Abstract
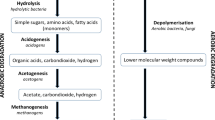
Bacterial counts on the contents of an anaerobic fixed-bed digester receiving a whey substrate were conducted using the modified roll tube technique. Average anaerobic counts after 48 hours incubation on lactate containing media were 3.12 × 109 and 3.7 × 109 ml−1, respectively. These counts were between 140 and 190 times higher than aerobic counts on the same media. Seventy-four strains from both media were isolated and characterized, and the relationship between the organisms was calculated according to the similarity coefficient of Sokal and Michener [20]. The organisms were clustered using the unweighted pair group method and the results were presented in the form of a simplified dendrogram. The isolates clustered in three major groups (A, B, and C) at a similarity level of 76%. A small diffuse group of five organisms was also found. The organisms in two (A and B) of the major clusters were obligate anaerobes. Cluster A represented 34% of the isolates, cluster B represented 50%, and the facultative streptococci in cluster C, 11%. The isolates in clusters A and B could only tentatively be identified as members of the generaBacteroides andPeptostreptococcus, respectively. The isolates could not be positively identified.
Similar content being viewed by others
References
Bryant MP (1976) The microbiology of anaerobic degradation and methanogenesis with special reference to sewage. In: Schlegel HG, Barnea J (eds) Microbial energy conversion. Pergamon Press, Oxford, pp 107–117
Bryant MP, Robinson IM (1961) An improved non-selective culture medium for ruminal bacteria and its use in determining diurnal variation in numbers of bacteria in the rumen. J Dairy Sci 44:1446–1456
Chartrain M, Zeikus JG (1986) Microbial ecophysiology of whey biomethanation: characterization of bacterial trophic populations and prevalent species in continuous culture. Appl Environ Microbiol 51:188–196
Chen M, Wolin MJ (1977) Influence of CH4 production byMethanobacterium ruminantium on the fermentation of glucose and lactate bySelenomonas ruminantium. Appl Environ Microbiol 341:756–759
Collins MD, Shah HN (1986) Reclassification ofBacteroides termitidis Sebald (Holdeman and Moore) in a new genusSebaldella, asSebaldella termitidis comb. nov. Intern J Sys Bacteriol 36:349–350
De Haast J, Britz TJ (1986) Characterization of aerobic and facultative anaerobic bacteria from the liquid phase of an anaerobic fixed-bed digester treating a cheese whey substrate. Microb Ecol 12:173–179
De Haast J, Britz TJ, Novello JC, Verwey EW (1985) Anaerobic digestion of deproteinated cheese whey. J Dairy Sci 52:457–467
Harrigan WF, McCance ME (1976) Laboratory methods in food and dairy microbiology. Academic Press, London
Hobson PN (1973) The bacteriology of anaerobic sewage digestion. Process Biochem 8: 19–25
Holdeman LV, Cato EP, Moore WEC (1977) Anaerobic laboratory manual, 4th ed. Virginia Polytechnic Institute and State University, Blacksburg, Virginia
Hungate RE (1969) A roll tube method for cultivation of strict anaerobes. In: Norris JR, Ribbons DW (eds) Methods in microbiology. Vol 3B. Academic Press, London, pp. 117–132
Iannotti EL, Fischer JR, Sievers DM (1978) Medium for the enumeration and isolation of bacteria from a swine waste digester. Appl Environ Microbiol 36:555–566
Iannotti EL, Fischer JR, Sievers DM (1982) Medium for enhanced growth of bacteria from a swine manure digester. Appl Environ Microbiol 43:247–249
Kistner A (1960) An improved method for viable counts of bacteria of the ovine rumen which ferment carbohydrates. J Gen Microbiol 23:565–576
Krieg NR, Holt JG (1984) Bergey's manual of systematic bacteriology. Vol 1. Williams & Wilkins, Baltimore, pp 602–662
Mayfield CI, Inniss WE (1977) A rapid, simple method for staining bacterial flagella. Can J Microbiol 23:1311–1313
Miller TL, Wolin MJ (1974) A serum bottle modification of the Hungate technique for cultivating obligate anaerobes. Appl Microbiol 27:985–987
Sahm H (1984) Anaerobic wastewater treatment. Adv in Biochem Eng/Biotechnol 29:83–115
Siebert ML, Toerien DF (1969) The proteolytic bacteria present in the anaerobic digestion of raw sewage sludge. Water Res 3:241–250
Sokal RR, Michener CD (1958) A statistical method for evaluating systematic relationships. Kansas University Science Bulletin 38:1409–1438
Toerien DF (1967) Enrichment culture studies on aerobic and facultative anaerobic bacteria found in anaerobic digesters. Water Res 1:147–155
Toerien DF (1970) Population description of the non-methanogenic phase of anaerobic digestion. II. Hierarchical classification of isolates. Water Res 4:285–303
Toerien DF, Hattingh WHJ (1969) Anaerobic digestion. I. The microbiology of anaerobic digestion. Water Res 3:385–416
Toerien DF, Siebert ML (1967) A method for the enumeration and cultivation of anaerobic “acid-forming” bacteria present in digesting sludge. Water Res 1:397–404
Toerien DF, Siebert ML, Hattingh WHJ (1967) The bacterial nature of the acid-forming phase of anaerobic digestion. Water Res 1:497–507
Turton LJ, Drucker DB, Hillier VF, Ganguli LA (1983) Effect of eight growth media upon fermentation profiles of ten anaerobic bacteria. J Appl Bacteriol 54:295–304
Ueki A, Minato H, Azuma R, Suto T (1980) Enumeration and isolation of anaerobic bacteria in sewage digestor fluids: isolation of lactate utilizers. J Gen Microbiol 26:15–24
Ueki A, Miyagawa E, Minato H, Azuma R, Suto T (1978) Enumeration and isolation of anaerobic bacteria in sewage digestor fluids. J Gen Microbiol 24:317–332
Varel VH (1984) Characteristics of some fermentative bacteria from a thermophilic methaneproducing fermenter. Microb Ecol 10:15–24
Zeikus JG (1980) Microbial populations in digesters. In: Stafford DA, Wheatly BI, Hughes DE (eds) Anaerobic digestion. Applied Science Publishers, London, pp 61–89
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
De Haast, J., Britz, T.J. Characterization of some anaerobic bacteria from the liquid phase of a mesophilic anaerobic digester fed with a prefermented cheese whey substrate. Microb Ecol 14, 167–177 (1987). https://doi.org/10.1007/BF02013021
Issue Date:
DOI: https://doi.org/10.1007/BF02013021