Abstract
Transactive response DNA-binding protein 43 kDa (TDP-43) encoded by the TARDBP gene is an evolutionarily conserved heterogeneous nuclear ribonucleoprotein (hnRNP) that regulates multiple steps of RNA metabolism, and its cytoplasmic aggregation characterizes degenerating motor neurons in amyotrophic lateral sclerosis (ALS). In most ALS cases, cytoplasmic TDP-43 aggregation occurs in the absence of mutations in the coding sequence of TARDBP. Thus, a major challenge in ALS research is to understand the nature of pathological changes occurring in wild-type TDP-43 and to explore upstream events in intracellular and extracellular milieu that promote the pathological transition of TDP-43. Despite the inherent obstacles to analyzing TDP-43 dynamics in in vivo motor neurons due to their anatomical complexity and inaccessibility, recent studies using cellular and animal models have provided important mechanistic insights into potential links between TDP-43 and motor neuron vulnerability in ALS. This review is intended to provide an overview of the current literature on the function and regulation of TDP-43-containing RNP granules or membraneless organelles, as revealed by various models, and to discuss the potential mechanisms by which TDP-43 can cause selective vulnerability of motor neurons in ALS.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Abnormal aggregation of proteins that normally function as components of ribonucleoprotein (RNP) granules is a hallmark of neurodevelopmental and neurodegenerative diseases [1,2,3]. Under normal physiological conditions, RNP granules form membraneless partitions in the nucleus and cytoplasm to control the flow of genetic information. Assembly of RNP granules may also be driven by external stimuli, such as during stress granule (SG) formation in response to cellular stresses. A remarkable feature of RNP granules is their compositional heterogeneity and structural flexibility. A single RNP, depending on the interacting partner (proteins or RNAs), can form either liquid-like physiological assemblies or solid-like fibers [4, 5]. Such solid-like RNPs are a candidate source for pathological aggregates that accumulate over time during the progression of diseases, although some evidence suggests that protein aggregates without RNA are associated with cellular toxicity [6, 7].
Transactive response DNA-binding protein 43 kDa (TDP-43) is an evolutionarily conserved RNA/DNA-binding protein encoded by the TARDBP gene and regulating transcription [8,9,10], RNA metabolism [11,12,13,14], anti-viral response [15], DNA damage response [16], and chromatin structure [17]. In 97% of amyotrophic lateral sclerosis (ALS) cases, the most common motor neuron disease, and in 45% of frontotemporal dementia (FTD) cases [18], aggregation of TDP-43 is detectable in degenerating neurons. TDP-43 protein has a homo-oligomerization domain, RNA-binding domains, and an intrinsically disordered region (IDR) in tandem, each containing amino acid sequence motifs for the various regulation, such as post-translational modification, nucleocytoplasmic transport, and proteolysis. In sporadic ALS, which accounts for approximately 90% of ALS cases, TDP-43 aggregation occurs without mutation in the coding sequence of the TARDBP gene, rendering the mechanism underlying aggregation of wild-type TDP-43 largely unknown. On the other hand, in familial ALS cases associated with the TARDBP locus, mutations have mostly, but not exclusively, been found in the IDR [19, 20]. Given that by mediating a multitude of intermolecular interactions, IDRs typically drive the transition from soluble protein to liquid droplets of protein [21], dysregulation of IDR-dependent homomeric and heteromeric TDP-43 assembly likely underlies the pathogenesis of ALS. Under physiological conditions, TDP-43-containing RNP granules exist in various subcellular compartments, each granule differing in its protein: RNA composition according to its function. Currently, an understanding of how a cell monitors the global and local levels of intracellular TDP-43 and specifies division of labor for granular and non-granular TDP-43 is far from complete.
At the systems level, TDP-43 is a ubiquitously expressed protein, like the misfolded proteins in other neurodegenerative diseases, such as amyloid β in Alzheimer’s disease, α-Synuclein in Parkinson’s disease, and huntingtin in Huntington’s disease [22]. However, as observed in these neurodegenerative diseases, selective subpopulations of neurons are affected in ALS: upper and lower motor neurons. Despite being a pathological hallmark of ALS, the extent to which TDP-43 aggregation accounts for the selective vulnerability of motor neurons is largely unknown. This is primarily due to the anatomical complexity and inaccessibility of motor neurons, hampering in vivo investigation of TDP-43 dynamics in live motor neurons [23, 24]. Thus, for an understanding of the pathogenesis of ALS associated with TDP-43 aggregation, it is imperative to fully figure out the functions and regulation of TDP-43-containing RNP complexes that assemble in the normal physiological conditions, investigate the nature of pathological changes occurring in wild-type TDP-43, and explore upstream intracellular and extracellular factors that promote the pathological transition of TDP-43 in motor neurons in vivo (Fig. 1). With these challenges in mind, in the present review, we present an overview of the domain structure of TDP-43 and its regulation. We subsequently discuss the known functions and properties of TDP-43 granules that have been elucidated via numerous cell culture and animal models. Finally, we review neuron-specific TDP-43 properties to explore the potential link between TDP-43 pathology and selective neuronal vulnerability in ALS.
Diagrams depicting TDP-43 in motor neurons under physiological (a) and pathological (b) conditions. a Under physiological conditions, TDP-43 (magenta) is primarily nuclear and regulates transcription and pre-mRNA processing. TDP-43 also forms mRNA transport granules and supports translation at neuromuscular synapses. Intracellular Ca2+ homeostasis is maintained by mitochondria. b In sporadic ALS or familial ALS associated with TARDBP mutations, the cytoplasmic pool of TDP-43 increases and forms aggregates (purple). The nuclear pool of TDP-43 is instead depleted. Transport granules containing pathological TDP-43 frequently display retrograde movement, which may result in diminished translation in the synaptic terminal and denervation. Dysregulated Ca2+ homeostasis and/or excessive Ca2+ influx due to neuronal hyperactivation may promote calpain-dependent TDP-43 cleavage, promoting TDP-43 aggregation. Neuronal hyperactivation produces the aggregation-prone splice isoforms of TDP-43
DNA and RNA-binding functions of TDP-43
TDP-43, a ubiquitous DNA/RNA binding protein, plays multiple roles in both the nucleus and cytoplasm. First identified as a cellular factor that bound to a regulatory element of the human immunodeficiency virus type 1 (HIV-1) long terminal repeat (LTR) and repressed its transcription [8], TDP-43 was later characterized as a binding protein for the spermatid-specific promoter of the SP-10 gene [9]. Beyond transcription, TDP-43 is also implicated in the maintenance of chromatin structure around long interspersed nuclear elements [17] and DNA damage response [16], both of which are presumably mediated by its DNA-binding capacity. TDP-43, as an RNA-binding protein, associates with more than 6000 target RNAs, including those encoding proteins for neuronal development and function [10, 25,26,27,28,29,30,31,32,33]. Encompassing a wide range of RNA metabolisms, the RNA-regulatory roles of TDP-43 include RNA splicing, RNA transport, translation [11, 12, 14], and biogenesis of non-coding RNAs [13, 34]. In the following sections, we present an overview of evidence that the diverse functions of TDP-43 are underpinned both by its modular molecular architecture and by its capacity for assembling RNP granules or other protein complexes.
TDP-43 structure and post-translational modification
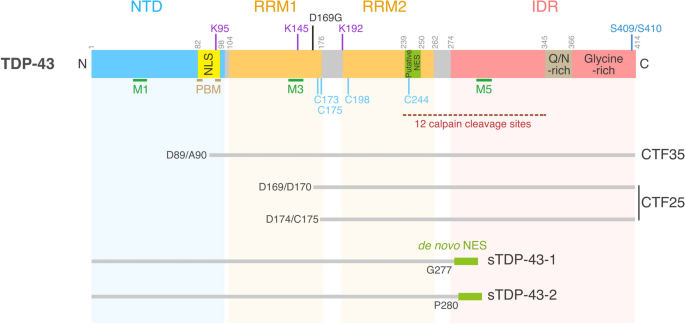
The multimerization status of TDP-43 governs its physiological and pathological functions. Under normal physiological conditions, the N-terminus of TDP-43 mediates homo-oligomerization, which is necessary for its role in RNA regulation (Fig. 2) [35,36,37,38] and can drive liquid–liquid phase separation (LLPS) [39]. The nuclear import receptor importin α recognizes the nuclear localization signal (NLS) embedded in the N-terminus of TDP-43 [40, 40], implying the close coordination of oligomerization and nuclear import. The NLS is subjected to ubiquitination [42], and the ubiquitination at lysine-95 within the NLS likely inhibits nuclear import, targeting TDP-43 for proteolysis in the cytoplasm [43]. The NLS also contains the poly (ADP-Ribose) (PAR)-binding motifs (PBMs) that regulate recruitment to SGs [44]. These observations suggest that the N-terminus domain of TDP-43 mediates the multilayer TDP-43 control of protein multimerization, localization, and stability under normal and stress conditions.
