Abstract
The expansion of oil palm and cattle grazing in the tropics continues to alter numerous ecosystem functions. The generated land-use change is potentially impacting stream leaf-litter breakdown, a fundamental process for freshwater ecosystems. To assess the effect of land-use change, we studied breakdown rates of forest (Pachira aquatica, Pouroma aspera, Sloanea ampla, and Hippocratea volubilis) and oil palm (Elaeis guineensis) leaves after a 26-day immersion in streams surrounded by rainforests, grazing lands, or oil palm plantations with and without riparian buffers. In addition, we assessed breakdown drivers by deploying litter bags (248) of two mesh sizes (15 mm and 0.5 mm) allowing or restricting macroinvertebrates’ access (134 coarse-mesh bags and 114 fine-mesh bags). Forest leaf breakdown by microbes (fine-mesh) was lower by 55% in the plantations compared to forests; while microbial oil palm-leaf breakdown was lower by 32% in the unbuffered plantations compared to forests (p < 0.05). Total litter breakdown was lower (p > 0.05) in the plantations but not when these preserved riparian buffers. Litter breakdown was driven primarily by microbes in all land uses except in the buffered plantations, possibly due to increased shredder biomass. These results suggest that oil palm agriculture may decrease microbial stream leaf-litter breakdown, especially in streams with no riparian buffers.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
The global agricultural expansion has finally slowed down, from 4854 million ha in 2000 to less than 4832 million ha in recent years (Klein Goldewijk et al. 2017). However, farmlands still occupy 38% of the planet’s ice-free surface and are responsible for nearly 90% of global deforestation (FAO 2022a), especially in numerous tropical countries (FAO 2022b) where farmlands expand at alarming rates, often at the expense of carbon-rich habitats such as tropical wet rainforests (FAO 2022a). Grazing lands, for example, replace 2.1 million hectares of tropical forest yearly (Ritchie and Roser 2021), and oil palm (Elaeis guineensis Jacq.) plantations are responsible for the loss of 10.5 million ha of tropical forest in the last two decades (Goldman et al. 2020). Furthermore, oil palm plantations are expected to expand even further as the demand for vegetable oil continues to increase (Meijaard et al. 2020). These land-use shifts severely damage biodiversity and ecosystem functioning (Laurance et al. 2014). Agriculture can affect the soil's physical, chemical, and/or biological properties along with many vital processes, including nutrient cycling, carbon sequestration, and litter breakdown and decomposition (Sanaullah et al. 2020). In this regard, stream ecosystems are of particular concern due to their direct connectivity to their surrounding terrestrial landscapes (Allan 2004) and their pivotal role in supporting numerous vulnerable and endemic species (Dudgeon et al. 2006; Burlakova et al. 2011). Streams are impacted by agriculture through inputs of nutrients, sediments, and contaminants, as well as physical changes in habitats and microclimate and the replacement or removal of riparian vegetation (Sala et al. 2000).
To mitigate some of the agricultural impacts on streams, a growing number of farmers in tropical countries are being incentivized to conserve riparian buffers in their lands via crop certification schemes (Luke et al. 2019). Riparian buffers are native vegetation strips, which can include trees, shrubs, or other perennial vegetation adjacent to streams and rivers. These can bring several benefits to terrestrial and freshwater ecosystems, including runoff filtering, habitat structure and connectivity conservation, and stabilization of eroding banks (Hickey and Doran 2004; Cole et al. 2020). Riparian buffers thus bring an overall positive effect on the aquatic and terrestrial biota (Deere et al. 2022) and ecosystem functioning (Luke et al. 2019). Nevertheless, there are several research gaps in the tropics regarding riparian buffers, as most research comes from Europe and North America (Luke et al. 2019; Hughes et al. 2021). Additionally, the policies for riparian buffers are often poorly defined or even absent in several of these countries (Luke et al. 2019), and to this day, numerous streams in tropical agricultural lands remain unbuffered (Rojas-Castillo et al. 2024).
Litter breakdown is among the most critical ecosystem processes in streams, as it is essential for energy acquisition, especially in shaded streams where leaves from the riparian vegetation constitute the most significant energy source (Gessner et al. 1999; Benfield et al. 2017). Litter breakdown is considered an adequate measure of the functional integrity of the streams, as it incorporates physical, chemical, and biological elements (Benfield et al. 2017). The process includes both abiotic and biotic mechanisms, as it involves the leaching (dissolution of labile compounds), microbial conditioning and consumption, macroinvertebrate fragmentation, and environmental abrasion of the leaf litter (Webster and Benfield 1986; Peralta-Maraver et al. 2019). In addition, the process constitutes a key step in the carbon cycle and reincorporation of the nutrients into the ecosystem (Battin et al. 2008; Darmawan et al. 2021).
Due to the dependency of leaf-litter breakdown on both abiotic and biotic features, the process is highly vulnerable to land-use change. Overall, fast litter breakdown and decomposition rates are indicators of pristine stream conditions (Young et al. 2008; Silva-Junior et al. 2014). However, other variables associated with land-use change may also impact litter breakdown in a complex way. Soil and water temperature tend to increase litter breakdown and decomposition (Gonçalves et al. 2013), as does organic pollution, and the presence of stable beds is also associated with increased breakdown rates (Silva-Junior et al. 2014). Oppositely, decreased litter breakdown rates are often observed in channelized streams, at reduced flow, low pH, or with high presence of mud, silt, fine particles, insecticides, and heavy metals (Silva-Junior et al. 2014). Riparian buffer removal may increase litter breakdown rates through increased temperatures, yet this could be offset by concurrent factors: a reduction in shredder macroinvertebrates biomass (Rojas-Castillo et al. 2023), an upsurge in fine particles and erosion (Silva-Junior et al. 2014), and decreased microbial richness (Chavarria et al. 2021), all prevalent in streams lacking riparian buffers.
Grazing lands for livestock in the neotropics have shown reduced litter breakdown compared to forests (Iñiguez-Armijos et al. 2016; Lemes da Silva et al. 2020). This reduction has been attributed to a loss of fungal biomass and shredders in impacted streams, increased water temperatures, phosphorus, and pH (Iñiguez‐Armijos et al. 2016), and decreased dissolved oxygen and water velocity (Lemes da Silva et al. 2020). Other studies have found no significant differences between grazing lands and forests even when forests were hosting a higher abundance and diversity of macroinvertebrates (Gutiérrez-López et al. 2016). The oil palm expansion may also decrease litter breakdown rates, since forest soil microbiota are more effective in litter breakdown compared to that of oil palm plantations (Elias et al. 2020). Yet, other studies show no differences between treatments (Foster et al. 2011) or even opposite results attributed to an increase in microbial activity due to the increased water temperature in plantations (Chellaiah and Yule 2018a).
Recent studies in streams in oil palm and grazing lands found significant decreases in microbial diversity and shredder macroinvertebrates together with increases in water temperature and changes in water quality (Rojas-Castillo 2023; Rojas-Castillo et al. 2023). In addition, oil palm streams showed a significant decrease in wood decomposers and fungal taxa richness (Rojas-Castillo 2023). All these changes could potentially affect leaf-litter breakdown. These land uses also modify the material entering the streams and thus the litter quality itself, which influences litter breakdown rates (Manzoni et al. 2010). Despite the foreseeable adverse effects on stream litter breakdown and decomposition by these land uses, few studies address the process, the ecological drivers, and the mitigation strategies, especially in oil palm plantations.
