Abstract
The inner-alpine dry valleys of the Swiss Alps are characterized by subcontinental climate, leading to many peculiarities in dry grassland species composition. Despite their well-known uniqueness, comprehensive studies on biodiversity patterns of the dry grasslands in these valleys were still missing. To close this gap, we sampled 161 10-m2 vegetation plots in the Rhône, Rhine and Inn valleys, recording vascular plants, terricolous bryophyte and lichen species, as well as environmental data. Additionally, we tested the scale-dependence of environmental drivers using 34 nested-plot series with seven grain sizes (0.0001–100 m2). We analysed the effects of environmental drivers related to productivity/stress, disturbance and within-plot heterogeneity on species richness. Mean species richness ranged from 2.3 species in 0.0001 m2 to 58.8 species in 100 m2. For all taxa combined, the most relevant drivers at the grain size of 10 m2 were southing (negative), litter (negative), mean annual precipitation (unimodal), gravel cover (negative), inclination (unimodal) and mean annual precipitation (unimodal). For vascular plants the pattern was similar, while bryophyte and lichen richness differed by the opposite relationship to mean annual precipitation as well as negative influences of mean herb layer height, grazing and mowing. The explained variance of the multiple regression model increased with grain size, with very low values for the smallest two grain sizes. While southing and litter had high importance for the fiver larger grain sizes, pH and gravel cover were particularly important at the intermediate grain sizes, and inclination and mean annual precipitation for the two largest grain sizes. The findings emphasize the importance of taxonomic group and grain size for patterns and drivers of species richness in vegetation, consistent with ecological theory. Differences in the diversity–environment relationships among the three taxonomic groups can partly be explained by asymmetric competition that leads to low bryophyte and lichen diversity where vascular plants do well and vice versa. The relatively low alpha diversity of vascular plants in dry grasslands in Swiss inner-alpine valleys compared to similar communities in other parts of the Palaearctic remains puzzling, especially because Swiss stands are often large and well-preserved.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The xerothermic vegetation of the central valleys of the Alps has long attracted the interest of botanists (Christ 1879; Braun-Blanquet and Richard 1950; Braun-Blanquet 1961; Schwabe and Kratochwil 2004; Dengler et al. 2019b). The macroclimate of these valleys strongly deviates from their surroundings, as they are situated between the barriers of high mountain systems which largely block rain-carrying clouds. These valleys, therefore, constitute dry islands within the generally precipitation-rich European Alps. Together with the partly very low valley bottoms (sometimes only a few hundred metres above sea level), this leads to a relatively warm and dry climate, in strong contrast to the surrounding mountains (Braun-Blanquet 1961; Dengler et al. 2020c).
The climate, topography and isolated position lead to many peculiarities in flora and vegetation, partly resembling the Eastern European steppe vegetation. Apart from xerothermic forests and shrublands, different types of dry grasslands are a dominating feature of these landscapes (Braun-Blanquet 1961). These dry grasslands comprise diverse vegetation types, some growing on rocky outcrops with very shallow soils, others on slightly deeper soils. Below the alpine zone, most of these dry grasslands belong to semi-natural grasslands originating from centuries of low-intensity agriculture. Natural grasslands are rare and restricted to sites where forest growth is limited by particular soil or topographic conditions, e.g. rocky slopes with skeletal soils (Braun-Blanquet 1961; Boch et al. 2020; Dengler et al. 2020c). In the central valleys of the Alps, widespread Central European dry grassland species meet with steppic species whose main distribution is in the central part of Eurasia. In addition, dealpine, submediterranean and Southwest European xerothermic species occur together with a few narrow endemics of the Alps (Becherer 1972; Wohlgemuth 1996; Dengler et al. 2019a).
While dry grasslands in most other parts of Europe have been intensively studied during recent decades with respect to biodiversity, ecology and conservation (see reviews by Dengler et al. 2014; 2020c, Dengler and Tischew 2018; Boch et al. 2020), comprehensive studies on diversity patterns in dry grasslands along environmental gradients from the Swiss Rhône, Rhine and Inn valleys are missing (but see Boch et al. 2019a, 2021; Dengler et al. 2019b). Older literature mainly focussed on vegetation classification and the floristic and ecological characterization of dry grasslands (e.g. Christ 1879; Frey 1934; Braun-Blanquet 1961; Schwabe and Kratochwil 2004). However, knowledge on the distribution, ecology and biodiversity status of these dry grasslands is critical for developing management and conservation strategies. In Switzerland, this is of particular importance, as dry grasslands are among the most threatened vegetation types (Delarze et al. 2016). It has been estimated that about 95% of the dry grassland area in Switzerland has been lost since 1900, mainly due to land-use intensification or abandonment (Lachat et al. 2010), and habitat quality is still decreasing (Boch et al. 2019b). Consequently, 35% of about 350 dry grassland vascular plant species in Switzerland are currently considered threatened (Bornand et al. 2016).
Species richness in plant communities is controlled by a multitude of drivers (Grace 1999) which act simultaneously. Applying the concepts of Grime (1973), potential drivers can be grouped into those related to the stress–productivity axis and those related to the disturbance axis (Huston 2014), both of which are strongly modified by changing land-use systems and intensity (Allan et al. 2014). Generally, maximum fine-grain richness should be expected at intermediate levels of productivity and disturbance (Grime 1973; Huston 2014). However, what can be considered intermediate productivity depends on the level of disturbance and what can be considered intermediate disturbance depends on the level of productivity; thus, looking at the variables individually might yield positive, unimodal or negative relationships (Huston 2014). More recently, the heterogeneity of either productivity- or disturbance-related factors became a focus of diversity theory as a variable that nearly universally increases species richness (Tamme et al. 2010; Stein et al. 2014). Previous studies in Palaearctic grasslands found that specific drivers might be particularly relevant in one region but not in others (reviewed by Dengler et al. 2014), thus demonstrating the need to study such relationships in multiple regions and consider a large set of different drivers to achieve a more systematic understanding.
Most of the studies dealing with drivers of species diversity in grasslands focused on vascular plants (see review in Dengler et al. 2014), while the knowledge for bryophytes and lichens is still fragmentary. However, these two taxonomic groups often constitute a large fraction of the overall species diversity in dry grassland vegetation. Particularly in rocky and sandy dry grasslands, the fraction of non-vascular taxa can be substantial, sometimes exceeding the number of vascular plant species (Löbel and Dengler 2008; Dengler et al. 2020b). Some studies found that bryophyte diversity is positively related to vascular plant diversity (Löbel et al. 2006; Müller et al. 2019) and to that of several invertebrate taxa, and even has the strongest relation with the diversity of a wide range of other taxa (“multidiversity”; Manning et al. 2015). However, studies investigating vascular plants, bryophytes and lichens simultaneously are rare (Löbel et al. 2006; Turtureanu et al. 2014; Zulka et al. 2014). This lack of multi-taxon studies in vegetation is problematic, as bryophytes and lichens are known to react sensitively to different environmental conditions, such as soil pH (Löbel et al. 2006), and nutrient supply (Boch et al. 2018b), vascular plant cover (Löbel et al. 2006; Boch et al. 2016, 2018a) and biomass (van Klink et al. 2017; Boch et al. 2018b). Therefore, it is important to understand how bryophyte and lichen richness in vegetation types is related to a wide array of potential drivers.