Structure of TDP-43 and its variants. TDP-43 contains 414 amino acid residues and comprises the N-terminal domain (NTD), two RNA recognition motif domains (RRM1 and RRM2), and a C-terminus intrinsically disordered region (IDR). The NTD includes the nuclear localization signal (NLS), overlapping with the poly(ADP-Ribose) (PAR)-binding motifs (PBMs). The NLS is ubiquitinated at K95. The RRM domains are acetylated at K145 and K192, and are subjected to cysteine-mediated disulfide cross-linking at C173, C175, C198 and C244. RRM2 includes a bioinformatically identified putative nuclear export signal (NES). Green bars indicate regions required for mitochondrial localization (M1, M3, and M5). RRM1 contains an ALS-associated mutation, D169G. For more comprehensive views of ALS-associated TDP-43 mutations and ubiquitination sites, see [20] and [42], respectively. The IDR domain contains regions rich in glutamine and asparagine residues (Q/N-rich) and glycine residues (Glycine-rich). The region containing 12 calpain-cleavage sites is indicated by a dashed line. Amino acid numbers are shown above the protein structure. CTF35 and CTF25 are generated by caspase-dependent cleavage. sTDP-43-1 and sTDP-43-2 are the products of neuronal activity-dependent alternative splicing
RNA binding of TDP-43 is mediated by the two RNA recognition motif (RRM) domains (RRM1 and RRM2) that reside in the middle of the primary protein structure [11]. RRM1 and RRM2 have differential affinities with different types of RNAs and, under stress conditions, have distinct functions in the assembly and maintenance of nuclear TDP-43 granules [45]. The RRM2 contains a putative nuclear export signal (NES) predicted via bioinformatics [40, 46, 47]. Mutations in the putative NES or inhibition of the nuclear export receptor exportin-1 (XPO1) by leptomycin B treatment lead to nuclear TDP-43 granule formation [40, 45]. However, independent studies have failed to establish TDP-43 as a direct substrate of XPO1 [47,48,49], and exact mechanisms for the nuclear TDP-43 granule formation caused either by the mutation of the putative NES or leptomycin B treatment remined to be clarified. The RRM domains are also dominant sites of acetylation and cysteine-mediated disulfide cross-linking, two processes that impair the RNA-regulatory functions of TDP-43 [50,51,52].
The IDR in the C-terminus comprises a glycine-rich domain and a region enriched in glutamine (Q) and asparagine (N). Proteins with IDRs reversibly phase separate into droplets; however, such assembly of IDR proteins may become irreversible when they aggregate due to mutations, prolonged stress, or changes in protein concentration [53]. Supporting this view, most of the ALS-linked TDP-43 mutations are found in this C-terminal IDR [19]. Peptides in this region can efficiently form amyloid-like fibrils in vitro that can exhibit prion-like infectious seeding ability in cells expressing the soluble TDP-43 [54,55,56,57]. Under pathological conditions, TDP-43 is hyperphosphorylated, ubiquitinated, and cleaved to generate aggregation-prone C-terminal fragments (CTFs) (Fig. 2) [58,59,60,61]. Intracellular aggregation of the full-length TDP-43 that is recognized by the antibody against S409/S410 phosphorylation (p409/410) precedes the generation of TDP-43 CTFs, suggesting that CTFs are not essential for the formation of intracellular TDP-43 aggregates [62].
TDP-43 granules in the nucleus and cytoplasm
Nuclear bodies (NBs)
In mammalian cells, both endogenous TDP-43 and exogenously expressed TDP-43 at a normal endogenous level show a demixed distribution as rounded particles in the nucleus [7, 63]. TDP-43 is a known component of several kinds of membraneless nuclear structures (i.e. nuclear bodies, or NBs), which are enriched with specific nuclear factors in continuous exchange with the surrounding nucleoplasm [64], such as paraspeckles (PSs) [65] and Cajal bodies [66,67,68]. When incorporated into the PSs, TDP-43 is prevented from regulating alternative polyadenylation of pluripotency factor mRNAs in embryonic stem cells, thereby influencing cellular differentiation [69]. On the inactive X chromosome, a set of RNA-binding proteins including TDP-43 form heteromeric condensates with the long non-coding RNA Xist to initiate and maintain gene silencing [70]. These observations elucidate the close linking of the intranuclear phase transition of TDP-43 to its RNA-regulatory roles. The granular appearance of nuclear TDP-43 is enhanced when cells are under stress, assembling dynamic and reversible TDP-43-containing NBs [45, 51, 71]. Upon arsenite treatment, TDP-43 associates with distinct RNA species, such as long non-coding RNA NEAT1 or short tRNAs, for NB assembly via its two RRM domains [45]. Sequestration of TDP-43 into stress-induced NBs may be a neuroprotective strategy, because the recruitment of TDP-43 to NBs is compromised by the ALS-causing D169G mutation in RRM1, resulting in the incorporation of the mutant TDP-43 into SGs in the cytoplasm [45]. TDP-43 is colocalized with PSs in the spinal motor neurons of sporadic ALS patients [67], and excessive PS formation has been observed in ALS-FUS [72]. Although PS hyper-assembly has been shown to have a protective effect [34], the significance of TDP-43 recruitment to PSs in ALS pathology remains elusive. An intranuclear spherical shell structure that is formed by an RNA-binding-deficient TDP-43 and includes HSP70 chaperones in the core was recently identified and termed anisosome [73]. This HSP70 chaperone-dependent droplet harbors a liquid crystalline property and prevents RNA-free TDP-43 from forming round cytoplasmic droplets and converting into gel/solid-states, which might be precursors of the TDP-43 aggregates observed in neurodegenerative diseases, including ALS. Besides its RNA-regulatory roles, TDP-43 is also implicated in DNA damage repair. TDP-43 depletion causes an accumulation of DNA double-strand breaks (DSBs), while TDP-43 overexpression is protective against DSBs [16, 74,75,76,77]. TDP-43 is rapidly recruited at DSB sites upon induction of DNA damage to stably interact with factors regulating DNA damage response and non-homologous end joining [74, 75]. The association between the role of TDP-43 in DNA damage repair and TDP-43-containing NB assembly remains to be determined.
RNP transport granules
Membraneless organelles formed via condensation of protein–RNA complexes, or phase transition, is relevant for the long-range transport of biomolecules particularly for large-sized and polarized cell types, including motor neurons, where transcription in the nucleus and translation at the synapse can be as far apart as the entire length of the spinal cord, arms, or legs. A fraction of cytoplasmic TDP-43 in neuronal cells functions as a component of RNP granules that undergo microtubule-dependent transport along the axon [12, 78]. Exogenous expression of human TDP-43 in the fly motor neurons and rodent primary cortical neurons results in the formation of RNP granules containing human TDP-43 in the axons [12, 78]. The TDP-43-containing RNP granules are transported bidirectionally, with brief pauses, for long distances, and display liquid-like properties such as fusion, fission, and exchange of TDP-43 with the cytoplasmic soluble TDP-43 pool. The biophysical traits of these TDP-43 granules vary depending on the axonal location; the TDP-43 granules in the mid-axon display a more enhanced motility, a rapid molecular exchange rate and higher sphericity, while those in the proximal axon tend to have limited motility, a lower molecular exchange rate, and more irregular contours [78]. LLPS mediates TDP-43 granule formation in the mid-axon; treatment with 1,6-hexanediol, which disrupts weak hydrophobic interactions in RNP granules [79], rapidly and reversibly dissolves TDP-43 granules in the mid-axon without affecting the integrity of those in the proximal axons. Remarkably, the ALS-linked mutations in the IDR increase the viscosity of the granules and promote retrograde, but not anterograde, transport, resulting in the accumulation of TDP-43-containg granules in the proximal axons [12, 78]. TDP-43 also displays granular localization in the dendritic arbors, enhanced by neuronal depolarization [14]. ALS-linked TDP-43 mutations reduced the depolarization-dependent dendritic localization [80]. Overall, the liquid-like properties of TDP-43-containing RNP transport granules are a critical determinant of the distance that mRNAs can travel along the axons and dendrites, and loss of these properties may underlie ALS pathology by affecting local proteomes in axons and dendrites of motor neurons.