This study aims at contrasting leaf-litter breakdown rates of local and introduced leaf species in streams in oil palm plantations, grazing lands, and primary rainforests (control group). It also explores the mitigating effect of riparian buffers on stream leaf-litter breakdown in plantations. To do so, we measured leaf breakdown of four local forest species (Pachira aquatica, Pouroma aspera, Sloanea ampla, and Hippocratea volubilis) and oil palm (Elaeis guineensis) in fine (0.5 mm, access only to microbes) and coarse-mesh bags (15 mm, easy access to macroinvertebrates) after a 26-day stream immersion on each land use. We sought to answer two research questions: (i) How do land use and riparian buffers affect total, microbial, and macroinvertebrate-mediated leaf-litter breakdown in the streams? and (ii) What are the drivers of leaf-litter breakdown in the different land uses? Based on previous findings of higher biomass of shredders in dense-canopy streams (Rojas-Castillo et al. 2023) and the lower richness of microbes in open-canopy streams (Chavarria et al. 2021), we hypothesized that leaf-litter breakdown would be higher in rainforests and plantations with riparian buffers compared to grazing lands and oil palm plantations without buffers. However, we also hypothesized that the process would still be driven primarily by microbes while macroinvertebrates would only play a minor role, as shown in previous tropical studies (Chellaiah and Yule 2018b; Pérez et al. 2023).
Methods
Streams and study area
This study was conducted in the Lachuá region in Alta Verapaz, Guatemala. The region covers an area of 535 km2 which includes a protected area (Lachuá Lake National Park) of tropical moist broadleaf rainforests (145 km2) which is part of the last and northernmost tropical hotspot in the neotropics (Mendoza and Dirzo 1999; Granados 2001). Lachuá also encompasses several smaller forest patches (143 km2), human settlements (5 km2), and agriculture and grazing lands (115 km2) (Escuela de Biología 2004). The region lies within the Usumacinta basin, bounded to the north, west, and east by the Chixoy and Icbolay rivers, and to the south by the Sultana mountains. The area's physiographic context is defined by the Lacandón Fold Belt province and a karstic Upper Cretaceous geology. The region has a dominant seasonal climate with annual rainfall exceeding 2500 mm, an average 91% air humidity, and a 25 °C averaged temperature, experiencing two main seasons: a dry (February–April) and a rainy season (June–October) (CONAP 2003; Rojas-Castillo et al. 2023). The area possesses a number of aquatic ecosystems and floodplains, e.g., the 4-km2 and 220 m deep Lachuá Lagoon, and the Lachuá, El Altar, and Peyán rivers that are recognized under the Ramsar Convention as wetlands of national and international significance (Escuela de Biología 2004).
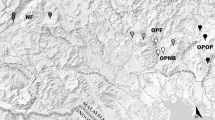
Our experiment was conducted in the 19 streams described by Rojas-Castillo et al. (2023) (Fig. 1), seven of which were located in rainforests (F), six in grazing lands (G), and six in oil palm plantations (three with a riparian buffer [P_RF] and three without [P]). The additional stream in the rainforest treatment was included as a precautionary measure to mitigate the risk of losing a sample unit due to potential drought in the smaller streams during the study period, as suggested by a park ranger. The canopy cover varies across these areas, with streams in grazing lands retaining a mean 14% cover, unbuffered streams in oil palms retaining 37% cover, while buffered streams in the plantations and forested streams retaining 81% and 82% cover, respectively. Notably, no significant differences were observed by Rojas-Castillo et al. (2023) across the streams within different land uses in terms of watershed area (mean = 46 ha, CI 22–71), width (mean = 1.69 m, CI 1.44–1.95), or depth (mean = 0.17 m, CI 0.15–0.19). For further insights into the study site and streams, refer to Rojas-Castillo et al. (2023).
Study area: stream network and land uses in catchment areas. Rainforest streams (F1–7), grazing land streams (G1–6), unbuffered streams in oil palm (P1–3), and buffered streams in oil palm (P4–6). Map from Google Earth (2021) edited in QGIS (Team QD 2015). Pictures on the right taken by Celia Diaz and Natalia Vargas, 2021
Leaf-litter breakdown
Using the litter bag method adapted from Boulton and Boon (1991), we measured stream leaf-litter breakdown during an immersion period of 26 days. We decided on this duration as previous studies have shown considerable weight loss after this period (Tschelaut et al. 2008; Blanco and Gutiérrez-Isaza 2014; Abelho and Descals 2019; Pérez et al. 2023). To quantify the role of macroinvertebrates and microbes in the litter breakdown, we used coarse-mesh (15 mm mesh size) and fine-mesh (0.5 mm) plastic bags (both 10 × 15 cm) either allowing or restricting the access of macroinvertebrates (adapted from Tiegs et al. 2009). In addition, we measured the effect of land use on introduced and local species by employing leaves from the exotic oil palm crop (Elaeis guineensis Jacq.) and four local forest species (Pachira aquatica Aubl., Pouroma aspera Trécul, Sloanea ampla I. M. Johnst., and Hippocratea volubilis L). We selected these local species based on their high abundance in riparian areas in the national park during the study period.
The introduced species, E. guineensis, is a widespread tropical palm tree crop (Choudhary and Grover 2019). It features tough (472 g/mm2 in penetrometer) compound pinnate leaves that are 3–5 m long and generally rich in nutrients (N = 2–2.6%, p = 0.12–0.22%, K = 0.88%, lignin = 25%, cellulose = 18%) (Chellaiah and Yule 2018a; Behera et al. 2022). The local forest species include three trees and one vine, commonly found in riparian ecosystems. Pachira aquatica is a fast-growing evergreen tree reaching heights of 15–30 m. Its palmate, leathery tough (443 g/mm2), and thick (0.16 mm) leaves are rich in antibiotics (Blanco and Gutiérrez-Isaza 2014; Rodrigues and Pastore 2021; Yoshikawa et al. 2022). Members of Pachira show moderate levels of foliar nitrogen (2.2%) and high foliar carbon (45%) (Werden et al. 2018). Pouroma is a genus of evergreen trees that prefers sunny conditions and it is characterized by hollow stems and palmately compound leaves (Berg et al. 1990). It belongs to the Cecropiaceae family whose members typically show moderately high foliar nitrogen content (2.5%) and phosphorus (0.78–0.91 mg g−1) and moderate to high levels of leaf toughness (364 g/mm2) (dos Santos et al. 2006; Blanco and Gutiérrez-Isaza 2014). Sloanea ampla is a medium-sized evergreen climax tree (15–18 m tall) from the Elaeocarpaceae family, endemic to the wet tropical biome in Central America and Mexico (Grobben 2022). It possesses alternate wide, ovoid, leathery leaves with wavy margins and pronounced venation. Members of this genus contain low foliar nitrogen (1.6%), high foliar carbon (50%), and leaf thickness of 0.18 mm (Umaña and Swenson 2019). Hippocratea is a climber vine with oblong-elliptic leaves, occasionally thin-coriaceous (Smith 1940) often found in riparian ecosystems and wetlands where they represent high litterfall productivity (up to 75%) (Infante Mata et al. 2012). Leaf material from this genus is high in lignin content and calcium oxalate crystals, and tends to decompose slowly (Gomes et al. 2005; Marín-Muñiz et al. 2014).
Forest leaves were picked up from the forest ground at the Lachuá Lake National Park, selecting only the freshly fallen brown ones and avoiding leaves with fungi colonization. These leaves were sourced from patches under one or two individual trees per species. In the oil palm plantations, we picked brown leaves that were cut 2 weeks before, and we then removed the leaflets from the rachis. We selected brown leaves over green ones to avoid the effect of defensive compounds which tend to be higher in green leaves (Burton et al. 2023). We collected the leaves in a large sack, separated by species, stored the leaves in hanging nets in a dry room for 1 week, air-dried them for 72 h (mean temperature 25–33 °C, max 40 °C), and cut and introduced 4–5 g of leaf material in each bag (one gram per each of the four forest species and 4–5 g of oil palms). We packed forest species together and oil palm leaves individually to recreate the natural conditions in which leaf litter is found in streams in forests and oil palm plantations. In total, we filled 247 plastic bags (133 coarse mesh and 114 fine mesh). In each stream, we placed 13 bags (seven coarse-mesh bags: three with oil palm and four with forest leaves; six fine-mesh bags: three with oil palm and three with forest leaves) separated by at least 10 m from each other. To ensure full immersion of the bags during the experiment, we secured them to the stream bed and placed them within pools and runs, deliberately avoiding riffles to prevent desiccation of the leaf litter (which could introduce additional confounding factors).
