Summary
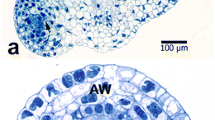
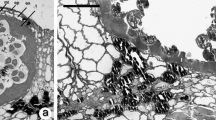
InMagnolia ×soulangeana pollen grains the generative cell (GC) does not become totally free within the vegetative cell (VC), at least until the pollen tube emergence. Due to a deviation in its detachment process from the sporoderm, the opposing ends of the VC plasmalemma do not fuse themselves when the GC moves away from the intine. Consequently, the interplasmalemmic space surrounding the GC does not become isolated but rather maintains continuity with the sporoderm through a complex formation that we have called plasmalemmic cord. The real existence of this formation was confirmed through serial sectioning showing the plasmalemmic cord to consist of the VC plasmalemma. In its initial portion it is occupied by a reasonably accentuated wall ingrowth of the inner layer of the intine (intine 3). In the remainder portion, neither of the cytochemical tests used in this work have revealed the presence of a significant amount of wall material. However, ultrathin sections of samples processed either chemically or by cryofixation showed the existence of an intricate system of tubules and vesicles, some of which are evaginations of the VC plasmalemma. The hypothesis that the plasmalemmic cord may have a role in the complex interactions between the two pollen cells is discussed.
Similar content being viewed by others
References
Albertini L, Grenet-Auberger H, Souvré A (1981) Polysaccharides and lipids in microsporocytes and tapetum ofRhoeo discolor Hance: cytochemical study. Acta Soc Bot Pol 50: 21–28
Bedinger P, Hardeman KJ, Loukides CA (1994) Travelling in style: the cell biology of pollen. Trends Cell Biol 4: 132–138
Currier HB, Strugger S (1956) Aniline blue and fluorescence microscopy of callose in bulb scales ofAllium cepa L. Protoplasma 45: 552–559
Dinis AM (1997) Estudo ultraestrutural e citoquímico da microsporogénese emMagnolia ×soulangeana Soul.-Bod. (Magnoliaceae). PhD thesis, Faculdade de Ciěncias e Tecnologia, Universidade de Coimbra, Coimbra, Portugal
—, Mesquita JF (1992) The ultrastructure of the vegetative cell during maturation of theMagnolia soulangeana Soul, pollen grain. In: Megías-Megías L, Rodríguez-García MI, Ríos A, Arias JM (eds) Electron microscopy 92, vol 3. Servicio de Publicaciones, Universidad de Granada, Granada, pp 429–430
— — (1994a) Ultrastructural and cytochemical evidence for the presence of peroxisomes in the generative cell ofMagnolia ×soulangeana pollen grain. Ann Bot 73: 83–90
— — (1994b) Intimate association between the generative cell and endoplasmic reticulum during the in vitro lag period ofMagnolia Xsoulangeana pollen germination. Int J Plant Sci 155: 158–166
Farragiana T, Marinozzi V (1979) Phosphotungstic acid staining of polysaccharides containing structures on epoxy embedded tissues. J Submicrosc Cytol 11: 263–265
Fitzgerald MA, Barnes SH, Blackmore S, Calder DM, Knox RB (1994) Pollen development and cohesion in a mealy and a hard type of orchid pollinium. Int J Plant Sci 155: 481–491
Galey FR, Nilsson SEG (1966) A new method for transferring sections from the liquid of the trough through staining solutions to the supporting film of a grid. J Ultrastruct Res 14: 405–410
Górska-Brylass A (1970) The “callose stage” of the generative cells in pollen grains. Grana 10: 21–30
Harris N, Oparka KJ, Walker-Smith DJ (1982) Plasmatubules: an alternative to transfer cells? Planta 156: 461–465
Humphrey CD, Pittman FE (1974) A simple methylene blue-azure II-basic fuchsin stain for epoxy-embedded tissue sections. Stain Technol 49: 9–14
Jensen WA (1962) Botanical histochemistry. WH Freeman, San Francisco
Kandasamy MK, Kappler R, Kristen U (1988) Plasmatubules in the pollen tubes ofNicotiana sylvestris. Planta 173: 35–41
Kaul V, Theunis CH, Palser BF, Knox RB, Williams EG (1987) Association of the generative cell and vegetative nucleus in pollen tubes ofRhododendron. Ann Bot 59: 227–235
Lancelle SA, Callaham DA, Hepler PK (1986) A method for rapid freeze fixation of plant cells. Protoplasma 131: 153–165
McCormick S (1993) Male gametophyte development. Plant Cell 5: 1265–1275
McCoy K, Knox RB (1988) The plasma membrane and generative cell organization in pollen of the mimosoid legume,Acacia retinodes. Protoplasma 143: 85–92
Mogensen HL (1992) The male germ unit: concept, composition, and significance. Int Rev Cytol 140: 129–147
Owens SJ, Westmuckett AD (1983) The structure and development of the generative cell wall inGibasis karwinskyana, G. venustula, andTradescantia blossfeldiana (Commelinaceae). In: Mulcahy DL, Ottaviano E (eds) Pollen: biology and implications for plant breeding. Elsevier Biomedical, New York, pp 149–158
Ramburg A (1969) Localisation ultrastructurale et nature du matériel coloré au niveau de la surface cellulaire par le mélange chromique-phosphotungstique. J Microsc 8: 325–342
Roland F (1971) Characterization and extraction of the polysaccharides of the intine and of the generative cell wall in the pollen grains of some Ranunculaceae. Grana 11: 101–106
Schlag M, Hesse M (1992) The formation of the generative cell inPolystachia pubescens (Orchidaceae). Sex Plant Reprod 5: 131–137
Spurr AR (1969) A low viscosity epoxy resin embedding medium for electron microscopy. J Ultrastruct Res 26: 31–43
Sunderland N, Huang B (1987) Ultrastructural aspects of pollen dimorphism. Int Rev Cytol 107: 175–220
Tanaka I (1997) Differentiation of generative and vegetative cells in angiosperm pollen. Sex Plant Reprod 10: 1–7
Testillano PS, Olmedilla A, Risueño MC, Colman O, Stockert JC (1988) Detección de polissacáridos en la pared del grano de polen. In: Civis Llovera J, Valle Hernández MF (eds) Actas del VI Simposio de Palinología (APLE). Universidad de Salamanca, Salamanca, pp 435–442
Theunis CH, McConchie CA, Knox RB (1985) Three-dimensional reconstruction of the generative cell and its wall connection in mature bicellular pollen ofRhododendron. Micron Microsc Acta 16: 225–231
Thiéry JP (1967) Mise en évidence des polysaccharides sur coupes fines en microscopie electronique. J Microsc 6: 987–1018
Tiwari SC (1994) An intermediate-voltage electron microscopic study of freeze-substituted generative cell in pear (Pyrus communis L.): features with relevance to cell-cell communication between the two cells of a germinating pollen. Sex Plant Reprod 7: 177–186
Twell D (1995) Diphtheria toxin-mediated cell ablation in developing pollen: vegetative cell ablation blocks generative cell migration. Protoplasma 187: 144–154
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Dinis, A.M., Mesquita, J.F. Ultrastructural study of the relationship between generative and vegetative cells inMagnolia ×soulangeana Soul.-Bod. pollen grains. Protoplasma 206, 87–96 (1999). https://doi.org/10.1007/BF01279255
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF01279255