Summary
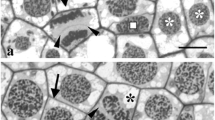
In mesophyll cells of the aquatic angiospermVallisneria gigantea Graebner, the endoplasm streams rotationally along the cell walls normal to the leaf surface in situ. Bundles of microfilaments anchored in the ectoplasm serve as tracks for the cytoplasmic streaming. In single mesophyll cells isolated by enzymatic digestion, hypertonic treatment induces abnormal streaming concomitant with plasmolysis, specifically at one or both of the shorter sides of an approximate rectangle. The disorderly arrangement of microfilaments in such cells has been confirmed by fluorescence microscopy of cells stained with FITC-phalloidin. While inhibitors of proteases added to the enzyme solution used for isolation of cells suppress the disturbance of rotational streaming, exogenously applied protease promotes it. The results suggest that bundles of microfilaments in the ectoplasm are stabilized by protease-sensitive factor(s) in the presence of the cell wall.
Similar content being viewed by others
Abbreviations
- APMSF:
-
(p-amidinophenyl)methanesulfonyl fluoride hydrochloride
- ATP:
-
adenosine 5′-triphosphate
- BS:
-
basal solution
- EGTA:
-
ethyleneglycol-bis(2-aminoethylether)-N,N,N′,N′-tetraacetic acid
- F-actin:
-
fibrous actin
- FITC:
-
fluorescein isothiocyanate
- FTC:
-
fluorescein thiocarbamoyl derivative
- G-actin:
-
globular actin
- MS:
-
Murashige and Skoog
- Pipes:
-
piperazine-N,N′-bis (2-ethanesulfonic acid)
- Tris:
-
tris(hydroxymethyl)aminomethane
References
Akashi T, Shibaoka H (1991) Involvement of transmembrane proteins in the association of cortical microtubules with the plasma membrane in tobacco BY-2 cells. J Cell Sci 98 (in press)
—, Kawasaki S, Shibaoka H (1990) Stabilization of cortical microtubules by the cell wall in cultured tobacco cells: effects of extensin on the cold-stability of cortical microtubules. Planta 182: 363–369
Brawley SH, Robinson KR (1985) Cytochalasin treatment disrupts the endogenous currents associated with cell polarization in fucoid zygotes: studies of the role of F-actin in embryogenesis. J Cell Biol 100: 1173–1184
Burridge K, Fath K, Kelly T, Nuckolls G, Turner C (1988) Focal adhesions: transmembrane junctions between the extracellular matrix and the cytoskeleton. Annu Rev Cell Biol 4: 487–525
Forde J, Steer MW (1976) Cytoplasmic streaming inElodea. Can J Bot 54: 2688–2694
Grolig F, Williamson RE, Parke J, Miller C, Anderton BH (1988) Myosin and Ca2+-sensitive streaming in the algaChara: detection of two polypeptides reacting with a monoclonal antimyosin and their localization in the streaming endoplasm. Eur J Cell Biol 47: 22–31
Hayashi T, Kamitsubo E (1959) Plasmolysis in Characeae. Bot Mag 72: 309–315
Ishigami M, Nagai R (1980) Motile apparatus inVallisneria leaf cells. II. Effects of cytochalasin B and lead acetate on the rate and direction of streaming. Cell Struct Funct 5: 13–20
Izutani Y, Takagi S, Nagai R (1990) Orientation movements of chloroplasts inVallisneria epidermal cells: different effects of light at low- and high-fluence rate. Photochem Photobiol 51: 105–111
Kakimoto T, Shibaoka H (1987) Actin filaments and microtubules in the preprophase band and phragmoplast of tobacco cells. Protoplasma 140: 151–156
Kamitsubo E (1966) Motile protoplasmic fibrils ofCharaceae. II. Linear fibrillar structure and its bearing on protoplasmic streaming Proc Jap Acad 42: 640–643