The litter bags were immersed in the streams in February 2021. We conducted the experiment during the dry season to minimize the risk of bag losses caused by flooding or mud covering and to catch the effect of the greater macroinvertebrate diversity found during this period compared to the rainy season (Pearson 2014; Mwaijengo et al. 2020). After their collection, the bags were immediately sealed in plastic Ziploc bags, preventing litter loss. These were then transported to the laboratory, where the leaves were carefully cleaned under water to remove macroinvertebrates and sediment particles. The leaves were then air-dried under the sun for 24 h and weighed.
Water quality and shredder biomass
To address water quality and shredder biomass, factors that can potentially affect leaf-litter breakdown (Silva-Junior et al. 2014), we used the water quality and macroinvertebrate biomass datasets compiled by Rojas-Castillo et al. (2023) for each of the study streams (conducted concurrently with our study). The water quality dataset included dissolved oxygen (measured with a handheld multiprobe, Model 6000, YSI, Yellow Springs, OH, USA), turbidity (measured with a Eutech-100 turbidity meter, Nijkerk, Netherlands), pH (measured with a pH meter ecoTestr pH 2), water temperature and light (measured by installing HOBO Pendant® MX2202 temperature/light data loggers for 30 days in the streams), conductivity (measured by installing HOBO U24 conductivity data loggers for 15 days), water level (measured by installing HOBO U20L-01 water level data loggers for 15 days), stream discharge and current velocity (measured by dilution gauging of 1 kg NaCl in 10 L of water/15 m transect) (White 1978). The macroinvertebrate dataset included the average biomass (WWg) of the different functional feeding groups (FFG) per stream, e.g., shredders. The average represented the macroinvertebrates collected in each stream, each calculated from six Surber-net samples (20 × 25 cm, mesh size 200 μm). These individuals were identified to genus level (Domínguez 2009; Springer et al. 2010; Hamada et al. 2018), were then classified into functional feeding group (FFG) (Oliveira and Nessimian 2010; Merritt et al. 2017; Pereira et al. 2021), and weighed with a Mettler Toledo AE50 analytical balance after a standard drying procedure (20 s in a paper towel) (Rojas-Castillo et al. 2023).
Statistical analysis
We calculated the leaf-litter breakdown coefficient (K) for each sample from the formula mf/mi = e−Kt where mf and mi are the final and initial litter mass (g), respectively, t is time in days, and K is the breakdown rate coefficient (Olson 1963). We obtained the total (K day−1 in coarse-mesh bags) and the microbial (K day−1 in fine-mesh bags) leaf-litter breakdown coefficients. Additionally, we calculated shredder and physical leaf-litter breakdown by subtracting the average microbial litter breakdown from the average overall litter breakdown per stream, as follows: (K day−1Total − K day−1microbial).
We explored the effect of land use, leaf litter, and mesh size on leaf-litter breakdown (K day−1) by contrasting different linear mixed-effect models (with and without interactions) performed in the nlme package (Pinheiro et al. 2022); these included the stream as a random effect following repeated measurements sampling (Forman 2019). We selected the best-fitting model based on the Akaike information criterion (AIC) and performed a marginal analysis of variance (ANOVA) on the best-fitting one employing the R stats package (R Core Team 2022). To address specific differences on the total and microbial breakdown between land uses, we performed post hoc Tukey tests on linear mixed-effect models for each leaf litter type in the multcomp package (Hothorn et al. 2008). For exploring land-use effect on shredder and physical leaf-litter breakdown, we performed ANOVAs on linear mixed-effect models including the stream as a random effect. For addressing specific differences across land uses, we ran ANOVAs on linear models for each type of leaf litter (rainforest and oil palm leaves), followed by Tukey analyses. We contrasted the average stream shredder biomass (WWg in 500 m2) from Rojas-Castillo et al. (2023) by performing an ANOVA followed by a post hoc Tukey test employing the stats package (R Core Team 2022). To explore the relation between macroinvertebrates, microbes, and litter breakdown, we performed a series of linear regressions (lm) between shredder biomass and total leaf-litter breakdown and between microbial leaf-litter breakdown and total leaf-litter breakdown employing the stats package (R Core Team 2022). Additionally, we compared Kc day−1 against Kf day−1 within each land use employing ANOVAs on linear mixed-effect models to quantify the importance of macroinvertebrates and physical factors on the total leaf-litter breakdown. Finally, we performed a series of linear regressions between the water and habitat quality parameters reported in Rojas-Castillo et al. (2023) and each stream's average litter breakdown (Kc day−1 and Kf day−1) to determine potential environmental factors associated with total and microbial leaf-litter breakdown. All statistical analyses were performed in R version 4.1.3 (R Core Team 2022) and RStudio version 2022.2.1.461 (RStudio Team 2022).
Results
Land use and riparian buffer effect on leaf-litter breakdown rates
During the 26 days of the study, forest leaves lost, on average, 6.4% (CI 5.6–7.1%) of their mass, reporting a mean breakdown rate of 0.0026 K day−1 (CI 0.0022–0.0029). The weight loss in oil palm leaves was higher, 21% (CI 19.9–22.0%) on average, with a rate of 0.009 K day−1 (CI 0.0086–0.0096). Litter, mesh, and land use showed a significant effect on K day−1 (Table 1). Total breakdown rate of oil palms and forests did not differ significantly among land uses (Table 2). Nevertheless, microbial breakdown rate (Kf day−1) of forest litter was higher in rainforests compare to oil palm plantations, and the one for oil palm litter was higher in rainforests compared to the plantations without buffers, but not compared to the buffered ones (Fig. 2). Surprisingly, microbial leaf-litter breakdown surpassed total leaf-litter breakdown for oil palm and forest litter (ANOVA, p = 3.97e−06, p = 0.082, respectively). The breakdown rate estimated for physical abrasion and macroinvertebrate activity (Kc − Kf day−1) for oil palm leaves did not differ among land uses (Table 3); however, the Tukey test showed significant differences for forest litter Kc − Kf day−1 between buffered plantations and all other land uses (Table 4 and Fig. 3).
Leaf breakdown (K day−1) by land use, mesh, and litter type. Boxplots: box (interquartile range), vertical lines (maximum and minimum values), small squares (mean), horizontal lines (median), circles (outliers). Land use: grazing land (G), oil palm without riparian buffer (P), oil palm with riparian buffer (P_RF), and rainforest (F). Significant differences (Tukey test, p ≤ 0.05) between treatments represented by different letters
K coarse mesh − K fine mesh (day−1) and shredder biomass (WWg per 0.05 m2). Boxplots: box (interquartile range), vertical lines (maximum and minimum values), small squares (mean), horizontal black lines (median), gray-circles (outliers). Land use: grazing land (G), oil palm without riparian buffer (P), oil palm with riparian buffer (P_RF), and rainforest (F). Significant differences between treatments (Tukey test: p < 0.05*, p < 0.1•)
Drivers of leaf-litter breakdown
Overall, the leaf-litter breakdown was almost exclusively driven by microbial activity measured in fine-mesh bags (lm, R2 = 0.76, p = 5.96e−13) (Fig. 4). However, shredders and physical breakdown (Kc − Kf day−1) appear to play an important role only in the leaf-litter breakdown of forest leaves in oil palm plantations with riparian buffers. This was the only land use where Kc was significantly higher than Kf (ANOVA, p = 0.05); here, microbes conducted ~ 52% of the total leaf-litter breakdown as opposed to the rest of land uses where the role of shredders and physical breakdown was negligible. Furthermore, shredder biomass was significantly higher in rainforests and oil palm plantations with riparian buffers compared to grazing lands and oil palm plantations without buffers (ANOVA, p = 0.03) (Fig. 3).
