Abstract
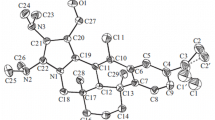
The sesquiterpene lactone argolide has been converted into its 1(10)-epoxide. The structure of the latter has been investigated by NMR, x-ray structural analysis, and molecular mechanics.
Similar content being viewed by others
References
S. M. Adekenov, K. A. Aituganov, K. M. Turdybekov, S. V. Lindeman, and Yu. T. Struchkov, Khim. Prir. Soedin., 653 (1991).
F. H. Allen, O. Kennard, D. G. Watson, L. Brammer, A. G. Orpen, and R. Taylor, J. Chem. Soc., Perkin Trans. II, S1 (1987).
J. L. Breton, F. Camps, J. Coll, L. Eguren, J. A. Gavin, A. G. Gonzalez, X. Martorell, C. Miravitlles, E. Molins, and J. Torramilans, Tetrahedron,41, 3141 (1985).
H. Haasnoot, F. A. A. M. de Leeuw, and C. Altona, Tetrahedron,36, 2783 (1981).
H. Günther, NMR Spectroscopy: An Introduction, Wiley, New York (1980).
W. Z. Wang, R. X. Tan, Y. M. Yao, Q. Wang, and F. X. Jiang, Phytochemistry,37, No. 5, 1347 (1994).
A. T. Kulyyasov, T. S. Seitembetov, K. M. Turdybekov, and S. M. Adekenov, Khim. Prir. Soedin., 879 (1996).
Author information
Authors and Affiliations
Additional information
Translated from Khimiya Prirodnykh Soedinenii, No. 1, pp. 60–64, January–February, 1998.
Rights and permissions
About this article
Cite this article
Turdybekov, K.M., Edil'baeva, T.T., Raldugin, V.A. et al. Molecular and crystal structures of argolide epoxide. Chem Nat Compd 34, 44–47 (1998). https://doi.org/10.1007/BF02249684
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF02249684