Kamiya N (1959) Protoplasmic streaming. Springer, Wien [Heilbrunn LV, Weber F (eds) Protoplasmatologia, vol VIII 3 a]
— Kuroda K (1956) Velocity distribution of protoplasmic streaming inNitella cells. Bot Mag 69: 543–554
Kato T, Tonomura Y (1977) Identification of myosin inNitella flexilis. J Biochem 82: 777–782
Kropf DL, Kloareg B, Quatrano RS (1988) Cell wall is required for fixation of the embryonic axis inFucus zygotes. Science 239: 187–190
Kuroda K (1983) Cytoplasmic streaming in Characean cells cut open by microsurgery. Proc Jp Acad (B) 59: 126–130
Küster E (1910) Über Inhaltsverlagerungen in plasmolysierten Zellen. Flora 100: 267–287
Lichtscheidl IK, Lancelle SA, Hepler PK (1990) Actin-endoplasmic reticulum complexes inDrosera. Their structural relationship with the plasmalemma, nucleus, and organelles in cells prepared by high pressure freezing. Protoplasma 155: 116–126
Murashige T, Skoog F (1962) A revised medium for rapid growth and bioassays with tobacco tissue cultures. Physiol Plant 15: 473–497
Nagai R, Hayama T (1979) Ultrastructure of the endoplasmic factor responsible for cytoplasmic streaming inChara internodal cells. J Cell Sci 36: 121–136
—, Rebhum LI (1966) Cytoplasmic microfilaments in streamingNitella cells. J Ultrastruct Res 14: 571–589
Ohsuka K, Inoue A (1979) Identification of myosin in a flowering plant,Egeria densa. J Biochem 85: 375–378
Palevitz BA, Ash JF, Hepler PK (1974) Actin in the green alga,Nitella. Proc Natl Acad Sci USA 71: 363–366
Schindler M, Meiners S, Cheresh DA (1989) RGD-dependent linkage between plant cell wall and plasma membrane: consequences for growth. J Cell Biol 108: 1955–1965
Sheetz MP, Spudich JA (1983) Movement of myosin-coated fluorescent beads on actin cables in vitro. Nature 303: 31–35
Shimmen T, Yano Y (1984) Active sliding movement of latex beads coated with skeletal muscle myosin onChara actin bundles. Protoplasma 121: 132–137
Shinmi T, Hase T, Sugiyama T (1989) Effect of ATP on the trypsin proteolysis of plasma membrane ATPase from corn roots. In: Tazawa M, Katsumi M, Masuda Y, Okamoto H (eds). Plant water relations and growth under stress. MYU K.K., Tokyo, pp 446–448
Sonobe S, Shibaoka H (1989) Cortical fine actin filaments in higher plant cells visualized by rhodamine-phalloidin after pretreatment with m-maleimidobenzoyl N-hydroxysuccinimide ester. Protoplasma 148:80–86
Takagi S, Nagai R (1983) Regulation of cytoplasmic streaming inVallisneria mesophyll cells. J Cell Sci 62: 385–405
Traas JA, Doonan JH, Rawlins DJ, Shaw PJ, Watts J, Lloyd CW (1987) An actin network is present in the cytoplasm throughout the cell cycle of carrot cells and associates with the dividing nucleus. J Cell Biol 105: 387–395
Twining SS (1984) Fluorescein isothiocyanate-labeled casein assay for proteolytic enzymes. Anal Biochem 143: 30–34
Williamson RE (1974) Actin in the alga,Chara corallina. Nature 248: 801–802
Yamaguchi Y, Nagai R (1981) Motile apparatus inVallisneria leaf cells. I. Organization of microfilaments. J Cell Sci 48: 193–205
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Masuda, Y., Takagi, S. & Nagai, R. Protease-sensitive anchoring of microfilament bundles provides tracks for cytoplasmic streaming inVallisneria . Protoplasma 162, 151–159 (1991). https://doi.org/10.1007/BF02562558
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02562558