SGs
To sustain cell survival, global repression of translation occurs in response to cellular stresses. SGs are cytoprotective membraneless organelles comprising RNA–protein complexes, and seen in the cytoplasm of cells under stress [81, 82]. TDP-43 is not a ubiquitous component of SGs, but is recruited in response to many, but not all, stressors [51, 83,84,85]. The recruitment of TDP-43 to SGs is promoted by the binding of PAR, a negatively charged biopolymer, to the PAR-binding motif (PBM) embedded in the NLS of TDP-43 (Fig. 2) [44]. The incorporation of TDP-43 into SGs protects it against pathological phosphorylation of the IDR at S409/S410, as shown by the formation of cytoplasmic granules distinct from SGs under stress by TDP-43 mutants defective in binding to PAR, and more prone to show the pathological phosphorylation [44]. Upon stress, the amount of TDP-43 recruited to SGs is also influenced by the assembly of TDP-43-containing NBs in the nucleus. The stress-dependent recruitment of TDP-43 to NBs is diminished by the ALS-causing D169G mutation within RRM1, while the formation of TDP-43-containing SGs is conversely significantly enhanced in the cytoplasm, raising the possibility that assembly of TDP-43-containing NBs works as the first line of defense against stress to prevent excessive recruitment and accumulation of TDP-43 in cytoplasmic SGs [45]. Although recruitment of TDP-43 to SGs may in the short term be beneficial, exposure to high levels of stress for a prolonged period leaves aggregates of pathologically phosphorylated TDP-43 after SG resolution [44]. Moreover, chronic optogenetic induction of SG assembly leads eventually to the deposition of pathologically phosphorylated TDP-43-containing aggregates and causes cytotoxicity [85]. These observations suggest that TDP-43 having experienced a prolonged SG incorporation may become a precursor for pathological TDP-43 aggregates. On the other hand, recent reports have described pathological TDP-43 granules devoid of SG-resident proteins and associated with cytotoxicity [6, 7, 44]. The extent of the contribution by SGs to the formation of pathological TDP-43 aggregates in ALS and a subtype of frontotemporal lobar degeneration (FTLD-TDP) remains to be evaluated.
TDP-43 in myo-granules
Cytoplasmic TDP-43 granules have been shown to play important physiological roles in skeletal muscles. Reduced levels of TDP-43 in skeletal muscles lead to age-related muscle weakness in mice and flies [86,87,88] and to muscle degeneration in zebrafish [89]. While TDP-43 is abundant in the nuclei of C2C12 myoblasts and primary mouse myoblasts, during their differentiation into multinucleated myotubes, cytosolic TDP-43 increases, resulting in the formation of 50–250 nm assemblies with amyloid-like properties, called myo-granules [90]. TDP-43 in myo-granules, which bind to mature mRNAs encoding sarcomeric components, is essential for skeletal muscle cell differentiation in culture and skeletal muscle regeneration in mice [90]. Analogously, in axotomized motor neurons in mice, cytoplasmic TDP-43 granules transiently accumulate that colocalize strongly with the RNA transport granule marker Sauften and moderately with the generic SG marker TIA-1 [91], implying that cytoplasmic TDP-43 redistribution is a part of the normal and physiological response to cellular injury. These findings have demonstrated that the multimerization status of TDP-43 differs depending on physiological cellular conditions and that TDP-43 oligomers can be both beneficial and harmful, depending on the cell-type and possibly the age. The ability of TDP-43 in myo-granules to seed TDP-43 aggregation in motor neurons, via its prion-like ability to spread across neuronal connectivity [92], remains undetermined.
Pathological TDP-43 aggregates
Cytoplasmic inclusions of TDP-43, appearing as rounded or skein-like inclusions in degenerating neurons, are a reliable pathological hallmark of ALS and FTLD-TDP [93, 94]. In FTLD-TDP, TDP-43 pathology can be categorized into four subtypes (types A–D) based on the histology of the TDP-43-positive structures, and disease severity is correlated with the distinct forms of pathological TDP-43 [95, 96]. The distinct histological traits of TDP-43 aggregate suggest multiple pathways for aggregation. Indeed, in cultured cells, cytoplasmic TDP-43 aggregation is driven by at least two distinct pathways upon expression of inherited ALS/FTLD causative genes: RNA-binding protein-mediated LLPS promoting granular-type aggregation and histone deacelylase 6 (HDAC6)-mediated aggresome formation promoting skein-like aggregation [97]. In the spinal cord of patients with ALS, most of the phosphorylated TDP-43 inclusions show significant skein-like immunoreactivity of lysine-145 acetylation in RRM1, which may be promoted by oxidative stress [50]. This implies that the cellular stresses specify the form of TDP-43 aggregates. The dipeptide repeat proteins expressed from the C9orf72 locus carrying repeat expansions, a cause of familial ALS, also causes TDP-43 aggregation [43, 98,99,100,101,102]. Intracellular TDP-43 is aggregated in a self-templating manner when the cultured cells are treated with seeds isolated from the brains of patients with ALS and FTLD-TDP, as well as synthetic peptide-derived TDP-43 CTF fibrils; this suggests that the alternate pathological TDP-43 conformations in ALS and FTLD-TDP could also arise from the prion-like properties of TDP-43 [57, 62, 103, 104]. Of note, in a synthetic peptide-dependent TDP-43 aggregation assay, phospho-deficient mutations in the CTF have little effect on the aggregation propensity of TDP-43 [57], hinting at the possibility of a toxic TDP-43 variant undetectable via conventional phospho-CTF immunostaining. A recent report of neurotoxicity observed in the presence of cytoplasmic TDP-43 granules lacking S409/S410 phosphorylation supports this view [6]. Intercellular transfer of TDP-43 has been demonstrated in vitro [103,104,105,106]. In vivo transmission of TDP-43 along neuronal connectivity was first demonstrated using animal models by injecting human brain-derived FTLD-TDP extract into mouse brain overexpressing TDP-43 [92]. Microvesicle/exosome-dependent intercellular TDP-43 transport might mediate transmission and de novo formation of pathological TDP-43 aggregates in a distant brain area [62, 103], which may not be dependent on SG formation because the assembled phosphorylated TDP-43-positive inclusions did not colocalize with SG markers in immunofluorescence [92].
TDP-43 and selective neuronal vulnerability
In sporadic ALS, pathological TDP-43 phosphorylation recognized by the pS409/S410 antibody is observed throughout many areas of the central nervous system (CNS) [107], showing that ALS is a multisystem TDP-43 proteinopathy. Studies using animal models also suggest that the deposition of cytoplasmic aggregates does not always accompany TDP-43 neurotoxicity [24, 108]. These observations question the extent to which cytoplasmic TDP-43 aggregates, as end products, explain the selective vulnerability of motor neurons in ALS. Although the answer is largely elusive at present, several studies have begun to reveal neuron-specific properties and regulatory function of TDP-43, including the alternative splicing of TDP-43 mRNA and proteolytic cleavage of TDP-43 protein. In both physiology and pathology, motor neuron-specific properties of TDP-43 could modify functions of TDP-43-containing membraneless organelles, and would be key to explaining selective vulnerability of motor neurons in ALS.