Turbidity correlated positively with total and microbial breakdown of forest litter. Physical properties, such as watershed area, depth, water current and discharge, and dissolved oxygen, correlated positively with total breakdown of oil palm litter, while turbidity and scrapper biomass correlated negatively with the oil palm microbial breakdown which correlated positively with silica (Table 5).
Discussion
Our findings suggest potential alterations in the stream ecosystem functioning associated with the oil palm expansion. However, even though we found statistical differences in the leaf-litter breakdown rate among land uses, the actual numerical differences were very small compared to estimates from other studies and systems (Petersen and Cummins 1974; Torres and Ramírez 2014; Rezende et al. 2017; Chellaiah and Yule 2018b). Furthermore, our results differed from the only other study that we know of that compares leaf-litter breakdown in oil palm plantations and forests, as this found higher leaf-litter breakdown rates in the plantations (Chellaiah and Yule 2018b). It is thus not possible to conclude on a general leaf-litter breakdown decline based solely on the existing studies, highlighting the need for more research of leaf-litter breakdown in oil palm plantations.
Leaf-litter breakdown rates
The overall breakdown rates observed for forest species in fine-mesh bags (k day−1 mean = 0.0029), and particularly in coarse-mesh bags (K day−1 mean = 0.0023), were considerably lower than those reported for forest species in tropical lowland streams elsewhere, such as Cecropia schreberiana Miq. (K day−1 = 0.017) in Puerto Rico (Torres and Ramírez 2014); Mauritia flexuosa L. fil. (K day−1 = 0.037) in southeastern Brazil (Rezende et al. 2017); Theobroma cacao L. (K day−1 = 0.008) and Hymenaea courbaril L. (Kc day−1 = 0.005; Kf day−1 = 0.004) in northern Brazil (Firmino et al. 2021); Dryobalanops beccarii Dyer (Kc day−1 = 0.0110, Kf day−1 = 0.0159), Koompassia malaccensis Maingay (Kc day−1 = 0.0112, Kf day−1 = 0.0264), and Shorea sp. (Kc day−1 0.0129, Kf day−1 0.0089) in a dipterocarp forest in Malaysia (Jinggut and Yule 2015); and Macaranga sp. (K day−1 = 0.06–0.08) in oil palm plantations in Borneo, Malaysia (Chellaiah and Yule 2018b).
The studied forest leaf litter consisted of four species, including Pachira aquatica, whose leaf extracts are rich in antimicrobial, antifungal, and nematocidal compounds (Rodrigues and Pastore 2021), potentially explaining the slow breakdown rates of our forest leaf mix. Additionally, leaf extracts from members of the genus Hippocratea have shown antimicrobial (Sojinu et al. 2022) and insecticidal activity (Oboho et al. 2022). Our mix also contained Sloanea leaves, known for their thickness and unpalatability to macroinvertebrates (K day−1 = 0.0094) (Tschelaut et al. 2008). The low breakdown rates observed in our forest mix may be attributed to factors such as high content of cellulose and leaf toughness, low nutritional value (Giweta 2020), and the presence of inhibitors or antimicrobials (Karavin et al. 2016; Luo et al. 2023). However, other studies have reported decomposition rates for species of the genus Pachira to be much higher (Kc day−1 = 0.0205 ± 0.016) than those reported in our study (Blanco and Gutiérrez-Isaza 2014). The same is true for Cecropia sp. (Kc day−1 = 0.0234) (Tschelaut et al. 2008), although Pouroma tends to decompose slower than Cecropia in land (Bakker et al. 2011).
The low breakdown rates observed in the forest mix may be attributed to several factors. Firstly, to the overall low nutritional value of brown leaves. Sampling brown leaves from the forest floor allows for the reabsorption of nutrients by the tree before leaf fall. This process is particularly efficient in nutrient-deficient soils (Xiong et al. 2022), such as those found in Lachuá. Additionally, the process of grouping several species together may impact decomposition rates. In mixed bags, antibiotic or inhibitory compounds released by one species could affect the overall decomposition process (Hättenschwiler et al. 2005). However, leaf mixing can also diversify limiting nutrients, potentially leading to accelerated decomposition (Cassart et al. 2020). But it is worth noting that brown leaves generally have lower concentrations of defensive compounds and nutrients compared to green leaves (Burton et al. 2023), which should theoretically decrease both effects. Another factor to consider is that oil palm leaves were cut from the trees and likely contained higher levels of nutrients compared to the forest mix, which naturally fell from the trees.
Litter breakdown rates can vary considerably among species and regions (Allan and Castillo 2007), and many factors can influence these rates (Giweta 2020), making comparisons between studies problematic. Even when comparing the same species, substantial differences in breakdown rates may also exist. In this study, oil palm leaves lost 21% of their mass after 26 days of immersion (K day−1 = 0.009). Conversely, in Malaysia, these leaves had a mass loss of 41–47% after a month of immersion (K day−1 = 0.01–0.02) (Chellaiah and Yule 2018b), and in northern Brazil, Firmino et al. (2021) reported an average loss of 26% after 60 days (Kc day−1 = 0.006; Kf day−1 = 0.005). Our values are comparable to those in Brazil during the dry season. However, the observed higher rates in Malaysia may be partially ascribed to increased physical abrasion in the streams during sampling, which occurred in the rainy season. In contrast, our study took place during the dry season, resulting in highly stagnant stream conditions (0.019 m s−1; 95% CI = 0.011–0.028). The fact that the breakdown in coarse mesh did not surpass that in fine mesh emphasizes the predominant role of microbes in leaf-litter breakdown. This finding aligns with the widely accepted notion that microbes are the primary drivers of litter breakdown in tropical streams, especially in the neotropics (Dobson et al. 2002; Yule et al. 2009; Boyero et al. 2016), while the role of shredders is often negligible due to their low diversity and low abundance (Boyero et al. 2012; Jinggut and Yule 2015; Chellaiah and Yule 2018b; Firmino et al. 2021).
The relative importance of shredders on litter breakdown varies dramatically among tropical streams, from studies reporting highly diverse and abundant shredder assemblages (e.g., Cheshire et al. 2005; Yule et al. 2009) to studies registering shredder scarcity (Dobson et al. 2002; Jacobsen et al. 2008; Chellaiah and Yule 2018a). Our streams seem to follow an intermediate pattern with highly variable biomass among land uses, from representing 31% and 56% of the total biomass in rainforests and oil palm plantations with riparian buffers, respectively, to 7% and 2% in the plantations lacking buffers and grazing lands (Rojas-Castillo et al. 2023). Shredder richness in our streams was very modest, especially in pools and runs (overall, 0.2–7.7 taxa per stream; 0.5–2.7 taxa per Surber sample); rainforest and plantations with buffer hosted on average 2–3 more taxa than the plantations with no buffers and the grazing lands. Among the most common shredders in our streams were the caddisflies Phylloicus (Mueller, 1880) and Triplectides (Kolenati, 1859), the beetle Anchytarsus (Guérin-Méneville, 1843), and unidentified species of shrimps (Caridae) (Rojas-Castillo et al. 2023).
Still, the overall slightly lower leaf-litter breakdown in coarse-mesh bags compared to the fine-mesh ones was surprising. The reason for this is not clear, but it could be attributed to a higher amount of “anoxic mud” entering the coarse mesh bags, as dissolved oxygen accelerates carbon and nitrogen release and promotes litter breakdown (Liu et al. 2022). This would also explain the overall low breakdown rates. The stream bed was particularly low in oxygen and rich in mud, and several of the bags were completely buried in mud when collected, which could have slowed down the breakdown process, especially in course-mesh bags. It is worth noting that other factors might have contributed to these findings, as previous studies have reported faster breakdown rates in fine-mesh bags compared to coarse-mesh bags, even in systems where mud is less abundant (Jinggut and Yule 2015; Gutiérrez-López et al. 2016).