The inconclusive results about the effects of different drivers on species richness might partly be due to different grain sizes that have been studied. It has been proposed theoretically (Shmida and Wilson 1985) and later shown in several meta-analyses that spatial scale can influence the relative importance (Field et al. 2009; Siefert et al. 2012) or even the direction of the impact of certain drivers (Tamme et al. 2010). Studies with nested-plot data of dry grasslands in different Palaearctic regions have confirmed strong scale effects (Turtureanu et al. 2014; Kuzemko et al. 2016; Polyakova et al. 2016; Dembicz et al. 2021a). In agreement with theory and the mentioned meta-analyses, these studies found the strongest effects of soil variables mostly at the smallest grain size and a prevalence of climate variables at larger grain sizes, but there were also many regional differences. Multi-scale sampling does not only allow an analysis of the drivers of α-diversity at different grain sizes, but also the application of species-area relationships (SARs) to study fine-grain β-diversity. SARs have been modelled with many different functions, with the power law (S = c Az with S = species richness, A = area, c and z modelled parameters) prevailing (Arrhenius 1921; Martín and Goldenfeld 2006; Dengler 2009). Recently it has been shown with an extensive dataset of nested-plot data from open vegetation types across the Palaearctic biogeographic realm that this function is generally the best SAR model also at fine grains in continuous habitats (Dengler et al. 2020a). The exponent z can then be used as a valid measure of β-diversity (Koleff et al. 2003; Jurasinski et al. 2009; Polyakova et al. 2016; Dembicz et al. 2021b). However, previous studies did not yield consistent results on the drivers of z values or the question whether z values are completely scale-invariant. Some studies found a slight peak of local z values for grain sizes around 0.01–0.1 m2 (Turtureanu et al. 2014; Polyakova et al. 2016), while others did not find any scale dependence (Kuzemko et al. 2016; Dembicz et al. 2021a).
To shed light on these open points, the Eurasian Dry Grassland Group conducted an international research expedition to the dry grasslands of the three valley systems of the Swiss Alps with pronounced continental climate (Dengler et al. 2020b). Combined with some other datasets using the same methodology, these data were then used to address the following questions: (i) How does species richness across scales in these grasslands compare to that of other grassland habitats in the region and elsewhere? (ii) Which structural and environmental drivers are most important for the biodiversity patterns across taxonomic groups and spatial scales? (iii) Which factors drive fine-grain β-diversity (z values), and is there a scale-dependency of local z values?
Methods
Study system
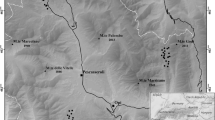
We selected 27 study sites along the inneralpine valley systems of Switzerland within the river catchments of the Rhône (14), Rhine (5) and Inn (8), the numbers reflecting the different spatial extents of dry grasslands in these catchments (Online Resource 1). The sites extended from 6.98° N – 10.38° N and 46.12° E – 46.98° E and covered an elevational gradient from 511 to 1574 m a.s.l. (Fig. 1). Bedrock composition was diverse, including limestone, granite, metamorphic rocks (gneiss, amphibolite), flysch, moraine and alluvial deposits, with base-rich substrata prevailing overall. Regarding climate, the Rhône valley is the driest and most continental, followed by the Inn valley, while the Rhine valley is the least continental. Mean annual precipitation varies considerably, from 670 to 1,345 mm (DaymetCH database, see below). Most sites are legally protected grasslands of national importance (Eggenberg et al. 2001; see Online Resource 1). Within a site, plots were selected to capture the existing diversity of ecologically and physiognomically varying dry grassland types (e.g. meso-xeric vs. xeric, skeleton-rich vs. fine-soil rich, north-facing vs. south-facing slopes). The studied communities belong to the vegetation classes Festuco-Brometea and Sedo-Scleranthetea (Dengler et al. 2020b).
Field sampling and lab measurements
We primarily used so-called “EDGG Biodiversity Plots” (n = 34; see Online Resource 1) to address the scale-dependence of plant diversity (Dengler et al. 2016). These are square plots of 100 m2, with two nested subplot series of 0.0001, 0.001, 0.01, 0.1, 1 and 10-m2 plots in two opposite corners of the largest plot. In addition, we sampled 93 10-m2 plots, resulting in a total of 161 10 m2 plots. Of these, 107 were located in the Rhône catchment, 23 in the Rhine catchment and 31 in the Inn catchment (Online Resource 1). Most of the plots (n = 139) were sampled during the 12th Field Workshop of the Eurasian Dry Grassland Group (EDGG; www.edgg.org) in 2019 (Dengler et al. 2020b), while a smaller proportion stems from sampling in the village Ausserberg in Valais in 2018 (published in Dengler et al. 2019b) and from a few other occasions in the years 2019–2020. Sampling was conducted in the months of May (n = 137), June (n = 8) and September (n = 16), i.e. at phenological stages at which experienced botanists could recognize more or less the complete species composition. To test whether sampling time could have introduced a bias, we compared total species richness (all taxonomic groups) of the 10-m2 plots sampled in May with those sampled later in the year, but the means were nearly identical (35.0 in May vs. 34.4 in later months).
