Abstract
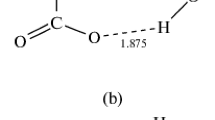
The interaction of O+ ion with several (from one to four) water molecules was studied by theab initio (UMP4/4-31G*) and semiempirical (AM1) quantum-chemical methods. It was found that the energy of binding the O+ ion to the first water molecule is appreciably higher than those of binding to the subsequent water molecules. In the complex with a water molecule, whose structure corresponds to that of water oxide, the O+ ion retains high reactivity. The barrier to the transfer of O+ ion to another water molecule is much lower than the barrier to analogous transfer of O atom from the molecule of water oxide, despite the lower dissociation energy of the H2O−O bond. Consideration of subsequent interactions with water molecules leads to an increase in the barrier to the transfer of O+ ion. The doublet and quadruplet excited states of the O++2 H2O system were also studied. In these cases, the formation energies are well described by the ion-dipole model.
Similar content being viewed by others
References
F. D. Furia and G. Modena,Pure Appl. Chem., 1982,54, 1853; A. E. Shilov, inActivation and Functionalization of Alkanes, Ed. C. L. Hill, Wiley, New York, 1989, 11.
Yu. V. Geletii, V. V. Lavrushko, and A. E. Shilov,Dokl. Akad. Nauk SSSR, 1986,288, 139 [Dokl. Chem., 1986 (Engl. Transl.)].
D. H. R. Barton, M. J. Gastiger, and W. B. Motherwell,J. Chem. Soc., Chem. Commun., 1983, 41; D. H. R. Barton, F. Halley, N. Ozbalik, M. Schmitt, E. Young, and G. Balavoine,J. Am. Chem. Soc., 1989,111, 7144.
Yu. V. Geletti, V. V. Lavrushko, and G. V. Lubimova,J. Chem. Soc., Chem. Commun., 1988, 936; Yu. V. Geletii, V. E. Zubarev, P. P. Levin, G. V. Lyubimova, and V. Ya. Shafirovich,Kinet. Katal., 1990,31, 802 [Kinet. Catal., 1990,31 (Engl. Transl.)]; D. H. R. Barton, D. Doller, and Yu. V. Geletii,Mendeleev Commun., 1991, 115.
A. E. Shilov,React. Kinet. Catal. Lett., 1990,41, 223; A. E. Shilov,Khim. Fiz., 1991,10, 758 [Sov Chem. Phys., 1991,10 (Engl. Transl.)].
E. M. Koldasheva, Yu. V. Geletii, V. V. Yanilkin, and V. V. Strelets,Izv. Akad. Nauk SSSR, Ser. Khim., 1990, 994 [Bull. Acad. Sci. USSR, Div. Chem. Sci., 1990,39, 886 (Engl. Transl.)]; E. M. Koldasheva, A. F. Shestakov, Yu. V. Geletii, and A. E. Shilov,Izv. Akad. Nauk SSSR, Ser. Khim., 1992, 845 [Bull. Acad. Sci. USSR, Div. Chem. Sci., 1992,41, 655 (Engl. Transl.)].
E. M. Koldasheva, Yu. V. Geletii, A. F. Shestakov, A. V. Kulikov, and A. E. Shilov,New. J. Chem., 1993,17, 421; E. M. Koldasheva, Yu. V. Geletii, A. F. Shestakov, A. V. Kulikov, and A. E. Shilov,Mendeleev Commun., 1993, 142.
G. V. Lyubimova and A. F. Shestakov,Kinet. Katal., 1994,35, 232 [Kinet. Catal., 1994,35 (Engl. Transl.)].
M. J. Frisch, G. W. Trucks, M. Head-Gordon, P. M. W. Gill, M. W. Wong, J. B. Foresman, B. G. Johnson, H. B. Schlegel, M. A. Robb, E. S. Replogle, R. Gomperts, J. L. Andres, K. Raghavachari, J. S. Binkley, C. Gonzalez, R. L. Martin, D. J. Fox, D. J. Defrees, J. Baker, J. J. P. Stewart, and J. A. Pople,Gaussian 92, Revision 1, Gaussian, Inc., Pittsburgh, PA, 1992.
M. J. S. Dewar, E. G. Zoebisch, E. F. Healy, and J. J. P. Stewart,J. Am. Chem. Soc., 1985,107, 3902.
A. A. Radtsig and B. M. Smirnov,Spravochnik po atomnoi i molekulyarnoi fizike [Handbook on Atomic and Molecular Physics], Atomizdat, Moscow, 1980, 240 pp. (in Russian).
R. D. Bach, A. L. Owensby, C. Gonzalez, H. B. Schlegel, and J. J. W. McDouall,J. Am. Chem. Soc., 1991,113, 6001.
S. P. de Visser, L. J. de Koning, and N. M. M. Nibbering,J. Phys. Chem., 1995,99, 15444.
Author information
Authors and Affiliations
Additional information
Translated fromIzvestiya Akademii Nauk. Seriya Khimicheskaya, No. 6, pp. 981–988, June, 2000.
Rights and permissions
About this article
Cite this article
Lyubimova, G.V., Shestakov, A.F. Interaction of O+ ion with water molecules: A quantum-chemical study. Russ Chem Bull 49, 976–983 (2000). https://doi.org/10.1007/BF02494879
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF02494879