Neural activity-dependent alternative splicing of TDP-43
Motor neurons are large cells with large membrane surface areas, which demand high levels of energy for generating an action potential, as well as for maintaining homeostatic ionic gradients across the plasma membrane in the resting state. The energetic demand is particularly high in large fast-fatigable motor neurons, the most vulnerable neuronal type in ALS [109], and these neurons are prone to hyperexcitation due to low GABAA and glycine receptor expression [110]. Accordingly, cortical hyperexcitability and mislocalization of TDP-43 are salient and highly conserved features of ALS. Recently, hyperexcitability was found to lead to the expression of two shortened splice isoforms of TDP-43 (sTDP-43-1 and sTDP-43-2), wherein the entire glycine-rich domain of TDP-43 is replaced by short tails generated by the inclusion of a new exon encoding a unique 18-amino acid C-terminus not found in the wild-type full-length TDP-43 (Fig. 2) [111]. The sTDP-43-1 isoform is prone to cytoplasmic localization due to a de novo NES created by the neural activity-dependent splicing. In rodent primary mixed cortical neurons, overexpression of sTDP-43-1 is neurotoxic, and promotes the cytoplasmic deposition and nuclear clearance of endogenous TDP-43 through N-terminus- and/or RRM-mediated aggregation. Moreover, significant expression of the sTDP-43-1 isoform is detectable in several different regions of the human CNS, including spinal motor neurons. Major challenges in proving a causal link between neuronal hyperexcitability and cytoplasmic TDP-43 aggregation in the selective vulnerability of human motor neurons in ALS include the elucidation of mechanisms underlying the neural activity-dependent alternative splicing and effects of the sTDP-43 isoforms on TDP-43-containing membraneless organelles.
Ca 2+ -dependent cleavage of TDP-43
Elevated intracellular Ca2+ levels caused by excessive stimulation of glutamate receptors have been implicated in the selective vulnerability of neurons in ALS [112,113,114,115]. Glutamate-mediated excitotoxicity is associated with the stoichiometry of both Ca2+-permeable subunits GluA1, 3, and 4 and the typically Ca2+-impermeable subunit GluA2 [116,117,118,119,120,121,122,123,124]. TDP-43 pathology in sporadic ALS is correlated with the downregulation of an RNA-editing enzyme, adenosine deaminase acting on RNA 2 (ADAR2), which edits GluA2 pre-mRNA to produce the Ca2+-impermeable GluA2 subunit [125,126,127,128], suggesting that exaggerated Ca2+ influx could lead to TDP-43 aggregation. The observation that TDP-43 is subject to proteolysis in a Ca2+ -dependent manner provides a mechanistic link between the exaggerated Ca2+ influx and TDP-43 aggregation [129] (Fig. 3). In mouse motor neurons, TDP-43 is cleaved by calpains (Ca2+-dependent cysteine proteases) at the C-terminus, which is distinct from the caspase-dependent cleavage sites (Fig. 2) [61, 130], to generate an aggregation-prone proteolytic product [129]. Calpain-dependent TDP-43 fragments are detectable in the spinal cord and brain of patients with ALS. Thus, the calpain-mediated TDP-43 cleavage is a crucial downstream target of an excessive intracellular Ca2+ load, potentially affecting TDP-43-containing membraineless organelles via generation of aggregation-prone TDP-43 fragments and contributing to the selective vulnerability of motor neurons in ALS.
Possible upstream mechanisms that promote pathological TDP-43 phase transition and aggregation. Cellular redox activity influences self-interaction, aggregation, and cytotoxicity of TDP-43 by promoting lysine acetylation, methionine oxidation, and cysteine disulfide bonding of TDP-43. An exaggerated Ca2+ influx elevates cytosolic Ca2+ concentration, which leads to calpain-mediated cleavage of TDP-43 that, in turn, generates aggregation-prone TDP-43 fragments. These post-translational TDP-43 modifications may frequently occur at mitochondria-rich subcellular compartments, including, but not restricted to, pre-synaptic axon swellings (illustrated), where ROS is actively produced due to high energy metabolism and efficient Ca2+ buffering is needed. Neuronal hyperexcitability drives the expression of two shortened splice isoforms of TDP-43 (sTDP-43-1 and sTDP-43-1), which are also aggregation-prone. The mechanisms underpinning how neuronal hyperexcitability drives TDP-43 alternative splicing are elusive. ER endoplasmic reticulum, MT microtubule, VGCC voltage-gated calcium channel
TDP-43 and mitochondria
The high energetic demand of motor neurons is met by ATP provision via mitochondrial metabolism. Inevitably, mitochondria are major sources of reactive oxygen species (ROS) within most mammalian cells, and modulation of cellular redox activity has been shown to influence self-interaction, aggregation, and cytotoxicity of TDP-43 [131,132,133]. Oxidative stress-induced cysteine oxidation and disulfide bond formation in the RRM domains lead to impaired splicing function and reduced solubility of TDP-43 [51, 52]. The RRM1 is also acetylated at lysine-145 in response to oxidative stress by histone acetylase cAMP response element-binding (CREB)-binding protein (CBP), impairing RNA-binding and promoting deposition of skein-like TDP-43 inclusions [50]. Furthermore, methionine oxidization in the IDR of TDP-43 affects its ability to self-assemble into an oligomeric structure [134]. These observations suggest that high mitochondrial metabolism could alter the phase behavior of TDP-43 via ROS production especially in a mitochondrial-rich subcellular compartment, such as pre-synaptic terminals [135, 135] (Fig. 3). Another essential function of mitochondria is the maintenance of cellular Ca2+ homeostasis, which is regulated through interactions between the endoplasmic reticulum (ER) and mitochondria via a region in the ER called mitochondria-associated membranes (MAM) [137, 138]. MAM is implicated in neurodegenerative diseases [139,140,141,142,143,144,145]. Tightening of MAM-mediated ER-mitochondria contacts can cause Ca2+ overload in mitochondria, whereas its loosening diminishes mitochondrial ATP synthesis and increases cytosolic Ca2+ [146,147,148,149]. Increases in cytosolic Ca2+ could in turn induce calpain-mediated cleavage of TDP-43, leading to TDP-43 aggregation [129] (Fig. 3); a direct link between MAM disruption and calpain-dependent TDP-43 cleavage, however, remains to be demonstrated. Taken together, mitochondria are key organelles influencing phase behaviors of TDP-43 through ROS generation and maintenance of Ca2+ homeostasis, whose dysfunction could contribute to selective vulnerability of motor neurons in ALS.
Of note, induced pluripotent stem cell (iPSC)-derived motor neurons from patients carrying TDP-43 mutation display high glutamate-induced Ca2+ release and delayed buffering of cytosolic Ca2+ [150], suggesting that TDP-43 regulates mitochondria, as well as being influenced by them. Indeed, endogenous TDP-43 is detectable in the mitochondria of brain samples with or without FTLD-TDP pathology, using immuno-electron microscopy [151]. Furthermore, in brain samples from patients with FTLD-TDP and ALS-FTLD-TDP, electron dense TDP-43-positive protein aggregates and impaired mitochondrial morphology have been observed [151]. These human studies suggest the mitochondrial roles of TDP-43, but in the studies using cellular and animal models, conflicting results have been reported for direct involvement of TDP-43 in mitochondrial respiratory complex and ATP synthesis [145, 151,152,153,154,155]. Further studies are thus necessary to understand the mitochondrial function of TDP-43 expressed at physiological levels. TDP-43-mediated mitochondrial control has also been suggested from overexpression experiments. Overexpression of TDP-43 disrupts MAM and increases cytosolic Ca2+ at the expense of mitochondrial Ca2+ storage [142]. The TDP-43-dependent MAM disruption is mediated via the activation of GSK -3β, although the mechanism whereby excessive TDP-43 is sensed by GSK-3β remains unknown [142]. In cellular and mice models, overexpression of TDP-43 also leads to invasion of TDP-43 into the mitochondria, the release of mitochondrial DNA (mtDNA) into the cytoplasm, and inflammation driven by the cytoplasmic DNA sensor cyclic guanosine monophosphate (GMP)-AMP synthase (cGAS)/STING pathway [156]. The mitochondrial dysfunctions caused by TDP-43 overexpression may be relevant to an understanding of TDP-43 pathology in ALS, and whether the TDP-43-dependent release of mtDNA and Ca2+ from mitochondria involves TDP-43 phase transition remains an open question.
