Abstract
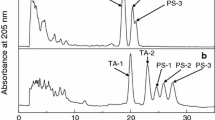
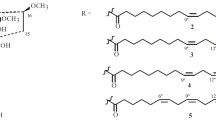
A reexamination of the flaxseed hydroperoxide isomerase reaction showed that a minor enzymic product (ca. 5%), identified as a γ-ketol, was present. The substrates were the 13- or 9-hydroperoxides of linolenic acid, which were converted to 9-hydroxy-12-oxo-cis-15-trans-11-octadecadienoic acid, respectively. These compounds were formed in addition to the major products reported earlier: a 12,13-α-ketol and 12-oxo-cis-10,15-phytodienoic acid from the 13-isomer, and a 9,10-α-ketol from the 9-isomer.
Similar content being viewed by others
References
Zimmerman, D.C., Biochem. Biophys. Res. Commun. 23:398 (1966).
Gardner, H.W., R. Kleiman, D.D. Christianson and D. Weisleder, Lipids 10:602 (1975).
Feng, P., and D.C. Zimmerman, Lipids 14:710 (1979).
Zimmerman, D.C., and P. Feng, Lipids 13:313 (1978).
Surrey, K., Plant Physiol. 39:65 (1964).
Matthew, J.A., H.W.-S. Chan and T. Galliard, Lipids 12:324 (1977).
Frankel, E.N., W.E. Neff, W.K. Rohwedder, B.P.S. Khambay, R.F. Garwood and B.C.L. Weedon, Lipids 12:901 (1977).
Silverstein, R.M., G.C. Bassler and T.C. Morrill, in “Spectrometric Identification of Organic Compounds,” 3rd Edition, John Wiley & Sons, Inc., New York, 1974, pp. 231–238.
Nakanishi, K., in “Infrared Absorption Spectroscopy,” Holden-Day, Inc., San Francisco, 1962, pp. 42–44.
Graveland, A., L. Pesman and P. van Eerde, Master Brewers Assoc. Am. Tech. Quart. 9:98 (1972).
Schauenstein, E., H. Esterbauer and H. Zollner, in “Aldehydes in Biological Systems,” Pion Ltd., London, 1977, pp. 42–102.
Author information
Authors and Affiliations
About this article
Cite this article
Feng, P., Vick, B.A. & Zimmerman, D.C. Formation of γ-ketols from 13- and 9-hydroperoxides of linolenic acid by flaxseed hydroperoxide isomerase. Lipids 16, 377–379 (1981). https://doi.org/10.1007/BF02534967
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF02534967