Abstract
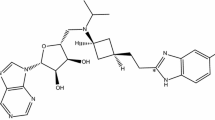
Izencitinib is a novel, orally administered pan-JAK inhibitor designed as a gut-selective therapy that was under development for the treatment of inflammatory bowel disease. The objectives of this study were to define the mass balance, routes and rates of excretion, and metabolic fate of izencitinib after oral administration of [14C]-izencitinib in humans. Six healthy adult male subjects were administered a single 100 mg (~300 μCi) oral dose of [14C]-izencitinib. Fecal excretion was the dominant route of elimination with >90% of the administered dose recovered in the feces. As expected by design, plasma concentrations of total radioactivity and izencitinib were low with the mean terminal half-life of total radioactivity (138 h) exceeding that of izencitinib (32.4 h). Izencitinib represented approximately 17% of the total circulating radioactivity, suggesting the presence of multiple circulating plasma metabolites. However, no individual metabolite exceeded 10% of total drug-related material in plasma. The major metabolites in feces, M18 and M9, were found to have unusual structures that reflected the presence of a nucleophilic carbon center in the naphthyridine ring of izencitinib. Proposed mechanisms for the formation of these metabolites involved oxidation and rearrangement (M18) and a one-carbon addition, potentially occurring through reaction with endogenous formaldehyde. Given the gut-selective properties of izencitinib, it is proposed that these novel fecal metabolites are the most relevant for evaluating the impact of metabolism on the pharmacological and toxicological properties of izencitinib, and that the circulating plasma metabolite profile is of little consequence in the assessment of the safety characteristics of izencitinib metabolites.






Similar content being viewed by others
Abbreviations
- ADME:
-
absorption, distribution, metabolism, and excretion
- AMS:
-
accelerator mass spectrometry
- AUC:
-
area under the curve
- AUC0-96h :
-
area under the curve from time zero to 96 hours
- AUC0-t :
-
area under the curve from time zero to the last measured concentration
- CID:
-
collision induced dissociation
- Cmax :
-
maximum concentration
- CD:
-
Crohn’s disease
- FTIH:
-
first-time-in human
- GFP-STAT1:
-
green fluorescent protein-signal transducer and activator of transcription
- GI:
-
gastrointestinal
- h:
-
hour or hours
- HLM:
-
human liver microsomes
- HPLC:
-
high-performance liquid chromatography
- HPLC-UV/MS:
-
HPLC coupled with mass spectrometry and ultraviolet detection
- HRMS:
-
high resolution mass spectra
- IBD:
-
inflammatory bowel disease
- IC50 :
-
half maximal inhibitory concentration
- JAK:
-
Janus kinase
- Ki:
-
dissociation constant
- LC-MS/MS:
-
HPLC with tandem mass spectrometry LC-UV/AMS
- MHRA:
-
Medicines and Healthcare products Regulatory Agency
- MIST:
-
metabolites in safety testing
- MS3 :
-
third generation product ions mass spectrometry
- ND:
-
not detected
- 1H NMR:
-
nuclear magnetic resonance
- PDA:
-
photodiode array
- pIC50 :
-
negative log of IC50
- pKi:
-
negative log of Ki
- PK:
-
pharmacokinetics
- RP:
-
reverse phase
- t1/2 :
-
half life
- Tmax :
-
time of maximum concentration
- S.D.:
-
standard deviation
- SEM:
-
standard error of the mean
- UC:
-
ulcerative colitis
- UV:
-
ultraviolet
References
Sobczak M, Fabisiak A, Murawska N, Wesolowska E, Wierzbicka P, Wlazlowski M, et al. Current overview of extrinsic and intrinsic factors in etiology and progression of inflammatory bowel diseases. Pharm Rep. 2014;66:766–75. https://doi.org/10.1016/j.pharep.2014.04.005
Segal JP, LeBlanc JF, Hart AL. Ulcerative colitis: an update. Clin Med (Lond). 2021;21:135–9. https://doi.org/10.7861/clinmed.2021-0080