In each plot and subplot, all vascular plants, terricolous bryophytes and lichens with shoot-presence in the plots were recorded. Most bryophyte and lichen species were collected and identified in the laboratory. At the grain size of 10 m2, the percent cover of each species was estimated, and we recorded the following structural and environmental parameters (Online Resource 2): mean annual temperature, mean annual precipitation, inclination, aspect, microrelief, herb layer height, soil pH, conductivity, soil depth, stone cover, litter cover, gravel cover, grazing and mowing. The coordinates and the elevation were determined using a handheld GPS. Aspect and inclination were measured in degrees using a compass, and inclinometer or a smartphone, respectively. For the analyses, we took the southing component of aspect, i.e. –cos (aspect), ranging from − 1 on northern to + 1 on southern slopes. The percent cover of the tree, shrub, herb and cryptogam layer was estimated, as well as the percent cover of all layers together (hereafter: ‘total vegetation’). In addition, the percent cover of abiotic layers like litter, dead wood, stone, gravel and fine soil was estimated. The vegetation height and soil depth were measured at five random points within each plot using a plastic disc and an iron pole, respectively. We then used mean vegetation height as a proxy for standing biomass and mean soil depth for the analysis. Further, we measured maximum microrelief perpendicular to a pole of 80 cm length placed on the soil surface where it showed the greatest difference in relief (Dengler et al. 2016). For soil property measures, soil samples of the uppermost 15 cm were taken at five random locations and then mixed. The soil samples were air-dried to measure soil pH and electrical conductivity with a multi-parameter probe (HANNA instruments HI 12,883, Woonsocket, Rhode Island, USA) in a suspension of 10 g soil and 25 ml distilled water. We assessed land use as the presence of grazing and mowing, respectively, both as binary variables by evaluating traces like faeces, signs of grazing or presence/absence of pasture weeds. Mean annual temperature and mean annual precipitation were derived from the DaymetCH dataset (D. Schmatz, WSL, Birmensdorf, unpublished, see https://www.wsl.ch/de/projekte/climate-data-portal.html#tabelement1-tab1, version of September 2021). This dataset of 100-m resolution for the period 1981–2010 was created with the interpolation software Daymet (Thornton et al. 1997), based on the daily measured values of all weather stations of MeteoSwiss and the digital elevation model DHM25 of SwissTopo. We decided against the widely used CHELSA climate dataset (Karger et al. 2017) because it underestimates the annual precipitation in our studied sites by on average nearly 200 mm as the comparison of DaymetCH and CHELSA data showed, while DaymetCH data were consistent with the climatic normal for the climate stations nearby. For the comprehensive list of variables, see Online Resource 1. The data are available from the GrassPlot database (dataset CH_D; Dengler and Tischew 2018, Dengler et al. 2018b; https://edgg.org/databases/GrassPlot).
Statistical analyses of species richness–environment relationships
In order to avoid multicollinearity, we first prepared a correlation matrix of all available metric predictor variables (environmental variables or proxies thereof) (Online Resource 3). Following the recommendation by Dormann et al. (2013), if two variables were highly correlated (Pearson’s │r│ ≥ 0.6), we always retained the ecologically more meaningful variable (Online Resource 3). The selection ended up with the following 14 predictors: southing, inclination, maximum microrelief, mean soil depth, mean herb layer height, litter cover, stone cover, gravel cover, grazing, mowing, soil pH, electrical conductivity, mean annual temperature and mean annual precipitation.
First, we modelled species richness at the 10-m2 grain size for vascular plants, bryophytes, lichens and all three groups combined. We checked if the models were improved by the addition of a quadratic term. The quadratic terms of inclination and stone cover were added to the full models as these strongly improved model performance (ΔAICc > 5 in the model with quadratic term compared to the one without). This led to a final selection of 17 predictor variables. We started by calculating generalised linear models (GLMs) with negative binomial distribution for each of the four richness variables using the MASS package (Venables and Ripley 2002). Spatial autocorrelation in the model residuals was tested using the Moran’s I test (Paradis and Schliep 2018). Only in the case of bryophyte richness did significant spatial autocorrelation occur. Thus, for this taxonomic group, we applied a generalised linear mixed-effect model (GLMM) with scaled values and plot ID nested in site ID as random factors. This random factor combination successfully removed spatial autocorrelation from the residuals. We then compared the GLMM to the corresponding negative binomial GLM using AICc values. We calculated the effect of the predictor variables on species richness by conducting a multimodel inference using the MuMIn package (Bartoń 2019). Using the dredge function, we generated a set of models with combinations of fixed effect terms. The relative importance value of the variables was derived as the sum of Akaike weights over all possible models containing the variable.
Additionally, we modelled total species richness for each of the seven grain sizes. Due to the smaller dataset size of the nested-plot series, in these models we dropped the four predictors with the lowest variable importance in the previous model (electrical conductivity, stone cover squared, grazing and mowing) and calculated a model for each grain size. No significant overdispersion or spatial autocorrelation occurred in these models, except for total species richness in three smallest grain sizes where we found overdispersion (thus the results of these models should be treated with caution). The effects of important predictors on species richness of taxonomic groups were visualised as predicted values based on the averaged models.
Analyses of species–area relationships and β-diversity
For each nested-plot series, we fitted a power-law species–area relationship (SAR) to the data of total species richness after averaging the richness values of the two subseries, using linear regression in double-log space. The slope parameter of the SAR (z value) can be used as a measure of fine-grain β-diversity (Jurasinski et al. 2009; Polyakova et al. 2016). When species richness equalled zero in smaller grain sizes, these grain sizes were not used in the regression of that nested-plot series. The dependence of z values on environmental predictors was then modelled similarly to the grain size models, but with a Gaussian error distribution.
To test for potential scale-dependence of z (Crawley and Harral 2001; Turtureanu et al 2014), we calculated “local z values” (Williamson 2003), i.e. the slopes of the SAR in double-log representation between two subsequent grain sizes (provided each had a richness > 0) for each of the 34 nested-plot series. These local z values of the six grain-size transitions were then compared with an ANOVA, taking series ID as error term, followed by Tukey’s post-hoc test.
Results
Species richness across scales
In total, we recorded 818 taxa, of which 629 were vascular plants (77%), 109 bryophytes (13%) and 80 lichens (10%). Mean total species richness ranged from 2.3 species in 0.0001 m2 to 58.8 species in 100 m2 (Table 1). Across all grain sizes, vascular plants had the highest mean species richness, followed by bryophytes and lichens. The maximum total species richness at 100-m2 scale was 113 species in a meso-xeric grassland in the Rhône catchment above the village of Ausserberg, from which 110 species were vascular plants and three bryophytes. The maximum richness at 100 m2 for bryophytes and lichens was 19 and 24 species, respectively (Table 1).
The three river catchments in comparison
The topographic, soil and land use conditions were rather similar in the three river catchments (Online Resource 4). Major differences only occurred for elevation and the elevation-related climatic variable mean annual temperature and mean annual precipitation (Online Resource 4). Mean annual temperature was highest for the plots in the Rhône catchment (8.3 °C), intermediate in the Rhine catchment (6.8 °C) and lowest in the Inn catchment (5.1 °C). With around 800 mm, mean annual precipitation was similar in the Rhône and Inn catchments, but less variable in the latter, while it was much higher in the Rhine catchment (1055 mm). At the 10-m2 scale, mean species richness values were very similar across the three catchments, e.g. for total species richness 34.0 species in the Rhône, 38.8 species in the Rhine and 36.0 species in the Inn catchment (Online Resource 4). There was no problematic autocorrelation for any taxonomic group that could be removed by adding catchment as a random factor to a GLMM (see Methods section), indicating that the small differences in richness patterns among the catchments were fully explained by the environmental predictors used in the regressions.