Effect of land use and riparian buffer on leaf-litter breakdown rates
Microbial leaf-litter breakdown (Kf day−1) of forest and oil palm leaves was significantly lower by 55% (CI 32–84) and by 28% (CI 0.11–0.47), respectively, in oil palm plantations compared to rainforest. This could be due to a different bacterial and fungi community in the plantations. Even though we did not sample the microbial community here, a more recent study in the region performed in the rainy season found differences in the communities across these land uses, reporting lower wood and leaf saprotroph species of fungi in the plantations lacking buffers (Rojas-Castillo 2023). It is possible that the fungi community in the plantations with riparian buffers resembled the community of the plantations lacking buffers in the dry season, as observed in the bacterial communities from intermediate-impacted streams (e.g., streams in silvopasture). These silvopasture communities resembled forest communities during the rainy season but grazing land communities in the dry season (Chavarria et al. 2021). This could explain why in our study, the riparian buffers appeared not to have an effect on the stream microbial leaf-litter breakdown of forest leaves. The lower breakdown by microbes in the plantations may also be attributed to the use of herbicides and pesticides in this agriculture, as this restricts aquatic hyphomycetes colonization (Sridhar et al. 1992). This would, however, also have affected litter breakdown in grazing lands, which was not the case.
The absence of significant differences between grazing lands and forests was highly surprising, as grazing lands typically exhibit slower breakdown rates (Zúñiga-Sarango et al. 2020; de Mello Cionek et al. 2021). This slower decomposition in grazing lands often results from riparian vegetation removal, frequent siltation (increasing leaf burial), and grass proliferation along the margins, which homogenize habitats, intensify solar exposure, and causes flow disruption. Consequently, these conditions lead to reduced richness and abundance of shredders (de Mello Cionek et al. 2021), findings consistent with observations in our streams within grazing lands (Rojas-Castillo et al. 2023). However, several studies have reported similar breakdown rates in grazing lands compared to forests (McTammany et al. 2008; Hladyz et al. 2010). These studies attribute the lack of differences to shifts in biological drivers, such as an increase in microbial activity alongside a decrease in invertebrate abundance (Hladyz et al. 2010). Microbes respond positively to environmental factors like temperature and nutrients, which are often higher in grazing lands. However, they respond negatively to high sediment load (McTammany et al. 2008), conditions present in our grazing lands, potentially explaining the lack of difference in microbial breakdown in these land uses. Another factor potentially minimizing differences in shredder and total leaf-litter breakdown could be the habitats sampled. To avoid leaf-litter desiccation, we did not sample riffles, which typically harbor the richest and most abundant shredder communities (Wang et al. 2023). The complex dynamics and high variability of litter breakdown in grazing lands underscore the importance of local studies for tailored livestock production farm management. Furthermore, it emphasizes the need for studies that compile and organize global data regionally to establish standardized management practices that consider location-specific variables.
In our study, the total leaf-litter breakdown rate (Kc day−1) of oil palm leaves was similar among land uses, and the breakdown rate of forest leaves was lower (not significantly) only in the plantations with no riparian buffers. These results differed from a Malaysian study, where the decay rates of oil palm and Macaranga leaves were higher in the plantations than in forests (Chellaiah and Yule 2018b). This variability may be associated with regional or local factors, highlighting the need for additional research both locally and globally. The higher forest-leaf breakdown rates in buffered plantations (as opposed to the unbuffered ones) may be associated with shredder activity, as the stream current and water flow (and thus physical abrasion) did not differ among land uses, while shredder biomass did. Shredders may play a role in the forest-leaf breakdown in the plantations conserving riparian buffers, as these streams hosted a higher biomass and diversity of shredders compared to the ones lacking these buffers (Rojas-Castillo et al. 2023). The low Kc − Kf (day−1) in rainforests could be attributed to the presence of preferable food sources in these streams, considering that the biomass of macroinvertebrate shredders was as high as in the plantations with riparian buffers.
Drivers of leaf-litter breakdown
Leaf-litter breakdown appeared to be driven primarily by microbial activity, which is known to be affected by factors such as temperature and water-quality parameters (nutrient concentrations, dissolved oxygen, and pH) (Suberkropp and Chauvet 1995; Rezende et al. 2017; Trevathan-Tackett et al. 2020; Liu et al. 2022). In our study, breakdown rates were not correlated with water temperature, pH, or any nutrient other than silica, which correlated positively. Litter breakdown showed a negative relation with turbidity and a positive relation with dissolved oxygen. Liu et al., (2022) found that litter breakdown levels in anaerobic conditions tend to be half of those at low oxygen levels (4 mg L−1) and one-third of those at higher levels (7 mg L−1). Thus, in our low-oxygenated streams, a difference of 2 mg L−1 in the stream bed could have had an effect. In addition, litter breakdown showed a positive relation with many physical factors including catchment area, stream depth, and water current and discharge. These parameters are associated with physical abrasion, but the gradients between the streams are very small (CI 95%: area = 22–70 ha, depth = 15–19 cm, current = 0.7–14 L s−1, current = 1.1–2.8 cm s−1).
The effect of shredder macroinvertebrates in rainforest streams appeared to be negligible, even if shredder biomass was significantly higher than in grazing lands and oil palm plantations with no buffers. This may be due to the abundance of leaf litter in the buffered streams in the plantations, especially during the dry season, when the lack of flow accumulates the resource. It may be that the shredders were not interested in the leaf litter from the bags since the streams were already rich in diverse leaf litter with already conditioned material (algae, fungi, and bacteria in the leaf litter), which is preferred by the shredders (Trochine et al. 2021). Streams surrounded by riparian buffers in the plantations were the only ones where Kf day−1 was significantly lower than Kc day−1. This difference of 48% could be attributed to the effect of shredders in these streams, as the biomass was as high as in rainforests (6.3 × 10–3 g/m2 in pools and runs). These riparian buffers provide abundant leaf material to the streams, but in contrast to what happens in rainforests, it is possible that there is less conditioned material (less fungi) in these plantations, and therefore the macroinvertebrate did not prefer the stream leaf litter over the material in the bags.
This study has several limitations, including the absence of a microbial community analysis, which restricts the detection of biological drivers. Additionally, the sampling was restricted to a limited number of streams and habitats, and breakdown rates for individual forest species were not measured. These limitations constrain, to some extent, the statistical power and the ability to compare results with previous studies. Despite these constraints, this research offers valuable insights into the potential impacts of land-use change on freshwater ecosystems. It underscores the urgent need to expand research on how activities such as oil palm plantations and other agricultural practices affect leaf-litter breakdown and their underlying biological processes.
Data availability
Data and R-code are available upon reasonable request.