Stability and dynamics of TDP-43 in motor neurons
Mainly due to anatomical inaccessibility, TDP-43 dynamics have rarely been explored in vivo in mammalian motor neurons. Direct observation of TDP-43 in live motor neurons is, however, feasible in an optical-friendly vertebrate model, zebrafish [23, 24]. We have found that optogenetic oligomerization of TDP-43 with the C-terminally tagged CRY2olig [157] resulted in an efficient increase in cytoplasmic TDP-43 in the spinal motor neurons of zebrafish, leading eventually to the accumulation of cytoplasmic TDP-43 aggregates positive for pS409/S410 immunoreactivity [24]. Intriguingly, in epithelial cells or differentiated myofibers, this light-dependent cytoplasmic shift of TDP-43 is not as efficient or is almost absent. While the mechanism underlying this motor neuron-specific cytoplasmic TDP-43 accumulation remains to be determined, this observation implies that motor neurons possess a unique mechanism for responding to oligomeric TDP-43. One possible explanation for the efficient cytoplasmic TDP-43 accumulation in motor neurons is that oligomeric TDP-43 is more stable in the cytoplasm of motor neurons than in that of other cell types. This idea might be consistent with the observations that TDP-43 has a longer half-life in primary rodent cortical neurons (approximately 18 h) [158], than in fibroblasts, HeLa cell lines (4–12 h) [159], and Neuro2a cell lines (12.6 h) [160]. It is also worth noting that in the cytoplasm, the TDP-43-containing transport granules display differential TDP-43 exchange rates between the mid and proximal regions of axons [78], suggesting that the dynamics and stability of TDP-43 granules differ locally, within the axons and possibly dendrites. The stability and dynamics of TDP-43 in motor neurons thus requires further study. How motor neurons control global and local levels of TDP-43 is a pivotal question to be addressed in the future, and is likely relevant to understanding the selective vulnerability of motor neurons in ALS.
Outlook
Here, we enumerated different forms of TDP-43 granules in different intracellular locations and contexts. However, these various forms of TDP-43 are insufficient in providing a complete understanding of multifaceted TDP-43 dynamics, in healthy and diseased states. Therefore, it is necessary to explore further TDP-43 granules and their regulatory mechanisms that have not yet been discovered. Aberrant phase behavior of TDP-43 in RNP granules results in at least three reasonably conceivable consequences, differing in their gain-/loss-of-function nature: (1) generation of toxic TDP-43 oligomers or aggregates that further propagate via phase transition in a dominant fashion, (2) loss/reduction of proteins that are otherwise normally expressed, due to TDP-43-mediated regulation, and (3) generation of truncated translation products of abnormally spliced transcripts causing proteostatic cellular stress. These multiple phenotypes are likely to occur simultaneously rather than sequentially. Thus, an understanding of the upstream cellular events causing abnormal TDP-43 phase transition may be as important as the conventional gain-of-function versus loss-of-function dichotomy. An emerging figure, while largely omitted from this review, is the multisystem nature of ALS and FTLD, where TDP-43 pathology in degenerating neurons has been linked to extracellular factors, including inflammation, microglial toxicity, and intercellular C9orf72-derived dipeptide transmission [43, 161,162,163]. Studies investigating the precise mechanisms of upstream events of TDP-43 pathology at the multisystem levels therefore warrant intensive efforts and potentially provide effective treatment targets applicable to a wide range of different ALS subtypes and other TDP-43 proteinopathies.
References
Lennox AL et al (2020) Pathogenic DDX3X mutations impair RNA metabolism and neurogenesis during fetal cortical development. Neuron 106(3):404-420e8
Nedelsky NB, Taylor JP (2019) Bridging biophysics and neurology: aberrant phase transitions in neurodegenerative disease. Nat Rev Neurol 15(5):272–286
Ramaswami M, Taylor JP, Parker R (2013) Altered ribostasis: RNA-protein granules in degenerative disorders. Cell 154(4):727–736
Zhang H et al (2015) RNA controls PolyQ protein phase transitions. Mol Cell 60(2):220–230
Elbaum-Garfinkle S et al (2015) The disordered P granule protein LAF-1 drives phase separation into droplets with tunable viscosity and dynamics. Proc Natl Acad Sci USA 112(23):7189–7194
Mann JR et al (2019) RNA binding antagonizes neurotoxic phase transitions of TDP-43. Neuron 102(2):321-338e8
Gasset-Rosa F et al (2019) Cytoplasmic TDP-43 de-mixing independent of stress granules drives inhibition of nuclear import, loss of nuclear TDP-43, and cell death. Neuron 102(2):339-357e7
Ou SH et al (1995) Cloning and characterization of a novel cellular protein, TDP-43, that binds to human immunodeficiency virus type 1 TAR DNA sequence motifs. J Virol 69(6):3584–3596
Acharya KK et al (2006) cis-requirement for the maintenance of round spermatid-specific transcription. Dev Biol 295(2):781–790
Sephton CF et al (2011) Identification of neuronal RNA targets of TDP-43-containing ribonucleoprotein complexes. J Biol Chem 286(2):1204–1215
Buratti E, Baralle FE (2001) Characterization and functional implications of the RNA binding properties of nuclear factor TDP-43, a novel splicing regulator of CFTR exon 9. J Biol Chem 276(39):36337–36343
Alami NH et al (2014) Axonal transport of TDP-43 mRNA granules is impaired by ALS-causing mutations. Neuron 81(3):536–543
Kawahara Y, Mieda-Sato A (2012) TDP-43 promotes microRNA biogenesis as a component of the Drosha and Dicer complexes. Proc Natl Acad Sci USA 109(9):3347–3352
Wang IF et al (2008) TDP-43, the signature protein of FTLD-U, is a neuronal activity-responsive factor. J Neurochem 105(3):797–806
Liu W et al (2020) LncRNA Malat1 inhibition of TDP43 cleavage suppresses IRF3-initiated antiviral innate immunity. Proc Natl Acad Sci USA 117(38):23695–23706
Hill SJ et al (2016) Two familial ALS proteins function in prevention/repair of transcription-associated DNA damage. Proc Natl Acad Sci USA 113(48):E7701–E7709
Liu EY et al (2019) Loss of nuclear TDP-43 is associated with decondensation of LINE retrotransposons. Cell Rep 27(5):1409-1421e6
Ling SC, Polymenidou M, Cleveland DW (2013) Converging mechanisms in ALS and FTD: disrupted RNA and protein homeostasis. Neuron 79(3):416–438
Pesiridis GS, Lee VM, Trojanowski JQ (2009) Mutations in TDP-43 link glycine-rich domain functions to amyotrophic lateral sclerosis. Hum Mol Genet 18(R2):R156–R162
Prasad A et al (2019) Molecular mechanisms of TDP-43 misfolding and pathology in amyotrophic lateral sclerosis. Front Mol Neurosci 12:25
Guo L, Shorter J (2015) It’s raining liquids: RNA tunes viscoelasticity and dynamics of membraneless organelles. Mol Cell 60(2):189–192
Saxena S, Caroni P (2011) Selective neuronal vulnerability in neurodegenerative diseases: from stressor thresholds to degeneration. Neuron 71(1):35–48
Svahn AJ et al (2018) Nucleo-cytoplasmic transport of TDP-43 studied in real time: impaired microglia function leads to axonal spreading of TDP-43 in degenerating motor neurons. Acta Neuropathol 136(3):445–459