Diversity–environment relationships of different taxonomic groups
The variance of species richness explained by our multiple regression models varied from only 15% for lichens via 34% for vascular plants and 36% for all taxa to 46% (66% including the random factors) for bryophytes (Table 2). The most important predictors for richness of all taxa were southing (negative), litter cover (negative), mean annual precipitation (unimodal with peak around 1100 mm), gravel cover (negative), inclination (unimodal with peak around 40°), maximum microrelief (positive) and stone cover (unimodal with peak around 55%) (Table 2, Fig. 2). When comparing the three taxonomic groups, only a few of the tested predictors had a consistent direction of the effect across all three, namely litter (negative), gravel cover (negative) and electrical conductivity (negative, but of low importance in all groups) (Table 2, Fig. 2). As the most species-rich taxonomic group, vascular plants showed patterns largely consistent with those reported for all taxa, except that soil pH (negative), mean soil depth (positive), mowing (positive), and grazing (positive) were more important. The species richness model for bryophytes differed from that for vascular plants mainly in the two climatic variables (mean annual precipitation being unimportant, while mean annual temperature being important) and mean herb layer height, grazing and mowing all having a clear negative relationship, while that of stone cover was pronouncedly unimodal with a peak around 45% (Fig. 2). Lichens differed from the two other taxonomic groups by showing positive relationship with soil pH. Although inclination had a negative quadratic term for all taxa combined and separately (Table 2), an actual unimodal relationship occurred only for all taxa together and vascular plants, while the curve was monotonously increasing for bryophytes and monotonously decreasing for lichens (Fig. 2).
Effects of inclination, southing, stone cover, mean annual temperature and mean annual precipitation on total, vascular plant, bryophyte and lichen species richness from the full multiple regression models. Displayed are the original richness data and the predictions from general linear models and linear mixed-effect model. Each dot represents one or several 10-m2 plots (n = 161). Inclination, stone cover and mean annual precipitation had quadratic relationships (see Table 2)
Diversity–environment relationships across spatial scales
The importance of the analysed variables differed strongly and systematically across the seven spatial grain sizes (Fig. 3; Online Resource 5). Southing and litter cover had a strong negative impact on species richness at most grain sizes, whereas inclination was a strong positive predictor for 10- and 100-m2 plots. Mean annual precipitation had strong unimodal impact on richness in larger grains (1–100 m2). The smallest grain sizes (0.0001 m2 and 0.001 m2) had no predictor with a relative importance greater than 0.5. The explained variance of species richness was highest for 100-m2 plots (R2adj. = 0.60) and almost negligible for the two smallest grain sizes (Fig. 4, Online Resource 5).
Species–area relationships
The overall z values of all taxa ranged from 0.16 to 0.31 with a mean of 0.24 (Table 1). The strongest predictors for z values were mean annual precipitation (unimodal), mean annual temperature (positive), and inclination (u-shaped relationship; Online Resource 5). The explained variance of z values was relatively high compared to that of species richness at the different spatial scales (R2adj. = 0.60). The local z values for all taxa ranged from 0 to 0.602 with a mean of 0.235. Across the considered spatial grain sizes, they were highest for the transition from 0.01 to 0.1 m2 with significant differences across grain sizes (Tukey’s post-hoc test, p < 0.05; Fig. 5).
Discussion
Species richness at different scales
Comparing the vascular plant data from our nested-plot series to the average of Festuco-Brometea nested plots from across the Palaearctic with identical or very similar sampling approaches (shoot presence, seven standard grain sizes; values available from the GrassPlot Diversity Explorer, v.2.10 at https://edgg.org/databases/GrasslandDiversityExplorer, accessed on 5 April 2022; see Biurrun et al. 2021), the stands in the central valleys of Switzerland were poorer. This is true across all seven studied grain sizes, with differences ranging from 8% at 0.0001 m2 to 27% at 0.1 m2. At 10 m2, our average was 28.9 species vs. the Palaearctic average of 36.9 (n = 358). Below-average vascular plant species richness (in relation to other stands of the class) was previously also observed for the Aosta valley (35 species at 10 m2; Wiesner et al. 2015) and the inneralpine dry valleys of Austria (34.2 species at 10 m2; Magnes et al. 2021). This pattern was unexpected, as dry grasslands, at least in the Rhone valley, the Inn valley and the Aosta valley still cover extensive areas and appear well-conserved compared to other regions in Europe. Moreover, the isolated character of continental central valleys in mountain massifs cannot be causal, as two other EDGG Field Workshops in the Apennines (49.5 species in 10 m2; Filibeck et al. 2018) and the Caucasus (48.7 species in 10 m2; Aleksanyan et al. 2020) in rather comparable situations found above-average richness. Also, base-rich alpine grasslands in the Swiss Alps are systematically richer (median of approx. 50 species in 10 m2: Dengler et al. 2020d). Similarly, bryophytes were systematically poorer in species in our plots than the Palaearctic average, except in 10 m2 and 100 m2 (https://edgg.org/databases/GrasslandDiversityExplorer). By contrast, lichen species richness was clearly higher in the Swiss inneralpine stands of Festuco-Brometea than those elsewhere, albeit at a very low level (e.g. on average 2.2 species vs. 1.1 species in 10 m2; https://edgg.org/databases/GrasslandDiversityExplorer).
One explanation for this puzzling finding might be the biogeographical history of the species pool (Zobel 2016). The Alps were covered by a huge ice shield during the last glacial period, preventing any in-situ survival of plants with the exception of nunataks on mountain tops (Pan et al. 2020). While the species pool of alpine grasslands probably found sufficiently large refugia along the ice-free margins of the Alps (Tribsch and Schönswetter 2003), the majority of more thermophilous steppe species probably went extinct. Recolonisation from steppe areas further to the east seems to have played a rather minor role for the inner valleys of the Alps (Kirschner et al. 2020), resulting in a significantly smaller pool of steppe species in the Alps compared to other regions.
Differentiated by catchment, our mean vascular plant species richness data at 10 m2 were 28.8 (Rhône), 33.6 (Rhine) and 27.7 (Inn) for. This corresponds quite well to the data from the national monitoring program of the sites of national importance in Switzerland (WBS), with higher number of replicates that were sampled at optimum phenological stage, with 26.7 for the Western Central Alps (Rhône) and 34.6 for the Eastern Central Alps (Rhine and Inn) (Bergamini et al. 2019). This confirms that our sampling was mostly comprehensive. Compared to the Jura Mts. in Switzerland with 40.4 vascular plants in 10 m2, the plots in the inneralpine dry valleys are generally poorer in species, which might be attributed to the more pronounced summer drought that excludes various less drought-tolerant species form the grasslands.
Factors influencing diversity of taxonomic groups at 10 m2
We found pronounced differences in the diversity–environment relationship between all three taxonomic groups considered. This is consistent with results of previous comparative studies on the plot-scale richness of these groups (Löbel et al. 2006; Turtureanu et al. 2014; Kuzemko et al. 2016; Polyakova et al. 2016; Dembicz et al. 2021a) and can be explained by their different ecological requirements and life histories.
