Abstract
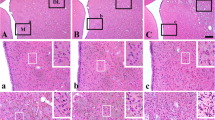
Morphological changes in the CA1 region of the hippocampus in the rat cardiac arrest model were studied with the in situ nick-end labeling (TUNEL) method and light and electron microscopy. The TUNEL-positive pyramidal cells first appeared on day 1, increased in number with time, and reached a peak at 7 days after recirculation. At the ultrastructural level, cell shrinkage, nuclear fragmentation, and an increased number of atuophagic vacuoles of the pyramidal cells were observed in the CA1 region. The brief ischemia activates the microglial cells in the CA1 region, and these cells were found to increase in number with time. The microglial cells were seen to adhere to degenerating pyramidal cells and to phagocytose the apoptotic neurons selectively.
Similar content being viewed by others
References
Kirino T (1982) Delaved neuronal death in the gerbil hippocampus following ischemia. Brain Res 239:57–69
Wyllie AH, Kerr JER, Currie AR (1980) Cell death: the significance of apoptosis. Int Rev Cytol 68:251–306
Pulsinelli WA, Brierley JB (1979) A new model of bilateral hemispheric ischemia in the unanesthetized rat. Stroke 10:267–272
Kirino T, Tamura A, Sano K (1984) Delayed neuronal death in the rat hippocampus following transient forebrain ischemia. Acta Neuropathol (Berl) 64:139–147
MacManus JP, Buchan AM, Hill IE, Rasquinha I, Preston E (1993) Global ischemia can cause DNA fragmentation indicative of apoptosis in rat brain. Neurosci Lett 164:89–92
Nitatori T, Sano N, Waguri S, Karasawa Y, Araki H., Shibanai K, Kominami E, Uchiyama Y (1995) Delayed neuronal cell death in the CA1 pyramidal cell layer of the gerbil hippocampus following transient ischemia is apoptosis. J. Neurosci 15:1001–1011
Schmidt-Kastner R, Fliss H, Hakim AM (1997) Subtle neuronal death in striatum after short forebrain ischemia in rats detected by in situ end-labeling for DNA damage. Stroke 28:163–170
Korpatchev WG, Lysenkov SP, Thieliz PS (1982) Modielirowanlie klinczeskoj smerti 1 postreanimationznoj bolezni u krys. Palol Fiziol Exp Ter 3:78–80
Kawai K, Nitecka L, Ruetzler CA, Nagshima G, Joo F, Mies G, Nowak TS, Saito N, Lohr JM, Klatzo I (1992) Global cerebral ischemia associated with cardiac arrest in the rat: I. Dynamics of early neuronal changes. J Cereb Blood Flow Metab 12:238–249
Brierley JB, Brown AW (1982) The origin of lipid phagocytes in the central nervous system: 1. The intrinsic microglia. J Comp Neurol 211:397–406
Morioka T, Kalechua AN, Streit W (1991) The microglial reaction in the rat hippocampus following transient forebrain ischemia. J Cereb Blood Flow Metabol 11:966–973
Gehrmann J, Banati RB, Hossmann KA, Kreutzberg GW (1995) Reactive microglia in cerebral ischaemia: an early mediator of tissue damage? Neuropathol Appl Neurobiol 21:277–289
Li Y, Chopp M, Jiang N, Zhang ZG, Zaloga C (1995) Induction of DNA fragmentation after 10 to 120 minutes of focal cerebral ischemia in rats. Stroke 26:1252–1258
Pollard H, Charriaut-Marlangue C, Cantagrel S, Represa A, Robain O, Moreau J, Ben-Ari Y (1994) Kainate-induced apoptotic cell death in hippocampal neurons. Neuroscience 63:7–18
Gehrmann J, Bonnekoh P, Miyazawa T (1992) The microglial reaction in the rat hippocampus following global ischaemia: immunoelectron microscopy. Acta Neuropathol 84:588–595
Deshpandel J, Bergstedt K, Linden T, Kalimo R, Wieloch T (1992) Ultrastructural changes in the hippocampal CA1 region following transient cerebral ischemia evidence against programmed cell death. Exp Brain Res 88:91–105
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Dohi, K., Shioda, S., Mizushima, H. et al. Delayed neuronal cell death and microglial cell reactivity in the CA1 region of the rat hippocampus in the cardiac arrest model. Med Electron Microsc 31, 85–93 (1998). https://doi.org/10.1007/BF01557785
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF01557785