References
Abelho M, Descals E (2019) Litter movement pathways across terrestrial–aquatic ecosystem boundaries affect litter colonization and decomposition in streams. Funct Ecol 33:1785–1797. https://doi.org/10.1111/1365-2435.13356
Allan JD (2004) Landscapes and riverscapes: the influence of land use on stream ecosystems. Annu Rev Ecol Evol Syst 35:257–284. https://doi.org/10.1146/annurev.ecolsys.35.120202.110122
Allan JD, Castillo MM (2007) Stream ecology: structure and function of running waters, 2nd edn. Chapman and Hall, New York
Bakker MA, Carreño-Rocabado G, Poorter L (2011) Leaf economics traits predict litter decomposition of tropical plants and differ among land use types. Funct Ecol 25:473–483. https://doi.org/10.1111/j.1365-2435.2010.01802.x
Battin TJ, Kaplan LA, Findlay S et al (2008) Biophysical controls on organic carbon fluxes in fluvial networks. Nat Geosci 1:95–100. https://doi.org/10.1038/ngeo101
Behera SK, Shukla AK, Suresh K et al (2022) Yield variability in oil palm plantations in Tropical India is influenced by surface and sub-surface soil fertility and leaf mineral nutrient contents. Sustainability 14:2672. https://doi.org/10.3390/su14052672
Benfield EF, Fritz KM, Tiegs SD (2017) Leaf-litter breakdown. In: Lamberti GA, Hauer FR (eds) Methods in stream ecology, 3rd edn. Academic Press, London, pp 71–82
Berg CC, Akkermans RWAP, van Heusden ECH (1990) Cecropiaceae: Coussapoa and Pourouma, with an Introduction to the Family. Flora Neotrop 51:1–208
Blanco JF, Gutiérrez-Isaza N (2014) Leaf litter mass loss rates and associated fauna of tree species commonly used in neotropical riparian reforestation. Acta Biol Colomb 19:91–100. https://doi.org/10.15446/abc.v19n1.38155
Boulton AJ, Boon PI (1991) A review of methodology used to measure leaf litter decomposition in lotie environments: Time to turn over an old leaf? Mar Freshw Res 42:1–43. https://doi.org/10.1071/MF9910001
Boyero L, Pearson RG, Dudgeon D et al (2012) Global patterns of stream detritivore distribution: implications for biodiversity loss in changing climates. Glob Ecol Biogeogr 21:134–141. https://doi.org/10.1111/j.1466-8238.2011.00673.x
Boyero L, Pearson RG, Hui C et al (2016) Biotic and abiotic variables influencing plant litter breakdown in streams: a global study. Proc R Soc B Biol Sci 283:20152664. https://doi.org/10.1098/rspb.2015.2664
Burlakova LE, Karatayev AY, Karatayev VA et al (2011) Endemic species: Contribution to community uniqueness, effect of habitat alteration, and conservation priorities. Biol Conserv 144:155–165. https://doi.org/10.1016/j.biocon.2010.08.010
Burton GP, Prescott TAK, Fang R, Lee MA (2023) Regional variation in the antibacterial activity of a wild plant, wild garlic (Allium ursinum L.). Plant Physiol Biochem 202:107959. https://doi.org/10.1016/j.plaphy.2023.107959
Cassart B, Angbonga Basia A, Jonard M, Ponette Q (2020) Average leaf litter quality drives the decomposition of single-species, mixed-species and transplanted leaf litters for two contrasting tropical forest types in the Congo Basin (DRC). Ann for Sci 77:33. https://doi.org/10.1007/s13595-020-00942-4
Chavarria KA, Saltonstall K, Vinda J et al (2021) Land use influences stream bacterial communities in lowland tropical watersheds. Sci Rep 11:21752. https://doi.org/10.1038/s41598-021-01193-7
Chellaiah D, Yule CM (2018a) Riparian buffers mitigate impacts of oil palm plantations on aquatic macroinvertebrate community structure in tropical streams of Borneo. Ecol Indic 95:53–62. https://doi.org/10.1016/j.ecolind.2018.07.025
Chellaiah D, Yule CM (2018b) Litter decomposition is driven by microbes and is more influenced by litter quality than environmental conditions in oil palm streams with different riparian types. Aquat Sci 80:43. https://doi.org/10.1007/s00027-018-0595-y
Cheshire K, Boyero L, Pearson RG (2005) Food webs in tropical Australian streams: shredders are not scarce. Freshw Biol 50:748–769. https://doi.org/10.1111/J.1365-2427.2005.01355.X
Choudhary M, Grover K (2019) Palm (Elaeis guineensis Jacq.) Oil. In: Ramadan MF (ed) Fruit oils: chemistry and functionality. Springer International Publishing, Cham, pp 789–802
Cole LJ, Stockan J, Helliwell R (2020) Managing riparian buffer strips to optimise ecosystem services: a review. Agric Ecosyst Environ 296:106891. https://doi.org/10.1016/J.AGEE.2020.106891
CONAP (2003) Plan Maestro del Parque Nacional Laguna Lachuá 2004–2009. Guatemala
Darmawan AA, Ariyanto DP, Basuki TM, Syamsiyah J (2021) Measuring of leaf litter decomposition rate and flux of carbon dioxide in various land cover in Gunung Bromo Education Forest, Karanganyar. IOP Conf Ser Earth Environ Sci 824:12055. https://doi.org/10.1088/1755-1315/824/1/012055
de Mello Cionek V, Fogaça FNO, Moulton TP et al (2021) Influence of leaf miners and environmental quality on litter breakdown in tropical headwater streams. Hydrobiologia 848:1311–1331. https://doi.org/10.1007/s10750-021-04529-6
Deere NJ, Bicknell JE, Mitchell SL et al (2022) Riparian buffers can help mitigate biodiversity declines in oil palm agriculture. Front Ecol Environ. https://doi.org/10.1002/FEE.2473
Dobson M, Magana A, Mathooko JM, Ndegwa FK (2002) Detritivores in Kenyan highland streams: more evidence for the paucity of shredders in the tropics? Freshw Biol 47:909–919. https://doi.org/10.1046/j.1365-2427.2002.00818.x
Domínguez E (2009) Macroinvertebrados bentónicos sudamericanos, 1st edn. Fundación Miguel Lillo, Tucumán
dos Santos UM, de Carvalho Gonçalves JF, Feldpausch TR (2006) Growth, leaf nutrient concentration and photosynthetic nutrient use efficiency in tropical tree species planted in degraded areas in central Amazonia. For Ecol Manag 226:299–309. https://doi.org/10.1016/j.foreco.2006.01.042
Dudgeon D, Arthington AH, Gessner MO et al (2006) Freshwater biodiversity: importance, threats, status and conservation challenges. Biol Rev 81:163–182. https://doi.org/10.1017/S1464793105006950
Elias DMO, Robinson S, Both S et al (2020) Soil microbial community and litter quality controls on decomposition across a tropical forest disturbance gradient. Front for Glob Change. https://doi.org/10.3389/ffgc.2020.00081
Escuela de Biología (2004) Ficha Informativa de los Humedales de Ramsar (FIR). Guatemala
FAO (2022a) FRA 2020 remote sensing survey. FAO Forest, Rome
FAO (2022b) FAOSTAT. https://www.fao.org/faostat/en/#data/. Accessed 19 Jun 2023
Firmino VC, Brasil LS, Martins RT et al (2021) Litter decomposition of exotic and native plant species of agricultural importance in Amazonian streams. Limnology 1:3. https://doi.org/10.1007/s10201-021-00655-1
Forman J (2019) Introduction to linear mixed models for repeated measurements data: analysis of single group studies with the nlme-package in R.