Du L, Ha C. Epidemiology and Pathogenesis of Ulcerative Colitis. Gastroenterol Clin North Am. 2020;49:643–54. https://doi.org/10.1016/j.gtc.2020.07.005
Petagna L, Antonelli A, Ganini C, Bellato V, Campanelli M, Divizia A, et al. Pathophysiology of Crohn’s disease inflammation and recurrence. Biol Direct. 2020;15:23. https://doi.org/10.1186/s13062-020-00280-5
Al-Bawardy B, Shivashankar R, Proctor DD. Novel and emerging therapies for inflammatory bowel disease. Front Pharm. 2021;12:651415. https://doi.org/10.3389/fphar.2021.651415
Santiago M, Stocker F, Ministro P, Goncalves R, Carvalho D, Portela F, et al. Incidence trends of inflammatory bowel disease in a Southern European Country: a mirror of the western world. Clin Transl Gastroenterol. 2022;13:e00481. https://doi.org/10.14309/ctg.0000000000000481
Boland BS, Sandborn WJ, Chang JT. Update on Janus kinase antagonists in inflammatory bowel disease. Gastroenterol Clin North Am. 2014;43:603–17. https://doi.org/10.1016/j.gtc.2014.05.011
Dudek P, Fabisiak A, Zatorski H, Malecka-Wojciesko E, Talar-Wojnarowska R. Efficacy, safety and future perspectives of JAK inhibitors in the IBD treatment. J Clin Med. 2021;10. https://doi.org/10.3390/jcm10235660
Harris C, Cummings JRF. JAK1 inhibition and inflammatory bowel disease. Rheumatology. 2021;60:ii45–ii51. https://doi.org/10.1093/rheumatology/keaa896
Cordes F, Foell D, Ding JN, Varga G, Bettenworth D. Differential regulation of JAK/STAT-signaling in patients with ulcerative colitis and Crohn’s disease. World J Gastroenterol. 2020;26:4055–75. https://doi.org/10.3748/wjg.v26.i28.4055
Meyer DM, Jesson MI, Li X, Elrick MM, Funckes-Shippy CL, Warner JD, et al. Anti-inflammatory activity and neutrophil reductions mediated by the JAK1/JAK3 inhibitor, CP-690,550, in rat adjuvant-induced arthritis. J Inflamm. 2010;7:41. https://doi.org/10.1186/1476-9255-7-41
Parmentier JM, Voss J, Graff C, Schwartz A, Argiriadi M, Friedman M. et al. In vitro and in vivo characterization of the JAK1 selectivity of upadacitinib (ABT-494). BMC Rheumatol. 2018;2:23. https://doi.org/10.1186/s41927-018-0031-x
FDA. Xeljanz Prescribing Information. 2018. https://www.accessdata.fda.gov/drugsatfda_docs/label/2018/203214s018lbl.pdf
EMA. Xeljanz Summary of Product Characteristics. 2018. https://www.ema.europa.eu/en/documents/product-information/xeljanz-epar-product-information_en.pdf
FDA. Rinvoq Prescribing Information. 2022. https://www.accessdata.fda.gov/drugsatfda_docs/label/2022/211675s007lbl.pdf
EMA. Rinvoq Summary of Product Characteristics. 2022. https://www.ema.europa.eu/en/documents/product-information/rinvoq-epar-product-information_en.pdf
MHRA. Rinvoq Summary of Product Characteristics. 2023. https://www.medicines.org.uk/emc/product/10972/smpc/print
Agrawal M, Kim ES, Colombel JF. JAK inhibitors safety in ulcerative colitis: practical implications. J Crohns Colitis. 2020;14:S755–S60. https://doi.org/10.1093/ecco-jcc/jjaa017
Sandborn WJ, Su C, Sands BE, D’Haens GR, Vermeire S, Schreiber S, et al. Tofacitinib as induction and maintenance therapy for ulcerative colitis. N Engl J Med. 2017;376:1723–36. https://doi.org/10.1056/NEJMoa1606910
Sandborn WJ, Panes J, Sands BE, Reinisch W, Su C, Lawendy N, et al. Venous thromboembolic events in the tofacitinib ulcerative colitis clinical development programme. Aliment Pharm Ther. 2019;50:1068–76. https://doi.org/10.1111/apt.15514
Sandborn WJ, Feagan BG, Loftus EV Jr., Peyrin-Biroulet L, Van Assche G, D’Haens G, et al. Efficacy and safety of upadacitinib in a randomized trial of patients with crohn’s disease. Gastroenterology. 2020;158:2123–38.e8. https://doi.org/10.1053/j.gastro.2020.01.047