Our proxies for the productivity–stress axis of Grime (1973) mostly showed inconsistent patterns across the three taxonomic groups. Only litter cover, which reflects both productivity and absence of disturbance, was negative for all of them. The strongest predictor of this variable group was mean annual precipitation, showing unimodal relationships for vascular plants and total richness, with maxima around 1000–1100 mm—as expected along a stress-productivity gradient (Grime 1973): below that amount of precipitation apparently edaphically dry sites become so stressful that only few species can exist, while above the maximum conditions become so benign that competition increasingly excludes species from the system. By contrast, lichen species richness peaked at lower precipitation and bryophyte richness even showed an inverse pattern compared to the vascular plants (u-shaped), which both probably could be explained by asymmetric competition, suppressing a diverse cryptogam layer where vascular plants thrive. Mean annual temperature showed opposite patterns for vascular plants (slightly richer at higher elevation with cooler climate) and non-vascular plants (particularly rich in the warmer lowlands). Our findings of richness–climate relationships for vascular plants, via the strong negative correlation with elevation (r = − 0.99) indicate increasing richness with elevation, which differs from the predominantly reported mid-elevational peak (McCain and Grytnes 2010). A possible explanation is that we sampled not the full elevation gradient, but only the increasing part of the hump, and thus missed the decreasing part at higher elevations. This fits to the fact that plot-based studies in Swiss grasslands found richness peaks between 1100 and 1600 m a.s.l. (Descombes et al. 2017; Boch et al. 2019b), while our highest plot was at 1574 m a.s.l. A particularly strong negative predictor of total, vascular and bryophyte richness was southing, which can be explained by the more stressful conditions in south-facing slopes vs. flat or north-facing areas. This is consistent with the strong negative effect of heat load index (a composite variable mainly based on aspect) found in Transylvanian dry grasslands (Turtureanu et al. 2014). Mean soil pH, which is often a particularly relevant parameter for plot-scale species richness (e.g. Schuster and Diekmann 2003; Löbel et al. 2006; Boch et al. 2016, 2018a), typically with a unimodal response, in our case showed opposing effects for the three taxonomic groups: negative for vascular plants, weak for bryophytes and positive for lichens. This might be related to the fact that vascular plants prefer slightly more developed soils which coincides with decalcification, whereas lichens profit among the base-rich dry grasslands from the most extreme sites (highest pH) due to lower vascular plant competition. Last, mean herb layer height, which, in the way it was measured, reflects standing biomass and vegetation density, was strongly negative for the two non-vascular groups, but slightly positive for the vascular plants themselves. The strong negative effect of standing biomass of vascular plants on richness of the two other groups again could be explained by asymmetric competition for light (Löbel et al. 2006; Boch et al. 2016, 2018a, b).
According to the Intermediate Disturbance Hypothesis (IDH; Connell 1978; see also Grime 1973; Huston 2014), one should expect maximum richness at intermediate levels of disturbance. We considered here three proxies of disturbance as follows: slope inclination related to erosion, and the two land-use variables grazing and mowing. While the quadratic term of inclination was negative in all models, we found a peak inside the possible values only for total richness and vascular plants, where highest diversity occurred at around 40°, thus in agreement with the IDH. By contrast, lichens showed a monotonously decreasing and bryophytes a monotonously increasing relationship with slope angle. This does not conflict with the IDH per se, but highlights the taxon and context dependency of what can be considered “intermediate” (see Huston 2014). Grazing and mowing, which we had only as rough binary variables, showed contrasting effects on vascular plant richness (weakly positive) and on the richness of the two non-vascular groups (negative). Since farmers who are managing these legally protected grasslands of national importance must follow restrictions regarding mowing, stocking density and fertilization, land-use intensity in these low-productivity systems can be described as low to moderate. Thus, our plots rather reflect the increasing part of the IDH, which is supported in the case of vascular plants. However, for bryophyte and lichen richness, even this low land-use intensity obviously was already too much. This is in agreement with Allan et al. (2014), who found bryophyte and lichen diversity to be highest at very low land-use intensities and rapidly declining values already at moderate land-use intensity in German grasslands. In addition, Boch et al. (2018b) found strong negative direct effects of land-use intensity and indirect negative effects of land-use intensity via increased vascular plant biomass on bryophyte richness in German and Swiss grasslands. Increased vascular plant biomass can lead to competitive exclusion of bryophyte and lichens species and consequently lower species richness of these groups (Löbel et al. 2006; Boch et al. 2016, 2018b).
There is comprehensive evidence that any type of environmental heterogeneity across grain sizes increases species richness (Stein et al. 2014), and this relationship only might be reversed at very fine grain sizes far below 1 m2 (Tamme et al. 2010). However, for maximum microrelief as our most direct measure of within-plot heterogeneity, we found mixed effects: While it positively influenced the species richness of all taxa and of lichens, there were only weak positive effects for bryophytes and even negative ones for vascular plants. This contrasts with the strong positive effect of microrelief on plot-scale richness of vascular plants found in dry grasslands of Sweden and Siberia (Löbel et al. 2006; Polyakova et al. 2016). We do not have a good explanation why the Swiss inne-ralpine dry grasslands deviate here from similar plant communities elsewhere and from theoretical expectations. We also considered stone cover as a measure of heterogeneity, assuming highest heterogeneity and thus highest richness at intermediate levels of stone cover. However, while the relationships had a negative quadratic term for all three taxonomic groups, only in bryophytes showed a peak at intermediate stone covers, while vascular plants showed a monotonously decreasing and lichens a monotonously increasing curve (Fig. 2). Apparently, the areas with very shallow soils directly surrounding rocks and stones created spaces with low competition from vascular plants, beneficial for terricolous bryophytes and even more so for terricolous lichens, which overcompensated the loss of area by the stones themselves (as we did not record saxicolous species). On the other hand, for vascular plants, additional rocks and stones inside the plot just meant a loss of inhabitable area. Finally, we tentatively also listed gravel cover under heterogeneity-related factors. However, here we found a negative effect on all taxonomic groups, but particularly so for vascular plants. The difference of gravel vs. stones and rocks could be that the former through its smaller weight can be more easily moved by various agents (wind, water, trampling) and thus damage surrounding plants, particularly small ones. Moreover, gravel, if not embedded in a matrix of fine soil, has an extremely low water-holding capacity and thus could even exclude most species due to desiccation.