Asakawa K, Handa H, Kawakami K (2020) Optogenetic modulation of TDP-43 oligomerization accelerates ALS-related pathologies in the spinal motor neurons. Nat Commun 11(1):1004
Klim JR et al (2019) ALS-implicated protein TDP-43 sustains levels of STMN2, a mediator of motor neuron growth and repair. Nat Neurosci 22(2):167–179
Melamed Z et al (2019) Premature polyadenylation-mediated loss of stathmin-2 is a hallmark of TDP-43-dependent neurodegeneration. Nat Neurosci 22(2):180–190
Honda D et al (2014) Corrigendum to “The ALS/FTLD-related RNA-binding proteins TDP-43 and FUS have common downstream RNA targets in cortical neurons” [FEBS Open Bio 4 (2014) 1–10]. FEBS Open Bio 4:1030
Lagier-Tourenne C et al (2012) Divergent roles of ALS-linked proteins FUS/TLS and TDP-43 intersect in processing long pre-mRNAs. Nat Neurosci 15(11):1488–1497
Polymenidou M et al (2011) Long pre-mRNA depletion and RNA missplicing contribute to neuronal vulnerability from loss of TDP-43. Nat Neurosci 14(4):459–468
Tollervey JR et al (2011) Characterizing the RNA targets and position-dependent splicing regulation by TDP-43. Nat Neurosci 14(4):452–458
Xiao S et al (2011) RNA targets of TDP-43 identified by UV-CLIP are deregulated in ALS. Mol Cell Neurosci 47(3):167–180
Herzog JJ et al (2020) TDP-43 dysfunction restricts dendritic complexity by inhibiting CREB activation and altering gene expression. Proc Natl Acad Sci USA 117(21):11760–11769
Nagano S et al (2020) TDP-43 transports ribosomal protein mRNA to regulate axonal local translation in neuronal axons. Acta Neuropathol 140(5):695–713
Shelkovnikova TA et al (2018) Protective paraspeckle hyper-assembly downstream of TDP-43 loss of function in amyotrophic lateral sclerosis. Mol Neurodegener 13(1):30
Shiina Y et al (2010) TDP-43 dimerizes in human cells in culture. Cell Mol Neurobiol 30(4):641–652
Zhang YJ et al (2013) The dual functions of the extreme N-terminus of TDP-43 in regulating its biological activity and inclusion formation. Hum Mol Genet 22(15):3112–3122
Afroz T et al (2017) Functional and dynamic polymerization of the ALS-linked protein TDP-43 antagonizes its pathologic aggregation. Nat Commun 8(1):45
Jiang LL et al (2017) The N-terminal dimerization is required for TDP-43 splicing activity. Sci Rep 7(1):6196
Schmidt HB, Rohatgi R (2016) In vivo formation of vacuolated multi-phase compartments lacking membranes. Cell Rep 16(5):1228–1236
Winton MJ et al (2008) Disturbance of nuclear and cytoplasmic TAR DNA-binding protein (TDP-43) induces disease-like redistribution, sequestration, and aggregate formation. J Biol Chem 283(19):13302–13309
Nishimura AL et al (2010) Nuclear import impairment causes cytoplasmic trans-activation response DNA-binding protein accumulation and is associated with frontotemporal lobar degeneration. Brain 133(Pt 6):1763–1771
Hans F et al (2018) Identification and characterization of ubiquitinylation sites in TAR DNA-binding protein of 43 kDa (TDP-43). J Biol Chem 293(41):16083–16099
Khosravi B et al (2020) Cell-to-cell transmission of C9orf72 poly-(Gly-Ala) triggers key features of ALS/FTD. EMBO J 39(8):e102811
McGurk L et al (2018) Poly (ADP-ribose) prevents pathological phase separation of TDP-43 by promoting liquid demixing and stress granule localization. Mol cell 71(5):703-717e9
Wang C et al (2020) Stress induces dynamic, cytotoxicity-antagonizing TDP-43 nuclear bodies via paraspeckle LncRNA NEAT1-mediated liquid-liquid phase separation. Mol Cell 79(3):443-458e7
Ayala YM et al (2008) Structural determinants of the cellular localization and shuttling of TDP-43. J Cell Sci 121(Pt 22):3778–3785
Ederle H et al (2018) Nuclear egress of TDP-43 and FUS occurs independently of Exportin-1/CRM1. Sci Rep 8(1):7084
Archbold HC et al (2018) TDP43 nuclear export and neurodegeneration in models of amyotrophic lateral sclerosis and frontotemporal dementia. Sci Rep 8(1):4606
Pinarbasi ES et al (2018) Active nuclear import and passive nuclear export are the primary determinants of TDP-43 localization. Sci Rep 8(1):7083
Cohen TJ et al (2015) An acetylation switch controls TDP-43 function and aggregation propensity. Nat Commun 6:5845
Cohen TJ et al (2012) Redox signalling directly regulates TDP-43 via cysteine oxidation and disulphide cross-linking. EMBO J 31(5):1241–1252
Shodai A et al (2013) Aberrant assembly of RNA recognition motif 1 links to pathogenic conversion of TAR DNA-binding protein of 43 kDa (TDP-43). J Biol Chem 288(21):14886–14905
Batlle C et al (2020) hnRNPDL phase separation is regulated by alternative splicing and disease-causing mutations accelerate its aggregation. Cell Rep 30(4):1117-1128e5
Chen AK et al (2010) Induction of amyloid fibrils by the C-terminal fragments of TDP-43 in amyotrophic lateral sclerosis. J Am Chem Soc 132(4):1186–1187
Guo W et al (2011) An ALS-associated mutation affecting TDP-43 enhances protein aggregation, fibril formation and neurotoxicity. Nat Struct Mol Biol 18(7):822–830
Jiang LL et al (2013) Structural transformation of the amyloidogenic core region of TDP-43 protein initiates its aggregation and cytoplasmic inclusion. J Biol Chem 288(27):19614–19624
Shimonaka S et al (2016) Templated aggregation of TAR DNA-binding protein of 43 kDa (TDP-43) by seeding with TDP-43 peptide fibrils. J Biol Chem 291(17):8896–8907
Arai T et al (2010) Phosphorylated and cleaved TDP-43 in ALS, FTLD and other neurodegenerative disorders and in cellular models of TDP-43 proteinopathy. Neuropathology 30(2):170–181
Hasegawa M et al (2008) Phosphorylated TDP-43 in frontotemporal lobar degeneration and amyotrophic lateral sclerosis. Ann Neurol 64(1):60–70
Hasegawa M et al (2011) Molecular dissection of TDP-43 proteinopathies. J Mol Neurosci 45(3):480–485
Li Q et al (2015) The cleavage pattern of TDP-43 determines its rate of clearance and cytotoxicity. Nat Commun 6:6183
Nonaka T et al (2013) Prion-like properties of pathological TDP-43 aggregates from diseased brains. Cell Rep 4(1):124–134
Dammer EB et al (2012) Coaggregation of RNA-binding proteins in a model of TDP-43 proteinopathy with selective RGG motif methylation and a role for RRM1 ubiquitination. PLoS ONE 7(6):e38658
Stanek D, Fox AH (2017) Nuclear bodies: news insights into structure and function. Curr Opin Cell Biol 46:94–101
West JA et al (2016) Structural, super-resolution microscopy analysis of paraspeckle nuclear body organization. J Cell Biol 214(7):817–830
Naganuma T et al (2012) Alternative 3ʹ-end processing of long noncoding RNA initiates construction of nuclear paraspeckles. EMBO J 31(20):4020–4034
Nishimoto Y et al (2013) The long non-coding RNA nuclear-enriched abundant transcript 1_2 induces paraspeckle formation in the motor neuron during the early phase of amyotrophic lateral sclerosis. Mol Brain 6:31
Izumikawa K et al (2019) TDP-43 regulates site-specific 2’-O-methylation of U1 and U2 snRNAs via controlling the Cajal body localization of a subset of C/D scaRNAs. Nucleic Acids Res 47(5):2487–2505
Modic M et al (2019) Cross-regulation between TDP-43 and paraspeckles promotes pluripotency-differentiation transition. Mol cell. 74(5):951-965e13
Pandya-Jones A et al (2020) A protein assembly mediates Xist localization and gene silencing. Nature 587(7832):145–151
Udan-Johns M et al (2014) Prion-like nuclear aggregation of TDP-43 during heat shock is regulated by HSP40/70 chaperones. Hum Mol Genet 23(1):157–170