Sandborn WJ, Ghosh S, Panes J, Schreiber S, D’Haens G, Tanida S, et al. Efficacy of upadacitinib in a randomized trial of patients with active ulcerative colitis. Gastroenterology. 2020;158:2139–49.e14. https://doi.org/10.1053/j.gastro.2020.02.030
Wyant T, Fedyk E, Abhyankar B. An overview of the mechanism of action of the monoclonal antibody vedolizumab. J Crohns Colitis. 2016;10:1437–44. https://doi.org/10.1093/ecco-jcc/jjw092
Colombel JF, Sands BE, Rutgeerts P, Sandborn W, Danese S, D’Haens G, et al. The safety of vedolizumab for ulcerative colitis and Crohn’s disease. Gut. 2017;66:839–51. https://doi.org/10.1136/gutjnl-2015-311079
Lukin D, Faleck D, Xu R, Zhang Y, Weiss A, Aniwan S, et al. Comparative safety and effectiveness of vedolizumab to tumor necrosis factor antagonist therapy for ulcerative colitis. Clin Gastroenterol Hepatol. 2022;20:126–35. https://doi.org/10.1016/j.cgh.2020.10.003
Loftus EV Jr., Colombel JF, Feagan BG, Vermeire S, Sandborn WJ, Sands BE, et al. Long-term efficacy of vedolizumab for ulcerative colitis. J Crohns Colitis. 2017;11:400–11. https://doi.org/10.1093/ecco-jcc/jjw177
Loftus EV Jr., Feagan BG, Panaccione R, Colombel JF, Sandborn WJ, Sands BE, et al. Long-term safety of vedolizumab for inflammatory bowel disease. Aliment Pharm Ther. 2020;52:1353–65. https://doi.org/10.1111/apt.16060
Vermeire S, Loftus EV Jr., Colombel JF, Feagan BG, Sandborn WJ, Sands BE, et al. Long-term efficacy of vedolizumab for crohn’s disease. J Crohns Colitis. 2017;11:412–24. https://doi.org/10.1093/ecco-jcc/jjw176
Beattie DT, Pulido-Rios MT, Shen F, Ho M, Situ E, Tsuruda PR, et al. Intestinally-restricted Janus Kinase inhibition: a potential approach to maximize the therapeutic index in inflammatory bowel disease therapy. J Inflamm. 2017;14:28. https://doi.org/10.1186/s12950-017-0175-2
Sandborn WJ, Nguyen DD, Beattie DT, Brassil P, Krey W, Woo J, et al. Development of Gut-Selective Pan-Janus Kinase Inhibitor TD-1473 for ulcerative colitis: a translational medicine programme. J Crohns Colitis. 2020;14:1202–13. https://doi.org/10.1093/ecco-jcc/jjaa049
Hardwick RN, Brassil P, Badagnani I, Perkins K, Obedencio GP, Kim AS, et al. Gut-Selective Design of Orally Administered Izencitinib (TD-1473) Limits Systemic Exposure and Effects of Janus Kinase Inhibition in Nonclinical Species. Toxicol Sci. 2022;186:323–37. https://doi.org/10.1093/toxsci/kfac002
Penner N, Xu L, Prakash C. Radiolabeled absorption, distribution, metabolism, and excretion studies in drug development: why, when, and how. Chem Res Toxicol. 2012;25:513–31. https://doi.org/10.1021/tx300050f
Spracklin DK, Chen D, Bergman AJ, Callegari E, Obach RS. Mini-review: comprehensive drug disposition knowledge generated in the modern human radiolabeled ADME study. CPT Pharmacomet Syst Pharm. 2020;9:428–34. https://doi.org/10.1002/psp4.12540
Young GC, Spracklin DK, James AD, Hvenegaard MG, Scarfe G, Wagner DS et al. Considerations for human ADME strategy and design paradigm Shift(s) - an industry white paper. Clin Pharmacol Ther. 2022. https://doi.org/10.1002/cpt.2691
FDA. Clinical Pharmacology Considerations for Human Radiolabeled Mass Balance Studies (Draft). 2022. https://www.fda.gov/regulatory-information/search-fda-guidance-documents/clinical-pharmacology-considerations-human-radiolabeled-mass-balance-studies
FDA. Safety Testing of Drug Metabolites Guidance for Industry. 2020 https://www.fda.gov/media/72279/download
ICH. ICH M3(R2) Guidance for industry: nonclinical safety studies for the conduct of human clinical trials and marketing authorization for pharmaceuticals. 2010. https://www.fda.gov/media/71542/download
