Abstract
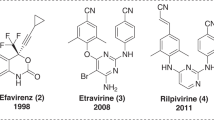
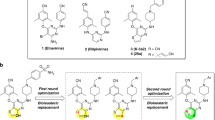
Non-nucleoside reverse transcriptase inhibitors have a definitive role and most commonly used in treatment of HIV-1 infection. A new series of 4-ethylidene/substituted-benzylidene-1-(4-hydroxy/chloro-6-methylpyrimidin-2-yl)-2-ethyl/phenyl-1H-imidazol-5(4H)-one were designed, synthesized, and evaluated for HIV-1 reverse transcriptase (RT) inhibitory activity. The results of in-vitro HIV-1 RT assay showed that some of the new compounds, such as 4c, 4d, 4e, 5a, and 5e effectively inhibit HIV-1 RT activity. 1-(4-Chloro-6-methylpyrimidin-2-yl)-4-(furan-2-ylmethylene)-2-methyl-1H-imidazol-5(4H)-one (5e) exerted most potent in-vitro HIV-1 RT inhibitory activity, among the group of compounds. Molecular docking studies were carried out to explore the binding affinity of imidazole-5-one analogs in active site of HIV-1 RT enzyme.




Similar content being viewed by others
References
Balzarini J (2004) Current status of the non-nucleoside reverse transcriptase inhibitors of human immunodeficiency virus type 1. Curr Top Med Chem 4:921–944
Balzarini J, De Clercq E (1998) Biochemical pharmacology of nucleoside and nonnucleoside analogues active against HIV reverse transcriptase. In: Bartlett JG, Bolognesi DP, Merigan TC (eds) Textbook of AIDS medicine, 2nd edn. Williams & Wilkins, Baltimore
Friesner RA, Murphy RB, Repasky MP, Frye LL, Greenwood JR, Halgren TA, Sanschagrin PC, Mainz DT (2006) Extra precision glide: docking and scoring incorporating a model of hydrophobic enclosure for protein–ligand complexes. J Med Chem 49:6177–6196
Furnis BS, Hannaford AJ, Smith PWG (2005) Vogel’s textbook of practical organic chemistry, 5th edn. Pearson Publication, London, p 1156
Mokale SN, Lokwani D, Shinde DB (2012) Synthesis, biological activity and docking study of imidazol-5-one as novel non-nucleoside HIV-1 reverse transcriptase inhibitors. Bioorg Med Chem 20:3119–3127
Pauwels R (2004) New non-nucleoside reverse transcriptase inhibitors (NNRTIs) in development for the treatment of HIV infections. Curr Opin Pharmacol 4:437–446
Rao A, Balzarini J, Carbone A, Chimmirri A, De Clercq E, Monforte AM, Monforte P, Pannecouque C, Zappalà M (2004a) 2-(2,6-Dihalophenyl)-3-(pyrimidin-2-yl)-1,3-thiazolidin-4-ones as non-nucleoside HIV-1 reverse transcriptase inhibitors. Antiviral Res 63:79–84
Rao A, Balzarini J, Carbone A, Chimmirri A, De Clercq E, Monforte AM, Monforte A, Pannecouque C, Zappala M (2004b) Synthesis of new 2,3-diaryl-1,3-thiazolidin-4-ones as anti-HIV agents. Farmaco 59:33–39
Rawal RK, Tripathi R, Katti SB, Pannecouquec C, De Clercqc E (2007) Synthesis and evaluation of 2-(2,6-dihalophenyl)-3-pyrimidinyl-1,3-thiazolidin-4-one analogues as anti-HIV-1 agents. Bioorg Med Chem 15:3134–3142
Acknowledgments
The authors are thankful to the Head, Department of Chemical Technology, Dr. Babasaheb Ambedkar Marathwada University, Aurangabad 431004 (MS), India for providing the laboratory facility.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Mokale, S.N., Lokwani, D.K. & Shinde, D.B. Synthesis, in-vitro reverse transcriptase inhibitory activity and docking study of some new imidazol-5-one analogs. Med Chem Res 23, 3752–3764 (2014). https://doi.org/10.1007/s00044-014-0954-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00044-014-0954-8