An H et al (2019) ALS-linked FUS mutations confer loss and gain of function in the nucleus by promoting excessive formation of dysfunctional paraspeckles. Acta Neuropathol Commun 7(1):7
Yu H et al (2020) HSP70 chaperones RNA-free TDP-43 into anisotropic intranuclear liquid spherical shells. Science 371(6529):eabb4309
Mitra J et al (2019) Motor neuron disease-associated loss of nuclear TDP-43 is linked to DNA double-strand break repair defects. Proc Natl Acad Sci USA 116(10):4696–4705
Konopka A et al (2020) Impaired NHEJ repair in amyotrophic lateral sclerosis is associated with TDP-43 mutations. Mol Neurodegener 15(1):51
Garcia-Muse T, Aguilera A (2019) R Loops: from physiological to pathological roles. Cell 179(3):604–618
Gianini M et al (2020) TDP-43 mutations link amyotrophic lateral sclerosis with R-loop homeostasis and R loop-mediated DNA damage. PLoS Genet 16(12):e1009260
Gopal PP et al (2017) Amyotrophic lateral sclerosis-linked mutations increase the viscosity of liquid-like TDP-43 RNP granules in neurons. Proc Natl Acad Sci USA 114(12):E2466–E2475
Kroschwald S et al (2015) Promiscuous interactions and protein disaggregases determine the material state of stress-inducible RNP granules. Elife 4:e06807
Liu-Yesucevitz L et al (2014) ALS-linked mutations enlarge TDP-43-enriched neuronal RNA granules in the dendritic arbor. J Neurosci 34(12):4167–4174
Hyman AA, Weber CA, Julicher F (2014) Liquid-liquid phase separation in biology. Annu Rev Cell Dev Biol 30:39–58
Wippich F et al (2013) Dual specificity kinase DYRK3 couples stress granule condensation/dissolution to mTORC1 signaling. Cell 152(4):791–805
Dewey CM et al (2011) TDP-43 is directed to stress granules by sorbitol, a novel physiological osmotic and oxidative stressor. Mol Cell Biol 31(5):1098–1108
Li YR et al (2013) Stress granules as crucibles of ALS pathogenesis. J Cell Biol 201(3):361–372
Zhang P et al (2019) Chronic optogenetic induction of stress granules is cytotoxic and reveals the evolution of ALS-FTD pathology. Elife. 20(8):e39578
Kraemer BC et al (2010) Loss of murine TDP-43 disrupts motor function and plays an essential role in embryogenesis. Acta Neuropathol 119(4):409–419
Diaper DC et al (2013) Drosophila TDP-43 dysfunction in glia and muscle cells cause cytological and behavioural phenotypes that characterize ALS and FTLD. Hum Mol Genet 22(19):3883–3893
Llamusi B et al (2013) Muscleblind, BSF and TBPH are mislocalized in the muscle sarcomere of a Drosophila myotonic dystrophy model. Dis Model Mech 6(1):184–196
Schmid B et al (2013) Loss of ALS-associated TDP-43 in zebrafish causes muscle degeneration, vascular dysfunction, and reduced motor neuron axon outgrowth. Proc Natl Acad Sci USA 110(13):4986–4991
Vogler TO et al (2018) TDP-43 and RNA form amyloid-like myo-granules in regenerating muscle. Nature 563(7732):508–513
Moisse K et al (2009) Divergent patterns of cytosolic TDP-43 and neuronal progranulin expression following axotomy: implications for TDP-43 in the physiological response to neuronal injury. Brain Res 1249:202–211
Porta S et al (2018) Patient-derived frontotemporal lobar degeneration brain extracts induce formation and spreading of TDP-43 pathology in vivo. Nat Commun 9(1):4220
Arai T et al (2006) TDP-43 is a component of ubiquitin-positive tau-negative inclusions in frontotemporal lobar degeneration and amyotrophic lateral sclerosis. Biochem Biophys Res Commun 351(3):602–611
Neumann M et al (2006) Ubiquitinated TDP-43 in frontotemporal lobar degeneration and amyotrophic lateral sclerosis. Science 314(5796):130–133
Mackenzie IR et al (2011) A harmonized classification system for FTLD-TDP pathology. Acta Neuropathol 122(1):111–113
Laferriere F et al (2019) TDP-43 extracted from frontotemporal lobar degeneration subject brains displays distinct aggregate assemblies and neurotoxic effects reflecting disease progression rates. Nat Neurosci 22(1):65–77
Watanabe S et al (2020) Aggresome formation and liquid-liquid phase separation independently induce cytoplasmic aggregation of TAR DNA-binding protein 43. Cell Death Dis 11(10):909
Chew J et al (2019) Aberrant deposition of stress granule-resident proteins linked to C9orf72-associated TDP-43 proteinopathy. Mol Neurodegener 14(1):9
Chew J et al (2015) C9orF72 repeat expansions in mice cause TDP-43 pathology, neuronal loss, and behavioral deficits. Science 348(6239):1151–1154
Jiang J et al (2016) Gain of toxicity from ALS/FTD-linked repeat expansions in C9orF72 Is alleviated by antisense oligonucleotides targeting GGGGCC-containing RNAs. Neuron 90(3):535–550
Liu Y et al (2016) C9orf72 BAC mouse model with motor deficits and neurodegenerative features of ALS/FTD. Neuron 90(3):521–534
Cook CN et al (2020) C9orf72 poly(GR) aggregation induces TDP-43 proteinopathy. Sci Transl Med 12(559):eabb3774
Feiler MS et al (2015) TDP-43 is intercellularly transmitted across axon terminals. J Cell Biol 211(4):897–911
Smethurst P et al (2016) In vitro prion-like behaviour of TDP-43 in ALS. Neurobiol Dis 96:236–247
Peled S et al (2017) Single cell imaging and quantification of TDP-43 and alpha-synuclein intercellular propagation. Sci Rep 7(1):544
Zeineddine R et al (2017) Flow cytometric measurement of the cellular propagation of TDP-43 aggregation. Prion 11(3):195–204
Tsuji H et al (2012) Molecular analysis and biochemical classification of TDP-43 proteinopathy. Brain 135(Pt 11):3380–3391
Arnold ES et al (2013) ALS-linked TDP-43 mutations produce aberrant RNA splicing and adult-onset motor neuron disease without aggregation or loss of nuclear TDP-43. Proc Natl Acad Sci USA 110(8):E736–E745
Roselli F, Caroni P (2014) Modeling neuronal vulnerability in ALS. Neuron 83(4):758–760
Lorenzo LE et al (2006) Differential expression of GABAA and glycine receptors in ALS-resistant vs. ALS-vulnerable motoneurons: possible implications for selective vulnerability of motoneurons. Eur J Neurosci 12:3161–3170
Weskamp K et al (2020) Shortened TDP43 isoforms upregulated by neuronal hyperactivity drive TDP43 pathology in ALS. J Clin Invest 130(3):1139–1155
Rothstein JD et al (1990) Abnormal excitatory amino acid metabolism in amyotrophic lateral sclerosis. Ann Neurol 28(1):18–25
Rothstein JD, Martin LJ, Kuncl RW (1992) Decreased glutamate transport by the brain and spinal cord in amyotrophic lateral sclerosis. N Engl J Med 326(22):1464–1468
Couratier P et al (1993) Cell culture evidence for neuronal degeneration in amyotrophic lateral sclerosis being linked to glutamate AMPA/kainate receptors. Lancet 341(8840):265–268
Rothstein JD et al (1995) Selective loss of glial glutamate transporter GLT-1 in amyotrophic lateral sclerosis. Ann Neurol 38(1):73–84
Vandenberghe W et al (2000) AMPA receptor current density, not desensitization, predicts selective motoneuron vulnerability. J Neurosci 20(19):7158–7166
Van Den Bosch L et al (2000) Ca (2+)-permeable AMPA receptors and selective vulnerability of motor neurons. J Neurol Sci 180(1–2):29–34
Kawahara Y et al (2004) Glutamate receptors: RNA editing and death of motor neurons. Nature 427(6977):801
Rembach A et al (2004) Antisense peptide nucleic acid targeting GluR3 delays disease onset and progression in the SOD1 G93A mouse model of familial ALS. J Neurosci Res 77(4):573–582
Kwak S, Kawahara Y (2005) Deficient RNA editing of GluR2 and neuronal death in amyotropic lateral sclerosis. J Mol Med (Berl) 83(2):110–120
