Abstract
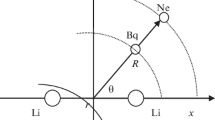
The geometrical structure and the vibrational spectrum of the LiClO3 molecule are studied by the Hartree-Fock-Roothaan (HF) and configurational interaction (CI) methods taking into account single and double excitations and Davidson's correction for quadruple excitations. Double-zeta basis sets of Huzinaga-Dunning and McLean-Chandler complemented with polarization and diffuse functions are used. Potential surface sections corresponding to migration of the Li+ cation around the ClO −3 fragment are investigated. It was found that the LiClO3 molecule has a single stable configuration of Cs symmetry with the bidentate coordination of the Li+ cation by the ClO −3 anion. The cyclic fragment

is nonplanar (the dihedral angle θ(LiO2Cl)=173°). The tridentate configuration of C3v symmetry lies higher than the equilibrium configuration by 24.5 (HF) or 18.3 (CI) kJ/mole and is not an isomer. The ab initio force field of the molecule was refined by the scaling method. Some assignments of the IR bands of the matrix-isolated molecular forms existing in vapor over lithium chlorate are corrected. The vibration frequencies (cm−1) and IR intensities (km/mole; in parentheses) are calculated with the refined force field: A′ type 1099(236), 856(81), 630(73), 557(119), 481(87), 156(66); A″ type 887(229), 459(35), 367(23).
Similar content being viewed by others
References
N. Smyrl and J. P. Devlin,J. Chem. Phys.,60, No. 6, 2540–2546 (1974).
I. R. Beattie and J. E. Parkinson,J. Chem. Soc., Dalton Trans., No. 6, 1185–1188 (1983).
L. Bencivenni, K. A. Gingerich and R. Teghil,Inorg. Chim. Acta,85, No. 1, 11–14 (1984).
L. Bencivenni, H. M. Nagarathna, K. A. Gingerich, and R. Teghil,J. Chem. Phys.,81, No. 8, 3415–3423 (1984).
G. Ritzhaupt, H. H. Richardson, and J. P. Devlin,High Temp. Sci.,19, 163–172 (1985).
G. Ritzhaupt and J. P. Devlin,J. Phys. Chem.,90, No. 26, 6764–6766 (1986).
J. S. Francisco and I. H. Williams,Chem. Phys.,114, No. 3, 339–345 (1987).
F. Ramondo, L. Bencivenni, and F. Grandinetti,Chem. Phys. Lett.,173, No. 5/6, 562–568 (1990).
M. W. Schmidt, K. K. Baldridge, J. A. Boatz, et al.,J. Comput. Chem.,14, 1347–1363 (1993).
S. Husinaga,J. Chem. Phys.,42, No. 4, 1293–1302 (1965).
T. H. Dunning and P. J. Hay,Methods of Electronic Structure Theory, Vol. 3, H. F. Schaefer (ed.), Plenum, New York (1977).
T. H. Dunning,J. Chem. Phys.,53, No. 7, 2823–2833 (1970).
A. D. McLean and G. S. Chandler, —Ibid.,,72, No. 10, 5639–5648 (1980).
H. Johansen,Chem. Phys. Lett.,11, No. 4, 466–471 (1971).
V. G. Solomonik, V. M. Ozerova, and V. V. Sliznev,Zh. Neorg. Khim.,27, No. 7, 1636–1642 (1982).
D. J. Gardiner, R. B. Girling, and R. E. Hester,J. Mol. Struct.,13, No. 1, 105–114 (1972).
G. Fogarasi and P. Pulay, in:Vibrational Spectra and Structure, Vol. 14, Amsterdam (1985), pp. 125–219.
N. Smyrl and J. P. Devlin,J. Phys. Chem.,77, No. 26, 3067–3070 (1973).
Additional information
Ivanovo State Chemical Technological Academy. Translated fromZhurnal Strukturnoi Khimii, Vol. 37, No. 3, pp. 440–449, May–June, 1996.
Rights and permissions
About this article
Cite this article
Solomonik, V.G., Pogrebnaya, T.P. & Sliznev, V.V. Ab initio study of the structure, force field, and vibrational spectrum of the LiClO3 molecule. J Struct Chem 37, 384–391 (1996). https://doi.org/10.1007/BF02578590
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF02578590