Summary
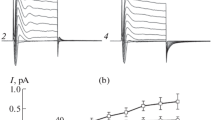
Outward rectifying. cation channels were observed in the epithelial cells of the urinary bladder of the toad.Bufo marinus. As studied in isolated cells using the patch-clamp technique, the channel has an average conductance of 24 and 157 pS for pipette potentials between 0 and +60 mV and −60 to −100 mV, respectively, when the major cation in both bath and pipette solutions is K+. The conductance of the cannel decreasen with increasing dehydration energy of the permeant monovalent cation in the oder Rb+=K+>Na+>Li+. Reversal potentials near zero under biionic conditions imply that the permeabilities for all four of these cations are smiliar. The channel is sensitive to quinidine sulfate but not to amiloride. It shares several pharmacological and biophysical properties with an outwardly-rectifying, vasopressin-sensitive pical K+ conductive pathway described previously for the toad urinary bladder. We demonstrate, in both single-channel and whole-bladder studies, that the outward rectification is a consequence of interaction of the chanel with extracellular divalent cations, particularly Ca2+, which blocks inward but not outward current. Various divalent cations impart different degrees of outward rectification to the conductive pathway. Concentrations of Mg2+ and Ca2+ required for halfmaximal effect are 3×10−4 and 10−4 m, resopectively. For Co2+ the values are 10−6 m at +50 mV and a 10−4 m at +200 mV. The mechanism of blockade by divalent cations is not established, but does not seem to involve a voltage-dependent interaction in which the blocker penetrates the transmembrane electric field. In the absence of divalent cations in the mucosal solution, the magnitudes of inward current carried by Rb+, K+, Na+ and Li+ through the apical K+ pathway at any transepithelial voltage, are in the same order as in the single-channel studies. We propose that the cation channel observed by us in isolated epithelial cells is the single-channel correlate of the vasopressin-sensitive apical K+ conductive pathway in the toad urinary bladder and is also related to the oxytocin- and divalent cation-sensitive apical condictivity observed in frog skin and urinary bladder.
Similar content being viewed by others
References
Aelvoet, I., Erlij, D., Van Driessche, W. 1988. Activation and blockage of a calcium-sensitive cation-selective pathway in the apical membrane of toad urinaty bladder.J. Physiol. (London) 398:555–574
Armstrong, C. M. 1971. Interaction of tetracthylammonium ion derivatives with the potassium channels of giant axion.J. Gen. Physiol. 58:413–437
Eisemann, G. 1962. Cation-selective glass electrodes and their mode of operation.Biophys. J. 2(Suppl. 2):259–323
Fabiato, A., Fabiato, F. 1979. Calculator programs for computing the composition of the solutions contraining multiple metals and ligand used for experiments in skinned muscle cells.J. Physiol. (Paris) 75:463–505
Garty, H., Benos, D.J. 1988. Characteristics and regulatory mechanisms of the amiloride-blockable Na+ channel.Physiol. Rev. 68:309–373
Hille, B. 1984. Ionic Channels of Excitable Membranes. Sinauer, Sunderland, MA
Jaslove, S. W., Brink P. R. 1986. The mechanism of recification at the electrotonic motor giant synapse of the crayfish.Nature (London) 323:63–65
Latorre, R., Miller, C. 1983. Conduction and selectivity in potasium channels.J. Membrane Biol. 71:11–30
Matsuda, H., Saigusa, A, Irisawa, H. 1987.Ohmic conductance through the inwardly rectifying K channel and blocking by internal Mg2+.Nature (London) 325:156–159
Mayer, M.L., Wesbrook, G.L., Guthrie, P.B. 1984. Voltagedependent block by Mg2+ of NMDA responses in spinal cord neurones.Nature (London) 309:261–263
Nowak, L., Bregestovsky, P., Ascher, P., Herbet, A., Prochiantz, A. 1984. Magnesium gates glutamate-activated channels in mouse central neurones.Nature (London) 307:462–465
Palmer, L. G. 1985. Interactions of amiloride and other blocking cations with the apical Na channel in the toad urinary bladder.J. Membrane Biol. 87:191–199
Palmer, L. G. 1986. Apical membrane K conductance in the toad urinary bladder.J. Membrane Biol. 92:217–226
Robinson, R. A., Stokes, R. H. 1965. Electrolyte Solution. Butterworths, London
Rodriguez, H. J., Scholer, D. W., Purkerson, M. L., Klahr, S. 1980. Isolation of epithelial cells from toad blaudder.Am. J. Physiol. 238:F140-F149
Van Driessche, W. 1987. Ca2+ channels in the apical membrane of the toad urinary bladder.Pluegers Arch. 410:239–249
Van Driesche, W., Aelvoel, I., Erdij, D. 1987. Oxytocin and cAMP stimulate monovalent cation movemetn through a Ca2+-sensitive, amiloride-insensitive channel in the apical membrane of thetoad urinary bladder.Proc. Natl. Acad. Sci USA 84:313–317
Van Driessche, W., Zeiske, W. 1985. Ca2+-sensitive spontaneously fluctuating, cation channels in the apical membrane of the adult frog skin epithelim.Pfluegers Arch. 405:250–259
Woodhull, A. M. 1973. Ionic blockage of sodium channels in nerve.J. Gen. Physiol. 61:687–708
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Das, S., Palmer, L.G. Extracellular Ca2+ controls outward rectification by apical cation channels in toad urinary bladder: Patch-clamp and whole-bladder studies. J. Membrain Biol. 107, 157–168 (1989). https://doi.org/10.1007/BF01871721
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF01871721