Diversity–environment relationships across spatial scales
We found that the explanatory power of our models for total richness was negligible for the two smallest grain sizes and then continuously increased towards 100 m2. Similarly, previous studies using the same method in dry grasslands of other regions (Kuzemko et al. 2016; Polyakova et al. 2016; Talebi et al. 2021; but see Dembicz et al. 2021a) found an increasing amount of explained variance with grain size. This makes sense for the following two reasons: (a) ecologically, one should expect species interactions (not covered by the measured environmental variables) to become more important towards finer grain sizes and thus lead to more variation and lower predictability, (b) methodologically, the scale mismatch increases also towards the smaller grain sizes as most predictors were measured for 10 m2 and the climate variables for even larger grains2.
While some predictors had a strong and consistent effect across the studied grain sizes (southing and litter cover), mean annual precipitation was important only for the three largest grain sizes, whereas gravel cover had high importance for the intermediate grain sizes only (0.01–1 m2) and soil pH even changed the sign of the impact from positive at smaller grain sizes to slightly negative at larger ones. These findings are in agreement with the general notion that diversity–environment relationships are strongly scale-dependent (Shmida and Wilson 1985; Field et al. 2009; Siefert et al. 2012). Specifically, the shift in the relative importance of soil factors (like pH and gravel cover) at smaller grain sizes to climatic variables (e.g. mean annual precipitation) at larger ones agrees with theoretical expectations (Shmida and Wilson 1985; Siefert et al. 2012). It is also largely consistent with the results from similar studies on dry grassland vegetation in other parts of the Palaearctic (Turtureanu et al. 2014; Kuzemko et al. 2016; Dembicz et al. 2021a; Talebi et al. 2021).
Species–area relationships
With a mean of 0.24, the z values found in the Swiss inner-alpine dry grasslands are intermediate. It exactly matches the mean value reported for more than 2000 nested-plot series of various grasslands and other open habitat types across the Palaearctic modelled in the same way (Dengler et al. 2020a: Supporting Information 9). Specifically for the two studied vegetation classes, Festuco-Brometea (large majority of plots) and Sedo-Scleranthetea, Dembicz et al. (2021c) report z values calculated in log-S space of 0.239 and 0.325, respectively. Thus, in contrast to α-diversity (see above), β-diversity (of which z values are a measure) is not outstanding in the region. Compared to other studies using the same method, z values were well explained by environmental variables, with an R2 of 0.60. The three most relevant predictors were mean annual precipitation (unimodal), mean annual temperature (positive) and inclination (negative). Maximum microrelief as our most direct measure of within-plot heterogeneity had a positive, but minor effect. These findings for z values of total species richness deviate quite strongly from the comprehensive study of Palaearctic open habitats (Dembicz et al. 2021b: Appendix S5) where the explained variance was low in general, and particularly low for the climate variables mean annual precipitation and mean annual temperature. Why these macroclimatic variables have such a strong influence on fine-grain beta diversity in our study system remains unclear for the time being.
Finally, when analysing the scale dependence of z values, we found a slight peak for the transition from 0.01 to 0.1 m2 with decreases towards both the smaller and the larger grain sizes. These findings correspond to those in regional studies of Turtureanu et al. (2014), Polyakova et al. (2016) and Talebi et al. (2021), while other studies did not find a scale dependence of z values (Kuzemko et al. 2016; Dembicz et al. 2021b). This demonstrates that the scale dependence of z values in dry grasslands of the Palaearctic is generally weak, but if there is one, it always exhibits a peak at grain sizes clearly below 1 m2, pointing to a very fine-grained community organisation. In a recent Palaearctic synthesis, Zhang et al. (2021) found the same when basing the calculation of z values on shoot presence as we did. From a methodological point of view this means that, except for very detailed studies, one can safely analyse dry grasslands using the “normal” power function with a constant z value, as Dengler et al. (2020a) already concluded for all open habitat types in the Palaearctic.
Conclusions and outlook
We found that patterns and drivers of species richness vary strongly between the three taxonomic groups (vascular plants, bryophytes and lichens) as well as across the seven studied grain sizes. This variation was mostly consistent with theoretical expectations and previous findings in other grassland types. Thus, our study contributes to an increasing knowledge of the often-neglected phenomena of taxon- and scale-dependence. It follows that one must be cautious, both in ecological research and biodiversity conservation, when transferring findings from other studies sampled with a different grain size or when considering one taxonomic group as surrogate for another. While the findings of this study largely confirmed our prior hypotheses, there were some unexpected deviations, e.g. the low importance of microrelief in the models and the high explained variance in the model for within-plot β-diversity (z values). Also, the low alpha diversity of vascular plants in Swiss inner-alpine valleys compared to dry grasslands in mountain valleys elsewhere in the Palaearctic remains puzzling. To understand the reasons for these unexpected patterns will require analysing alpha diversity of standard grain sizes with high-quality data across many regions in the Palaearctic simultaneously and including many potential drivers, an enterprise for which the steadily growing GrassPlot database (Dengler and Tischew 2018a, Dengler et al. 2018b; Biurrun et al. 2021) provides good opportunities.
Data availability
The vegetation-plot data underlying this study are stored in the GrassPlot database (https://edgg.org/databases/GrassPlot), from which they can be requested according to the GrassPlot Bylaws.
Code availability
The R code used for the analysis is available upon request from the first author.