Petri S et al (2005) The cellular mRNA expression of GABA and glutamate receptors in spinal motor neurons of SOD1 mice. J Neurol Sci 238(1–2):25–30
Kuner R et al (2005) Late-onset motoneuron disease caused by a functionally modified AMPA receptor subunit. Proc Natl Acad Sci USA 102(16):5826–5831
Tortarolo M et al (2006) Glutamate AMPA receptors change in motor neurons of SOD1G93A transgenic mice and their inhibition by a noncompetitive antagonist ameliorates the progression of amytrophic lateral sclerosis-like disease. J Neurosci Res 83(1):134–146
Zhao P et al (2008) Altered presymptomatic AMPA and cannabinoid receptor trafficking in motor neurons of ALS model mice: implications for excitotoxicity. Eur J Neurosci 27(3):572–579
Melcher T et al (1996) A mammalian RNA editing enzyme. Nature 379(6564):460–464
Higuchi M et al (2000) Point mutation in an AMPA receptor gene rescues lethality in mice deficient in the RNA-editing enzyme ADAR2. Nature 406(6791):78–81
Aizawa H et al (2010) TDP-43 pathology in sporadic ALS occurs in motor neurons lacking the RNA editing enzyme ADAR2. Acta Neuropathol 120(1):75–84
Hideyama T et al (2012) Co-occurrence of TDP-43 mislocalization with reduced activity of an RNA editing enzyme, ADAR2, in aged mouse motor neurons. PLoS ONE 7(8):e43469
Yamashita T et al (2012) A role for calpain-dependent cleavage of TDP-43 in amyotrophic lateral sclerosis pathology. Nat Commun 3:1307
Zhang YJ et al (2007) Progranulin mediates caspase-dependent cleavage of TAR DNA binding protein-43. J Neurosci 27(39):10530–10534
Oberstadt M et al (2018) TDP-43 self-interaction is modulated by redox-active compounds Auranofin, Chelerythrine and Riluzole. Sci Rep 8(1):2248
Parakh S et al (2020) The redox activity of protein disulfide isomerase inhibits ALS phenotypes in cellular and zebrafish models. Iscience. 23(5):101097
Zuo X et al (2021) TDP-43 aggregation induced by oxidative stress causes global mitochondrial imbalance in ALS. Nat Struct Mol Biol 28(2):132–142
Lin Y et al (2020) Redox-mediated regulation of an evolutionarily conserved cross-beta structure formed by the TDP43 low complexity domain. Proc Natl Acad Sci USA 117(46):28727–28734
Devine MJ, Kittler JT (2018) Mitochondria at the neuronal presynapse in health and disease. Nat Rev Neurosci 19(2):63–80
Wu Y et al (2017) Contacts between the endoplasmic reticulum and other membranes in neurons. Proc Natl Acad Sci USA 114(24):E4859–E4867
Rizzuto R et al (1998) Close contacts with the endoplasmic reticulum as determinants of mitochondrial Ca2+ responses. Science 280(5370):1763–1766
Csordas G et al (2006) Structural and functional features and significance of the physical linkage between ER and mitochondria. J Cell Biol 174(7):915–921
Schon EA, Area-Gomez E (2013) Mitochondria-associated ER membranes in Alzheimer disease. Mol Cell Neurosci 55:26–36
Bernard-Marissal N et al (2015) Dysfunction in endoplasmic reticulum-mitochondria crosstalk underlies SIGMAR1 loss of function mediated motor neuron degeneration. Brain 138(Pt 4):875–890
Prause J et al (2013) Altered localization, abnormal modification and loss of function of Sigma receptor-1 in amyotrophic lateral sclerosis. Hum Mol Genet 22(8):1581–1600
Stoica R et al (2014) ER-mitochondria associations are regulated by the VAPB-PTPIP51 interaction and are disrupted by ALS/FTD-associated TDP-43. Nat Commun 5:3996
Cali T et al (2013) Enhanced parkin levels favor ER-mitochondria crosstalk and guarantee Ca (2+) transfer to sustain cell bioenergetics. Biochim Biophys Acta 1832(4):495–508
Guardia-Laguarta C et al (2014) alpha-Synuclein is localized to mitochondria-associated ER membranes. J Neurosci 34(1):249–259
Ottolini D et al (2013) The Parkinson disease-related protein DJ-1 counteracts mitochondrial impairment induced by the tumour suppressor protein p53 by enhancing endoplasmic reticulum-mitochondria tethering. Hum Mol Genet 22(11):2152–2168
De Vos KJ et al (2012) VAPB interacts with the mitochondrial protein PTPIP51 to regulate calcium homeostasis. Hum Mol Genet 21(6):1299–1311
Rowland AA, Voeltz GK (2012) Endoplasmic reticulum-mitochondria contacts: function of the junction. Nat Rev Mol Cell Biol 13(10):607–625
Paillusson S et al (2016) There’s Something wrong with my MAM; the ER-mitochondria axis and neurodegenerative diseases. Trends Neurosci 39(3):146–157
Gomez-Suaga P et al (2017) The ER-mitochondria tethering complex VAPB-PTPIP51 regulates autophagy. Curr Biol 27(3):371–385
Dafinca R et al (2020) Impairment of mitochondrial calcium buffering links mutations in C9orF72 and TARDBP in iPS-derived motor neurons from patients with ALS/FTD. Stem Cell Rep 14(5):892–908
Wang P et al (2019) TDP-43 induces mitochondrial damage and activates the mitochondrial unfolded protein response. PLoS Genet 15(5):e1007947
Wang W et al (2013) The ALS disease-associated mutant TDP-43 impairs mitochondrial dynamics and function in motor neurons. Hum Mol Genet 22(23):4706–4719
Finelli MJ et al (2015) Oxr1 improves pathogenic cellular features of ALS-associated FUS and TDP-43 mutations. Hum Mol Genet 24(12):3529–3544
Kawamata H et al (2017) Mutant TDP-43 does not impair mitochondrial bioenergetics in vitro and in vivo. Mol Neurodegener 12(1):37
Salvatori I et al (2018) Differential toxicity of TAR DNA-binding protein 43 isoforms depends on their submitochondrial localization in neuronal cells. J Neurochem 146(5):585–597
Yu CH et al (2020) TDP-43 Triggers Mitochondrial DNA Release via mPTP to activate cGAS/STING in ALS. Cell 183(3):636–649
Taslimi A et al (2014) An optimized optogenetic clustering tool for probing protein interaction and function. Nat Commun 5:4925
Barmada SJ et al (2014) Autophagy induction enhances TDP43 turnover and survival in neuronal ALS models. Nat Chem Biol 10(8):677–685
Ling SC et al (2010) ALS-associated mutations in TDP-43 increase its stability and promote TDP-43 complexes with FUS/TLS. Proc Natl Acad Sci USA 107(30):13318–13323
Watanabe S, Kaneko K, Yamanaka K (2013) Accelerated disease onset with stabilized familial amyotrophic lateral sclerosis (ALS)-linked mutant TDP-43 proteins. J Biol Chem 288(5):3641–3654
Swarup V et al (2011) Deregulation of TDP-43 in amyotrophic lateral sclerosis triggers nuclear factor kappaB-mediated pathogenic pathways. J Exp Med 208(12):2429–2447
Xu D et al (2018) TBK1 suppresses RIPK1-driven apoptosis and inflammation during development and in aging. Cell 174(6):1477-1491e19
Zhang J et al (2020) Neurotoxic microglia promote TDP-43 proteinopathy in progranulin deficiency. Nature 588(7838):459–465
Acknowledgements
This work was supported by SERIKA FUND (KA), KAKENHI Grant numbers JP19K06933 (KA) and JP20H05345 (KA).
Author information
Authors and Affiliations
Contributions
KA wrote the manuscript and generated figures, with inputs from HH and KK. All authors approved the submitted version.
Corresponding author
Ethics declarations
Conflict of interest
All authors declare that there is no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Asakawa, K., Handa, H. & Kawakami, K. Multi-phaseted problems of TDP-43 in selective neuronal vulnerability in ALS. Cell. Mol. Life Sci. 78, 4453–4465 (2021). https://doi.org/10.1007/s00018-021-03792-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00018-021-03792-z