Abstract
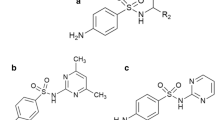
The bioavailability of205Bi from various205Bilabelled pharmaceutical oral bismuth preparations was studied in rats. The intestinal absorption, calculated from205Bi whole body retention and accumulated205Bi urinary excretion, was small in general, but significantly higher (0.26–0.33% of dose) from oral bismuth citrates (basic bismuth citrate, colloidal bismuth subcitrate) as compared to basic bismuth nitrate, salicylate, gallate, and bismuth aluminate (0.04–0.11% of dose). After oral administration, the retained bismuth was mainly accumulated in the kidney, followed by bone, red blood cells and the lung. The whole body retention, faecal and urinary excretions of205Bi were described by a three-compartment model. Biological205Bi half-lives of 10, 36 and 295 h were derived in rats.
Similar content being viewed by others
References
Bevington PA (1969) Data reduction and error analysis for the physical sciences. McGraw-Hill, New York
Bierer DW (1990) Bismuth subsalicylat: history, chemistry, and safety. Rev Infect Dis 12 [suppl. 1]: 3–8
Braunsfurth JS, Gabbe EE, Heinrich HC (1976) Performance of the Hamburg 4Π whole body radioactivity counter. Phys Med Biol 22: 1–17
DAB (Deutsches Arzneibuch) (1968), 7. Ausgabe, Kommentar Böhme H, Hartke K (eds). Wissenschaftliche Verlagsgesellschaft, Stuttgart, pp 1547–1558
Fischer R, Dresow B, Wendel J, Bechthold V, Heinrich HC (1991) Bi-205/Bi-206 cyclotron production from Pb-isotopes for absorption studies in man. Int J Appl Radiat Isot (in press)
Froomes PRA, Wan AT, Keech AC, McNeil JJ, McLean AJ (1989) Absorption and elimination of bismuth from oral doses of tripotassium dicitrato bismuthate. Eur J Clin Pharmacol 37: 533–536
Galland MC, Rodor F, Jouglard J (1979) Bismuth et encephalopathie. Med Hyg 37: 2579–2582
Gaucher A, Netter P, Faure G (1979) Bismuth-induced osteoarthropathies. Med J Aust 66: 129–130
Gmelins Handbuch der anorganischen Chemie (1964) Ergänzungsband 8. Auflage. Verlag Chemie Weinheim, p 840
Hillemand P, Palliere M, Laquais B, Bouvet P (1977) Traitment bismuthique et bismuthemie. Semin l'Hospital de Paris 53: 1663–1669
Lee SP, Lim TH, Pybus J, Clarke AC (1980) Tissue distribution of orally administered bismuth in the rat. Clin Exp Pharmacol Physiol 7: 319–324
Marshall BJ, McGechie DB, Rogers PA, Glancy RJ (1985) Pyloric campylobacter infection and gastroduodenal disease. Med J Aust 142: 439–444
Rao N, Feldman St (1990) Disposition of bismuth in the rat. I. Red blood cell and plasma protein binding. Pharm Res 7: 188–191
Raedsch R, Walter-Sack I, Weber E, Blessing J (1990) Pharmakokinetik von Wismut — Präparaten bei Patienten mit Gastritis und Ulkuskrankheiten. Klin Wochenschr 68: 488
Russ GA, Bigler RE, Tilbury RS, Woodard H, Laughlin J (1975) Metabolic studies with radiobismuth. Rad Res 63: 443–454
Vienet R, Bouvet P, Istin M (1983) Cinetique et distribution du206Bi chez le rat et le lapin: un modele. Int J Appl Radiat Isot 34: 747–753
Zidenberg-Cherr S, Parks NJ, Keen CL (1987) Tissue and subcellular distribution of bismuth radiotracer in the rat: considerations of cytotoxicity and microdosimetry for bismuth radiopharmaceuticals. Radiat Res 111: 119–129
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Dresow, B., Nielsen, P., Fischer, R. et al. Bioavailability of bismuth from205Bi-labelled pharmaceutical oral Bi-preparations in rats. Arch Toxicol 65, 646–650 (1991). https://doi.org/10.1007/BF02098030
Issue Date:
DOI: https://doi.org/10.1007/BF02098030