References
Aleksanyan A, Biurrun I, Belonovskaya E et al (2020) Biodiversity of dry grasslands in Armenia: first results from the 13th EDGG Field Workshop in Armenia. Palaearct Grassl 46:12–51
Allan E, Bossdorf O, Dormann CF et al (2014) Inter-annual variation in land-use intensity enhances grassland multidiversity. Proc Natl Acad Sci USA 111:308–313
Arrhenius O (1921) Species and area. J Ecol 9:95–99
Bartoń K (2019) MuMIn: multi-model inference (R package version 1.43.6). https://CRAN.R-project.org/package=MuMIn
Becherer A (1972) Führer durch die Flora der Schweiz. Schwabe & Co, Basel
Bergamini A, Ginzler C, Schmidt BR et al (2019) Zustand und Entwicklung der Biotope von nationaler Bedeutung: Resultate 2011–2017 der Wirkungskontrolle Biotpschutz Schweiz. WSL Ber 85:1–104
Biurrun I, Pielech R, Dembicz I et al (2021) Benchmarking plant diversity of Palaearctic grasslands and other open habitats. J Veg Sci 32:e13050
Boch S, Prati D, Schöning I, Fischer M (2016) Lichen species richness is highest in non-intensively used grasslands promoting suitable microhabitats and low vascular plant competition. Biodivers Conserv 25:225–238
Boch S, Müller J, Prati D et al (2018a) Low-intensity management promotes bryophyte diversity in grasslands. Tuexenia 38:311–328
Boch S, Allan E, Humbert JY et al (2018b) Direct and indirect effects of land use on bryophytes in grasslands. Sci Tot Environ 644:60–67
Boch S, Bedolla A, Ecker KT et al (2019a) Threatened and specialist species suffer from increased wood cover and productivity in Swiss steppes. Flora 258:151444
Boch S, Bedolla A, Ecker KT et al (2019b) Mean indicator values suggest decreasing habitat quality in Swiss dry grasslands and are robust to relocation error. Tuexenia 39:315–334
Boch S, Biurrun I, Rodwell J (2020) Grasslands of Western Europe. In: Goldstein MI, DellaSala DA, DiPaolo DA (eds) Encyclopedia of the world’s biomes. Volume 3: Forests – trees of life. Grasslands and shrublands – sea of plants. Elsevier, Amsterdam, pp 678–688
Boch S, Kurtogullari Y, Allan E et al (2021) Effects of fertilization and irrigation on vascular plant species richness, functional composition and yield in mountain grasslands. J Environ Manag 279:111629
Bornand C, Gygax A, Juillerat P et al (2016) Rote Liste Gefässpflanzen. BAFU, Bern
Braun-Blanquet J (1961) Die inneralpine Trockenvegetation. Fischer, Stuttgart
Braun-Blanquet J, Richard R (1950) Groupement vegetaux et sols du bassin de Sierre. Bull Murithienne 66:106–134
Christ H (1879) Das Pflanzenleben der Schweiz. Schulthess, Zurich
Connell JH (1978) Diversity in tropical rain forests and coral reefs. Science 199:1302–1310
Crawley MJ, Harral JE (2001) Scale dependence in plant biodiversity. Science 291:864–868
Delarze R, Eggenberg S, Steiger P et al (2016) Rote Liste Lebensräume – Gefährdete Lebensräume der Schweiz 2016. BAFU, Bern
Dembicz I, Velev N, Boch S et al (2021a) Drivers of plant diversity in Bulgarian dry grasslands vary across spatial scales and functional-taxonomic groups. J Veg Sci 32(1):e12935
Dembicz I, Dengler J, Steinbauer MJ et al (2021b) Fine-grain beta diversity of Palaearctic grassland vegetation. J Veg Sci 32:e13045
Dembicz I, Dengler J, Gillet F et al (2021c) Fine-grain beta diversity in Palaearctic open vegetation: variability within and between biomes and vegetation types. Veg Classif Surv 2:293–304
Dengler J (2009) Which function describes the species-area relationship best? – a review and empirical evaluation. J Biogeogr 36:728–744
Dengler J, Tischew S (2018) Grasslands of Western and Northern Europe – between intensification and abandonment. In: Squires VR, Dengler J, Feng H et al (eds) Grasslands of the world: diversity, management and conservation. CRC Press, Boca Raton, pp 27–63
Dengler J, Janišová M, Török P et al (2014) Biodiversity of Palaearctic grasslands: a synthesis. Agric Ecosys Environ 182:1–14
Dengler J, Boch S, Filibeck G et al (2016) Assessing plant diversity and composition in grasslands across spatial scales: the standardised EDGG sampling methodology. Bull Eurasian Dry Grassl Group 32:13–30
Dengler J, Wagner V, Dembicz I et al (2018b) GrassPlot – a database of multi-scale plant diversity in Palaearctic grasslands. Phytocoenologia 48:331–347
Dengler J, Gehler J, Aleksanyan A et al (2019a) EDGG Field Workshops 2019 – the international research expeditions to study grassland diversity across multiple scales and taxa: Call for participation. Palaearct Grassl 41:9–22
Dengler J, Widmer S, Staubli E et al (2019b) Dry grasslands of the central valleys of the Alps from a European perspective: the example Ausserberg (Valais, Switzerland). Hacquetia 18:155–177
Dengler J, Matthews TJ, Steinbauer MJ et al (2020a) Species-area relationships in continuous vegetation: Evidence from Palaearctic grasslands. J Biogeogr 60:72–86
Dengler J, Guarino R, Moysiyenko I et al (2020b) On the trails of Josias Braun-Blanquet II: first results from the 12th EDGG Field Workshop studying the dry grasslands of the inneralpine dry valleys of Switzerland. Palaearct Grassl 45:59–88
Dengler J, Biurrun I, Boch S et al (2020c) Grasslands of the Palaearctic biogeographic realm: introduction and synthesis. In: Goldstein MI, DellaSala DA, DiPaolo DA. (eds) Encyclopedia of the world’s biomes. Volume 3: Forests – trees of life. Grasslands and shrublands – sea of plants. Elsevier, Amsterdam, pp 617–637
Dengler J, Cykowska-Marzencka B, Bruderer T et al (2020d) Sampling multi-scale and multi-taxon plant diversity data in the subalpine and alpine habitats of Switzerland: report on the 14th EDGG Field Workshop. Palaearct Grassl 47:14–42
Descombes P, Vittoz P, Guisan A, Pellissier L (2017) Uneven rate of plant turnover along elevation in grasslands. Alp Bot 127:53–63
Dormann CF, Elith J, Bacher S et al (2013) Collinearity: a review of methods to deal with it and a simulation study evaluating their performance. Ecography 36:27–46
Eggenberg S, Dalang T, Dipner M et al (2001) Kartierung und Bewertung der Trockenwiesen und -weiden von nationaler Bedeutung – Technischer Bericht. BUWAL, Bern
Field R, Hawkins BA, Cornell HV et al (2009) Spatial species-richness gradients across scales: a meta-analysis. J Biogeogr 36:132–147
Filibeck G, Cancellieri L, Sperandii MG et al (2018) Biodiversity patterns of dry grasslands in the Central Apennines (Italy) along a precipitation gradient: experiences from the 10th EDGG Field Workshop. Bull Euras Grassl Group 36:25–41
Frey H (1934) Die Walliser Felsensteppe. PhD thesis, Universität Zürich, Zürich
Grace JB (1999) The factors controlling species density in herbaceous plant communities: an assessment. Perspect Plant Ecol Evol Syst 2:1–28
Grime JP (1973) Control of species density in herbaceous vegetation. J Environ Manag 1:151–167
Huston MA (2014) Disturbance, productivity, and species diversity: empirism vs. logic in ecological theory. Ecology 95:2382–2396
Jurasinski G, Retzer V, Beierkuhnlein C (2009) Inventory, differentiation, and proportional diversity: a consistent terminology for quantifying species diversity. Oecologia 159:15–26
Karger DN, Conrad O, Böhner J et al (2017) Climatologies at high resolution for the earth’s land surface areas. Sci Data 4:170122