Foster WA, Snaddon JL, Turner EC et al (2011) Establishing the evidence base for maintaining biodiversity and ecosystem function in the oil palm landscapes of South East Asia. Philos Trans R Soc B Biol Sci 366:3277–3291. https://doi.org/10.1098/rstb.2011.0041
Gessner MO, Chauvet E, Dobson M (1999) A perspective on leaf litter breakdown in streams. Oikos 85:377–384. https://doi.org/10.2307/3546505
Giweta M (2020) Role of litter production and its decomposition, and factors affecting the processes in a tropical forest ecosystem: a review. J Ecol Environ 44:11. https://doi.org/10.1186/s41610-020-0151-2
Goldman ED, Weisse M, Harris N, Schneider M (2020) Estimating the role of seven commodities in agriculture-linked deforestation: oil palm, soy, cattle, wood fiber, cocoa, coffee, and rubber. Tech Note, World Resour Inst 22. https://doi.org/10.46630/writn.na.00001
Gomes SMA, da Silva EAM, Lombardi JA et al (2005) Anatomia foliar como subsídio à taxonomia de Hippocrateoideae (Celastraceae) no Sudeste do Brasil. Acta Bot Brasilica 19:945–961. https://doi.org/10.1590/S0102-33062005000400029
Gonçalves AL, Graça MAS, Canhoto C (2013) The effect of temperature on leaf decomposition and diversity of associated aquatic hyphomycetes depends on the substrate. Fungal Ecol 6:546–553. https://doi.org/10.1016/j.funeco.2013.07.002
Google Earth (2021) Lachuá Ecoregion, Alta Verapaz.15°56´07.40´´N, 90°39´50.00´´O. Eye alt 5.75km. 2022 CNES/Airbus
Granados P (2001) Ictiofauna de la Laguna Lachuá, Parque Nacional Laguna Lachuá, Cobán. Universidad de San Carlos de Guatemala, Alta Verapaz
Grobben YJH (2022) Forest Regeneration on previous pastureland in the Talamanca Mountain Range, Costa Rica. Costa Rica
Gutiérrez-López A, Meza-Salazar AM, Guevara G (2016) Leaf decomposition and colonization by aquatic macroinvertebrates in two tropical microcatchments (Manizales, Colombia). Hidrobiológica 26:347–357. https://doi.org/10.24275/uam/izt/dcbs/hidro/2016v26n3/Guevara
Hamada N, Thorp JH, Rogers DC (2018) Thorp and covich’s freshwater invertebrates, vol 3. Keys to neotropical Hexapoda. Academic Press, London
Hättenschwiler S, Tiunov AV, Scheu S (2005) Biodiversity and litter decomposition in terrestrial ecosystems. Annu Rev Ecol Evol Syst 36:191–218. https://doi.org/10.1146/annurev.ecolsys.36.112904.151932
Hickey MBC, Doran B (2004) A review of the efficiency of buffer strips for the maintenance and enhancement of riparian ecosystems. Water Qual Res J 39:311–317. https://doi.org/10.2166/WQRJ.2004.042
Hladyz S, Tiegs SD, Gessner MO et al (2010) Leaf-litter breakdown in pasture and deciduous woodland streams: a comparison among three European regions. Freshw Biol 55:1916–1929. https://doi.org/10.1111/j.1365-2427.2010.02426.x
Hothorn T, Bretz F, Westfall P (2008) Simultaneous inference in general parametric models. Biometrical J 50:346–363
Hughes AC, Orr MC, Ma K et al (2021) Sampling biases shape our view of the natural world. Ecography (cop) 44:1259–1269. https://doi.org/10.1111/ecog.05926
Infante Mata D, Moreno-Casasola P, Madero-Vega C (2012) Litterfall of tropical forested wetlands of Veracruz in the coastal floodplains of the Gulf of Mexico. Aquat Bot 98:1–11. https://doi.org/10.1016/j.aquabot.2011.11.006
Iñiguez-Armijos C, Rausche S, Cueva A et al (2016) Shifts in leaf litter breakdown along a forest–pasture–urban gradient in Andean streams. Ecol Evol 6:4849–4865. https://doi.org/10.1002/ece3.2257
Jacobsen D, Cressa C, Mathooko JM, Dudgeon D (2008) Macroinvertebrates: composition, life histories and production. In: Dudgeon D (ed) Tropical Stream ecology. Academic Press, London, pp 65–105
Jinggut T, Yule CM (2015) Leaf-litter breakdown in streams of East Malaysia (Borneo) along an altitudinal gradient: initial nitrogen content of litter limits shredder feeding. Freshw Sci 34:691–701. https://doi.org/10.1086/681256
Karavin N, Cımık A, Ural Z, Erdem E (2016) Dynamics of litter decomposition: effect of antimicrobial features on leaf decomposition. Int J Pure App Biosci 4:24–27. https://doi.org/10.18782/2320-7051.2213
Klein Goldewijk K, Beusen A, Doelman J, Stehfest E (2017) Anthropogenic land use estimates for the Holocene–HYDE 3.2. Earth Syst Sci Data 9:927–953. https://doi.org/10.5194/essd-9-927-2017
Laurance WF, Sayer J, Cassman KG (2014) Agricultural expansion and its impacts on tropical nature. Trends Ecol Evol 29:107–116. https://doi.org/10.1016/j.tree.2013.12.001
Lemes da Silva AL, Lemes WP, Andriotti J et al (2020) Recent land-use changes affect stream ecosystem processes in a subtropical island in Brazil. Austral Ecol 45:644–658. https://doi.org/10.1111/aec.12879
Liu S, He G, Fang H et al (2022) Effects of dissolved oxygen on the decomposers and decomposition of plant litter in lake ecosystem. J Clean Prod 372:133837. https://doi.org/10.1016/j.jclepro.2022.133837
Luke SH, Slade EM, Gray CL et al (2019) Riparian buffers in tropical agriculture: scientific support, effectiveness and directions for policy. J Appl Ecol 56:85–92. https://doi.org/10.1111/1365-2664.13280
Luo Y, Wang L, Cao T et al (2023) Legacy effect of plant chemical defence substances on litter decomposition. Plant Soil 487:93–108. https://doi.org/10.1007/s11104-023-05954-3
Manzoni S, Trofymow JA, Jackson RB, Porporato A (2010) Stoichiometric controls on carbon, nitrogen, and phosphorus dynamics in decomposing litter. Ecol Monogr 80:89–106. https://doi.org/10.1890/09-0179.1
Marín-Muñiz JL, Hernández ME, Moreno-Casasola P (2014) Comparing soil carbon sequestration in coastal freshwater wetlands with various geomorphic features and plant communities in Veracruz, Mexico. Plant Soil 378:189–203. https://doi.org/10.1007/s11104-013-2011-7
McTammany ME, Benfield EF, Webster JR (2008) Effects of agriculture on wood breakdown and microbial biofilm respiration in southern Appalachian streams. Freshw Biol 53:842–854. https://doi.org/10.1111/j.1365-2427.2007.01936.x
Meijaard E, Brooks TM, Carlson KM et al (2020) The environmental impacts of palm oil in context. Nat Plants 6:1418–1426. https://doi.org/10.1038/s41477-020-00813-w
Mendoza E, Dirzo R (1999) Deforestation in Lacandonia (southeast Mexico): evidence for the declaration of the northernmost tropical hot-spot. Biodivers Conserv 8:1621–1641. https://doi.org/10.1023/A:1008916304504
Merritt RW, Cummins KW, Berg MB (2017) Trophic relationships of macroinvertebrates. Methods Stream Ecol Third Ed 1:413–433. https://doi.org/10.1016/B978-0-12-416558-8.00020-2
Mwaijengo GN, Vanschoenwinkel B, Dube T et al (2020) Seasonal variation in benthic macroinvertebrate assemblages and water quality in an Afrotropical river catchment, northeastern Tanzania. Limnologica 82:125780. https://doi.org/10.1016/j.limno.2020.125780
Oboho DE, Akwaowo UN, Edeke A et al (2022) Phytochemical screening, GC-MS and histological effects of methanolic leaf extract of Hippocratea africana (Willd) on the midgut of Sitophilus zeamais (Motsch.). Lond J Sci Res 24:11–21
Oliveira ALHD, Nessimian JL (2010) Distribuição espacial e categorização funcional trófica de comunidades de insetos aquáticos em rios na Serra da Bocaina, Sudeste Brasil. Acta Limnol Bras 22:424–441. https://doi.org/10.4322/actalb.2011.007
Olson JS (1963) Energy storage and the balance of producers and decomposers in ecological systems. Ecology 44:322–331. https://doi.org/10.2307/1932179
Pearson RG (2014) Dynamics of invertebrate diversity in a tropical stream. Diversity 6:771–791. https://doi.org/10.3390/d6040771
Peralta-Maraver I, Perkins DM, Thompson MSA et al (2019) Comparing biotic drivers of litter breakdown across stream compartments. J Anim Ecol 88:1146–1157. https://doi.org/10.1111/1365-2656.13000