ICH. ICH Guideline M12 on drug interaction studies (Step 2b, Draft). 2022. https://www.ema.europa.eu/en/documents/scientific-guideline/draft-ich-guideline-m12-drug-interaction-studies-step-2b_en.pdf
Bourdet DL, Yeola S, Hegde SS, Colson PJ, Barnes CN, Borin MT. Revefenacin absorption, metabolism, and excretion in healthy subjects and pharmacological activity of its major metabolite. Drug Metab Dispos. 2020;48:1312–20. https://doi.org/10.1124/dmd.120.000103
Hamilton RA, Garnett WR, Kline BJ. Determination of mean valproic acid serum level by assay of a single pooled sample. Clin Pharm Ther. 1981;29:408–13. https://doi.org/10.1038/clpt.1981.56
Penner N, Klunk LJ, Prakash C. Human radiolabeled mass balance studies: objectives, utilities and limitations. Biopharm Drug Dispos. 2009;30:185–203. https://doi.org/10.1002/bdd.661
Cheng Y, Prusoff WH. Relationship between the inhibition constant (K1) and the concentration of inhibitor which causes 50 per cent inhibition (I50) of an enzymatic reaction. Biochem Pharmacol. 1973;22:3099–108. https://doi.org/10.1016/0006-2952(73)90196-2
Martin S, Lenz EM, Temesi D, Wild M, Clench MR. Reaction of homopiperazine with endogenous formaldehyde: a carbon hydrogen addition metabolite/product identified in rat urine and blood. Drug Metab Dispos. 2012;40:1478–86. https://doi.org/10.1124/dmd.112.044917
Zhang M, Resuello CM, Guo J, Powell ME, Elmore CS, Hu J, et al. Contribution of artifacts to N-methylated piperazine cyanide adduct formation in vitro from N-alkyl piperazine analogs. Drug Metab Dispos. 2013;41:1023–34. https://doi.org/10.1124/dmd.112.050450
Wei Y, Wang M, Liu H, Niu Y, Wang S, Zhang F, et al. Simultaneous determination of seven endogenous aldehydes in human blood by headspace gas chromatography-mass spectrometry. J Chromatogr B Anal Technol Biomed Life Sci. 2019;1118-1119:85–92. https://doi.org/10.1016/j.jchromb.2019.04.027
Luo W, Li H, Zhang Y, Ang CY. Determination of formaldehyde in blood plasma by high-performance liquid chromatography with fluorescence detection. J Chromatogr B Biomed Sci Appl. 2001;753:253–7. https://doi.org/10.1016/s0378-4347(00)00552-1
European Food Safety Authority, 2014. Endogenous formaldehyde turnover in humans compared with exogenous contribution from food sources. EFSA J. 2014;12:3550. https://doi.org/10.2903/j.efsa.2014.3550
Peppercorn MA. Sulfasalazine. pharmacology, clinical use, toxicity, and related new drug development. Ann Intern Med. 1984;101:377–86. https://doi.org/10.7326/0003-4819-101-3-377
Wilson ID, Nicholson JK. Gut microbiome interactions with drug metabolism, efficacy, and toxicity. Transl Res. 2017;179:204–22. https://doi.org/10.1016/j.trsl.2016.08.002
Acknowledgements
The authors and Theravance Biopharma thank the subjects for their participation in the clinical trial, and the principal investigator and clinical site staff at Celerion, Inc. in Lincoln, Nebraska. The study was funded by Theravance Biopharma Ireland Limited.
Author information
Authors and Affiliations
Contributions
IB, GPO, MR, and DLB are employees of Theravance Biopharma US, Inc., and shareholders in Theravance Biopharma, Inc. MTB, WLF, TAB are paid consultants for Theravance Biopharma US, Inc. SY, NLS, and XH are former employees of Theravance Biopharma US, Inc., and may hold shares in Theravance Biopharma, Inc.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Yeola, S., Badagnani, I., Huang, X. et al. The metabolic fate of izencitinib, a gut-selective pan-JAK inhibitor, in humans. Identification of unusual fecal metabolites and implications for MIST evaluation. Med Chem Res 32, 2071–2088 (2023). https://doi.org/10.1007/s00044-023-03143-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00044-023-03143-6