Kirschner P, Záveská E, Gamisch A et al (2020) Long-term isolation of European steppe outposts boosts the biome’s conservation value. Nat Comm 11:1–10
Koleff P, Gaston KJ, Lennon JJ (2003) Measuring β-diversity for presence-absence data. J Anim Ecol 72:367–382
Kuzemko AA, Steinbauer MJ, Becker T et al (2016) Patterns and drivers of phytodiversity of steppe grasslands of Central Podolia (Ukraine). Biodivers Conserv 25:2233–2250
Lachat T, Pauli D, Gonseth Y et al (eds) (2010) Wandel der Biodiversität in der Schweiz seit 1900. Ist die Talsohle erreicht? Haupt, Bern
Löbel S, Dengler J (2008) Dry grassland communities on southern Öland: phytosociology, ecology, and diversity. Acta Phytogeogr Suec 88:13–31
Löbel S, Dengler J, Hobohm C (2006) Species richness of vascular plants, bryophytes and lichens in dry grasslands: The effects of environment, landscape structure and competition. Folia Geobot 41:377–393
Magnes M, Willner W, Janišová M et al (2021) Xeric grasslands of the inner-alpine dry valleys of Austria – new insights into syntaxonomy, diversity and ecology. Veg Classif Surv 2:133–157
Manning P, Gossner MM, Bossdorf O et al (2015) Grassland management intensification weakens the associations among the diversities of multiple plant and animal taxa. Ecology 96:1492–1501
Martín HG, Goldenfeld N (2006) On the origin and robustness of the power-law species-area relationships in ecology. Proc Natl Acad Sci USA 103:10310–10315
McCain CM, Grytnes J (2010) Elevational gradients in species richness – encyclopedia of life sciences. John Wiley and Sons, Chichester. https://doi.org/10.1002/9780470015902.a0022548
Müller J, Boch S, Prati D et al (2019) Effects of forest management on bryophyte species richness in Central European forests. For Ecol Manag 432:850–859
Pan D, Hülber K, Willner W et al (2020) An explicit test of Pleistocene survival in peripheral versus nunatak refugia in two high mountain plant species. Mol Ecol 29:172–183
Paradis E, Schliep K (2018) ape 5.0: an environment for modern phylogenetics and evolutionary analyses in R. Bioinformatics 35:526–528
Polyakova MA, Dembicz I, Becker T et al (2016) Scale- and taxon-dependent patterns of plant diversity in steppes of Khakassia, South Siberia (Russia). Biodivers Conserv 25:2251–2273
Schuster B, Diekmann M (2003) Changes in species density along the soil pH gradient – evidence from German plant communities. Folia Geobot 38:367–379
Schwabe A, Kratochwil A (2004) Festucetalia valesiacae communities and xerothermic vegetation complexes in the Central Alps related to environmental factors. Phytocoenologia 34:329–446
Shmida A, Wilson MV (1985) Biological determinants of species diversity. J Biogeogr 12:1–20
Siefert A, Ravenscroft C, Althoff D et al (2012) Scale dependence of vegetation-environment relationships: a meta-analysis of multivariate data. J Veg Sci 23:942–951
Stein A, Gerstner K, Kreft H (2014) Environmental heterogeneity as a universal driver of species richness across taxa, biomes and spatial scale. Ecol Lett 17:866–880
Talebi A, Attar F, Naqinezhad A et al (2021) Scale-dependent patterns and rivers of plant diversity in the steppe grasslands of the Central Alborz Mts., Iran. J Veg Sci 32:e13005
Tamme R, Hiiesalu I, Laanisto L et al (2010) Environmental heterogeneity, species diversity and co-existence at different spatial scales. J Veg Sci 21:796–801
Thornton PE, Running SW, White MA (1997) Generating surfaces of daily meteorological variables over large regions of complex terrain. J Hydrol 190:214–251
Tribsch A, Schönswetter P (2003) Patterns of endemism and comparative phylogeography confirm palaeo-environmental evidence for Pleistocene refugia in the Eastern Alps. Taxon 52:477–497
Turtureanu PD, Palpurina S, Becker T et al (2014) Scale- and taxon-dependent biodiversity patterns of dry grassland vegetation in Transylvania (Romania). Agric Ecosyst Environ 182:15–24
van Klink R, Boch S, Buri P et al (2017) No detrimental effects of delayed mowing or uncut grass refuges on plant and bryophyte community structure and phytomass production in low-intensity hay meadows. Basic Appl Ecol 20:1–9
Venables WN, Ripley BD (2002) Modern applied statistics with S, 4th edn. Springer, New York
Wiesner L, Baumann E, Weiser F et al (2015) Scale-dependent species diversity in two contrasting dry grassland types of an inner alpine dry valley (Cogne, Aosta Valley, Italy). Bull Eurasian Dry Grassl Group 29:10–17
Williamson M (2003) Species-area relationships at small scales in continuum vegetation. J Ecol 91:904–907
Wohlgemuth T (1996) Ein floristischer Ansatz zur biogeographischen Gliederung der Schweiz. Bot Helv 106:227–260
Zhang J, Gillet F, Bartha S et al (2021) Scale dependence of species-area relationships is widespread but generally weak in Palaearctic grasslands. J Veg Sci 32:e13044
Zobel M (2016) The species pool concept as a framework for studying patterns of plant diversity. J Veg Sci 27:8–18
Zulka KP, Abensperg-Traun M, Milasowszky N et al (2014) Species richness in dry grassland patches in eastern Austria: a multi-taxon study on the role of local, landscape and habitat quality variables. Agric Ecosyst Environ 182:25–36
Acknowledgements
We thank Regula Billeter, Stefan Eggenberg, Daniel Hepenstrick, Martina Monigatti, Susanne Riedel and Eline Staubli as well as students of the Bachelor and CAS classes of JD for help with the field sampling. Christine Keller kindly determined some lichen specimens with thin-layer chromatography. Hallie Seiler edited the English language usage. D. Schmatz and K. Ecker (WSL, Birmensdorf) kindly provided the DaymetCH climate model for our study.
Funding
Open access funding provided by ZHAW Zurich University of Applied Sciences. The 12th EDGG Field Workshop, during which most of the data were sampled, received financial support from the Eurasian Dry Grassland Group (EDGG) and the International Association for Vegetation Science (IAVS).
Author information
Authors and Affiliations
Contributions
JD planned the research. All authors except MBe and SB were involved in the field sampling, ID identified the collected vascular plants, BC-M the bryophytes and SB the lichens, while MBe analysed the soils. MBe conducted the statistical analyses under the supervision of ID and JD. He also led the writing of the Methods and Results sections, while JD and SB drafted the Introduction and Discussion sections, and ID and WW gave major inputs. All authors checked, revised and approved the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
We declare no conflict of interest.
Ethical approval
The field work in protected areas was permitted by the conservation authorities of the cantons of Vaud, Valais and Grisons.
Consent to participate
Not applicable.
Consent for publication
All authors have approved the submitted manuscript.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Bergauer, M., Dembicz, I., Boch, S. et al. Scale-dependent patterns and drivers of vascular plant, bryophyte and lichen diversity in dry grasslands of the Swiss inneralpine valleys. Alp Botany 132, 195–209 (2022). https://doi.org/10.1007/s00035-022-00285-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00035-022-00285-y