Abstract
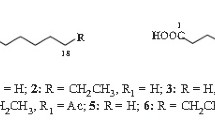
One of the products formed upon incubation of linolenic acid (cis9,12,15-octadecatrienoic acid) with an extract of flaxseed acetone powder has been characterized as 8-[2-(cis-pent-2′-enyl)-3-oxo-cis-cyclopent-4-enyl]octanoic acid. The cyclopentenone ring structure of this acid is analogous to that of the A-type prostaglandins produced in mammalian systems.
Similar content being viewed by others
References
Axelrod, B., Adv. Chem. Ser. 136:324 (1974).
Zimmerman, D.C., and B.A. Vick, Plant Physiol. 46:445 (1970).
Gardner, H.W., J. Lipid Res. 11:311 (1970).
Vick, B.A., and D.C. Zimmerman, Plant Physiol. 57:780 (1976).
Galliard, T., and D.R. Phillips, Biochem. J. 129:743 (1972).
Surrey, K., Plant Physiol. 39:65 (1964).
Nakaniski, K., in “Infrared Absorption Spectroscopy,” Holden-Day, Inc., San Francisco, CA, 1962, pp. 42–44.
Bergstrom, S., L.A. Carlson, and J.R. Weeks, Pharmacol. Rev. 20:1 (1968).
Ramwell, R.W., J.E. Shaw, G.B. Clarke, M.F. Grostic, D.G. Kaiser, and J.E. Pike, in “Progress in the Chemistry of Fats and Other Lipids,” Vol. 9, Edited by R.T. Holman, Pergamon Press, Oxford and New York, 1968, pp. 233–273.
Casida, J.E., in “Pyrethrum,” Academic Press, New York, 1973, pp. 108–109.
Fukui, H., K. Koshimizu, Y. Yamazaki, and S. Usuda, Agric. Biol. Chem. 41:189 (1977).
Author information
Authors and Affiliations
About this article
Cite this article
Zimmerman, D.C., Feng, P. Characterization of a prostaglandin-like metabolite of linolenic acid produced by a flaxseed extract. Lipids 13, 313–316 (1978). https://doi.org/10.1007/BF02533720
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF02533720