Abstract
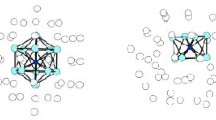
Stabilization of cyclic mercury clusters is discussed using the MNDO method. The bonding character of the highest occupied molecular orbital is shown to be one of the essential criteria of the relative stability of these systems. This criterion max be satisfied by structures with both direct and indirect (via the bridging chlorine atoms) bonding of mercury atoms.
Similar content being viewed by others
References
L. A. Akamova. V. V. Pinyaskin. O. B. Tomilin. et al.,Zh. Strukt. Khim.,36, No. 4, 623–629 (1995).
L. A. Akamova, O. B. Tomilin, I. I. Terekhin, et al.,Zh. Strukt. Khim..39, No. 1, 26–34 (1998).
M. J. Dewar and W. Thiel,J. Am. Chem. Soc.,99, 4899 (1977).
M. J. Dewar, G. L. Grady, K. M. Merz, and J. F. Stewart,Organometallics, No. 4, 1964-1966 (1985).
T. Clark,A Hundbook of Computational Chemistry: a Practical Guide to Chemical Structure and Energy Calculations, Wiley, New York (1985).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Tomilin, O.B., Terekhin, I.I. & Akamova, L.V. On the relative stability of cyclic mercury clusters. J Struct Chem 40, 843–848 (1999). https://doi.org/10.1007/BF02700689
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF02700689