Pereira PDS, de Souza NF, Baptista DF et al (2021) Functional Feeding Group composition and attributes: evaluation of freshwater ecosystems in Atlantic Forest, Brazil. Biota Neotrop. https://doi.org/10.1590/1676-0611-BN-2020-1016
Pérez J, Boyero L, Raquel Tuñón A et al (2023) Agricultural impacts on lowland tropical streams detected through leaf litter decomposition. Ecol Indic 154:110819. https://doi.org/10.1016/j.ecolind.2023.110819
Petersen RC, Cummins KW (1974) Leaf processing in a woodland stream. Freshw Biol 4:343–368. https://doi.org/10.1111/j.1365-2427.1974.tb00103.x
Pinheiro J, Bates D, R Core Team (2022) nlme: linear and nonlinear mixed effects models
R Core Team (2022) R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna
Rezende RS, Santos AM, Medeiros AO, Gonçalves-Júnior JF (2017) Temporal leaf litter breakdown in a tropical riparian forest with an open canopy. Limnetica 36:445–459. https://doi.org/10.23818/limn.36.14
Ritchie H, Roser M (2021) Forests and deforestation. Our World Data
Rodrigues AP, Pastore GM (2021) A review of the nutritional composition and current applications of monguba (Pachira aquatica Aubl.) plant. J Food Compos Anal 99:103878. https://doi.org/10.1016/j.jfca.2021.103878
Rojas-Castillo OA (2023) The impacts of the oil palm expansion on freshwater biodiversity and ecosystem functioning: The role of riparian buffers in mitigating stream biodiversity loss. University of Copenhagen
Rojas-Castillo OA, Kepfer-Rojas S, Vargas N, Jacobsen D (2023) Forest buffer-strips mitigate the negative impact of oil palm plantations on stream communities. Sci Total Environ 873:162259. https://doi.org/10.1016/J.SCITOTENV.2023.162259
Rojas-Castillo OA, Rojas SK, Juen L et al (2024) Meta-analysis contrasting freshwater biodiversity in forests and oil palm plantations with and without riparian buffers. Conserv Biol 38:e14172. https://doi.org/10.1111/COBI.14172
RStudio Team (2022) RStudio: integrated development environment for R
Sala OE, Stuart Chapin F, Armesto JJ et al (2000) Global biodiversity scenarios for the year 2100. Science (80-) 287:1770–1774. https://doi.org/10.1126/science.287.5459.1770
Sanaullah M, Usman M, Wakeel A et al (2020) Terrestrial ecosystem functioning affected by agricultural management systems: a review. Soil till Res 196:104464. https://doi.org/10.1016/j.still.2019.104464
Silva-Junior EF, Moulton TP, Boëchat IG, Gücker B (2014) Leaf decomposition and ecosystem metabolism as functional indicators of land use impacts on tropical streams. Ecol Indic 36:195–204. https://doi.org/10.1016/j.ecolind.2013.07.027
Smith AC (1940) Hippocratea. Brittonia 3:356–367. https://doi.org/10.2307/2804625
Sojinu OS, Akinloye DI, Mosaku AM et al (2022) Chemical constituents and an antimicrobial assay of methanol root and leaves extracts of Hippocratea Indica Willd. J Chem Soc Niger. https://doi.org/10.46602/jcsn.v67i3.753
Springer M, Ramírez A, Hanson P (2010) Macroinvertebrados de agua dulce de Costa Rica I. Rev Biol Trop 58:151–198. https://doi.org/10.15517/rbt.v58i4
Sridhar KR, Chandrashekar KR, Kaveriappa KM (1992) Research on the Indian subcontinent. The ecology of aquatic hyphomycetes. Springer, Berlin, pp 182–211
Suberkropp K, Chauvet E (1995) Regulation of leaf breakdown by fungi in streams: influences of water chemistry. Ecology 76:1433–1445. https://doi.org/10.2307/1938146
Team QD (2015) QGIS geographic information system. Open source geospatial Found. Proj
Tiegs SD, Akinwole PO, Gessner MO (2009) Litter decomposition across multiple spatial scales in stream networks. Oecologia 161:343–351. https://doi.org/10.1007/s00442-009-1386-x
Torres PJ, Ramírez A (2014) Land-use effects on leaf litter breakdown in low-order streams draining a rapidly developing tropical watershed in Puerto Rico. Rev Biol Trop 62:129–142. https://doi.org/10.15517/rbt.v62i0.15783
Trevathan-Tackett SM, Brodersen KE, Macreadie PI (2020) Effects of elevated temperature on microbial breakdown of seagrass leaf and tea litter biomass. Biogeochemistry 151:171–185. https://doi.org/10.1007/s10533-020-00715-1
Trochine C, Diaz Villanueva V, Brett MT (2021) The ultimate peanut butter on crackers for Hyalella: diatoms on macrophytes rather than bacteria and fungi on conditioned terrestrial leaf litter. Freshw Biol 66:599–614. https://doi.org/10.1111/fwb.13664
Tschelaut J, Weissenhofer A, Schiemer F (2008) Macroinvertebrates and leaf litter decomposition in a neotropical lowland stream, Quebrada Negra, Costa Rica. Stapfia 88:457–466
Umaña MN, Swenson NG (2019) Does trait variation within broadly distributed species mirror patterns across species? A case study in Puerto Rico. Ecology 100:e02745. https://doi.org/10.5061/dryad.6505nf4
Wang L, Xia L, Li J et al (2023) Winter dynamics of functional diversity and redundancy of riffle and pool macroinvertebrates after defoliation in a temperate forest stream. Front Microbiol 14:1105323. https://doi.org/10.3389/fmicb.2023.1105323
Webster JR, Benfield EF (1986) Vascular plant breakdown in freshwater ecosystems. Annu Rev Ecol Syst 17:567–594. https://doi.org/10.1146/annurev.es.17.110186.003031
Werden LK, Alvarado JP, Zarges S et al (2018) Using soil amendments and plant functional traits to select native tropical dry forest species for the restoration of degraded Vertisols. J Appl Ecol 55:1019–1028. https://doi.org/10.5061/dryad.fd57r
White KE (1978) Dilution methods. Wiley, Chichester, pp 47–55
Xiong H, Ma H, Zhao H et al (2022) Integrated physiological, proteome and gene expression analyses provide new insights into nitrogen remobilization in citrus trees. Tree Physiol 42:1628–1645. https://doi.org/10.1093/treephys/tpac024
Yoshikawa VN, Duarte MC, Ferreira CDM (2022) Typification of names in Pachira aquatica Aubl. (Malvaceae, Bombacoideae) with a new combination and new status from the Brazilian Amazon forest. Acta Bot Bras 36:e2021abb0117. https://doi.org/10.1590/0102-33062021abb0117
Young RG, Matthaei CD, Townsend CR (2008) Organic matter breakdown and ecosystem metabolism: functional indicators for assessing river ecosystem health. J N Am Benthol Soc 27:605–625. https://doi.org/10.1899/07-121.1
Yule CM, Leong MY, Liew KC et al (2009) Shredders in Malaysia: abundance and richness are higher in cool upland tropical streams. J N Am Benthol Soc 28:404–415. https://doi.org/10.1899/07-161.1/ASSET/IMAGES/LARGE/I0887-3593-028-02-0404-F04.JPEG
Zúñiga-Sarango W, Gaona FP, Reyes-Castillo V, Iñiguez-Armijos C (2020) Disrupting the biodiversity–ecosystem function relationship: response of shredders and leaf breakdown to urbanization in Andean streams. Front Ecol Evol 8:592404. https://doi.org/10.3389/fevo.2020.592404
Acknowledgements
We want to thank the personnel of the Lachuá National Park, Ricardo Cac, the Rojas and Vargas family, Hans Graff, and Natalia Vargas for their support in the field.
Funding
Open access funding provided by Copenhagen University. This project received funding from the European Union’s Horizon 2020 research and innovation programme under the Marie Skłodowska-Curie agreement no. 801199.
Author information
Authors and Affiliations
Contributions
Conceptualization: OARC, DJ, and SKR. Developing methods: OARC, DJ, and SKR. Conducting the research: OARC. Data analysis: OARC. Data interpretation: OARC. Preparation of figures and tables: OARC. Writing: OARC, DJ, and SKR.
Corresponding author
Ethics declarations
Conflict of interest
The authors have no competing interests to declare that are relevant to the content of this article.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Rojas-Castillo, O.A., Kepfer-Rojas, S. & Jacobsen, D. Land-use effects on leaf-litter breakdown in streams in a tropical lowland catchment. Aquat Sci 86, 67 (2024). https://doi.org/10.1007/s00027-024-01079-6
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00027-024-01079-6