Abstract
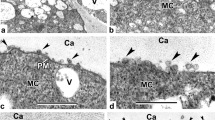
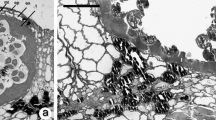
The morphological patterns of sporoderm development are illustrated by the example of pollen grains with different aperture types based on published results of transmission electron microscopy (TEM). It is noted that all studied pollen grains are characterized by the development of the periplasmic space between the plasmalemma and callose, where the primexine matrix and primexine begin to form. The aperture areas remain almost invariable during marking and formation of ectexine. The aperture plug and endexine are formed before dissolution of the callose. As the callose is dissolved, microspore is surrounded by a young sporoderm throughout the perimeter. Intine begins to form in the aperture with the pollen grain maturation. Intine displaces the aperture plug from the aperture center (pollen tube exit site) by the time of anthers opening. Direct conversion of the aperture plug into a complex multilayer intine occurs only during the formation of the outer layer of intine of some monocots.
Similar content being viewed by others
References
Ariizumi, T. and Toriyama, K., Genetic regulation of sporopollenin synthesis and pollen development, Ann. Rev. Plant. Biol., 2011, vol. 62, pp. 1–24.
Clement, C., Laporte, P., and Andran, J.C., The loculus content and tapetum during pollen development in Lilium, Sex. Plant. Rep., 1998, vol. 11, pp. 11–94.
El-Gazaly, G., Huysmans, S., and Smets, E.F., Pollen development of Rondeletia odorata (Rubiaceae), Am. J. Bot., 2001, vol. 88, no. 1, pp. 14–30.
Firon, N., Nepi, M., and Pacini, E., Water status and associated processes mark critical stages in pollen development and functioning, Ann. Bot., 2012a, vol. 109, pp. 1201–1213.
Firon, N., Nepi, M., and Pacini, E., Water status and associated processes mark critical stages in pollen development and functioning, Ann. Bot., 2012b, vol. 109, no. 7, pp. 1–13.
Gabaraeva, N.I., Ultrastructure and development of endextine lamellae in Manglietia tenuipes” (Magnoliaceae), Bot. Zh., 1987, vol. 72, no. 10, pp. 1310–1317.
Gabaraeva, N.I., Ultrastructural bases of the development of sporoderm in members of the dicotyledon subclass Magnoliidae, Doctoral Dissertation in Biology, St. Petersburg, 1997.
Gabaraeva, N.I. and Grigorjeva, V.V., Sporoderm development in Acer tataricum (Aceraceae): An interpretation, Protoplasma, 2010b, vol. 247, pp. 65–81.
Gabaraeva, N., Grigorjeva, V., and Kosenko, Y., 1. Primexine development in Passiflora racemosa Brot.: Overlooked aspects of development, Plant. Syst. Evol., 2012a, DOI 10.1007/s00606-013-0757-2 Print ISSN 0378-2697 Online ISSN 1615-6110.
Gabaraeva, N., Grigorjeva, V., and Kosenko, Y., 2. Exine development in Passiflora racemosa Brot.: Overlooked aspects of development, Plant. Syst. Evol., 2012b, DOI 10.1007/s00606-013-0756-3 Print ISSN 0378-2697 Online ISSN 1615-6110.
Gabaraeva, N., Grigorjeva, V., and Polevova, S., Exine and tapetum development in Symphytum officinale (Boraginaceae): Exine substructure and its interpretation, Plant. Syst. Evol., 2011, vol. 296, pp. 101–120.
Gabaraeva, N., Grigorjeva, V., Rowley, J.R., and Hemsley, A.R., Sporoderm development in Trevesia burckii (Araliaceae): 1. Tetrad period: Further evidence for the participation of self-assembly processes, Rev. Palaeobiol. Palyn., 2009a, vol. 156, pp. 211–232.
Gabaraeva, N., Grigorjeva, V., Rowley, J.R., and Hemsley, A.R., Sporoderm development in Trevesia burckii (Araliaceae): 2. Post-tetrad period: Further evidence for the participation of self-assembly processes, Rev. Palaeobiol. Palyn., 2009b, vol. 156, pp. 233–247.
Gabarayeva, N.I. and Grigorjeva, V.V., Sporoderm ontogeny in Chamaedorea micro-spandix (Arecaceae): Selfassembly as the underlying cause of development, Grana, 2010a, vol. 49, pp. 91–114.
Hess, M.W., Cell-wall development in freeze-fixed pollen: Intine formation of Ledebouria socialis (Hyacinthaceae), Planta, 1993, vol. 189, pp. 139–149.
Hess, M.W. and Frosch, A., Subunits of forming pollen exine and Ubish bodies as seen in freeze substituted Ledebouria socialis (Hyacinthaceae), Protoplasma, 1994, vol. 182, pp. 10–14.
Horner, H.T. and Pearson, C.B., Pollen wall and aperture development in Helianthus annuus (Compositae: Heliantheae), Am. J. Bot., 1978, vol. 65, no. 3, pp. 293–309.
Jimenez, J.U., Fernandez, P.H., Schlag, M.G., and Hesse, M., Pollen and tapetum development in male fertile Rosmarinus officinalis L. (Lamiaceae), Grana, 1996, vol. 34, pp. 305–316.
Kreunen, S.S. and Osborn, J.M., Pollen and anther development in Nelumbo (Nelumbonaceae), Am. J. Bot., 1999, vol. 86, no. 12, pp. 1662–1676.
Matveeva, N.P., Polevova, S.V., Smirnova, A.V., and Ermakov, I.P., Accumulation of sporopollenin in the envelope of microspore of Nicotiana tabacum L. during its development, Tsitologiya, 2012, vol. 54, no. 2, pp. 176–184.
Meier-Melikyan, N.R., Bovina, I.Yu., Kosenko, Ya.V., et al., Atlas pyl’tsevykh zeren astrovykh Asteraceae (Atlas of Pollen Grains of Asteraceae), Moscow: KMK, 2004.
Osborn, J.M., El-Ghazaly, G., and Cooper, R.L., Development of exineless pollen wall in Callitriche truncata (Callitrichaceae) and the evolution of underwater pollination, Plant. Syst. Evol., 2001, vol. 228, pp. 81–87.
Owens, S.J. and Dickinson, H.G., Pollen wall development in Gibasis (Commelinaceae), Ann. Bot., 1983, vol. 51, no. 1, pp. 1–15.
Polowick, P.L. and Sawnhey, V.K., An ultrastructural study of pollen development in tomato (Lycopersicon esculentum): 1. Tetrad to early binucleate microspore stage, Can. J. Bot., 1992, vol. 71, pp. 1039–1047.
Romero, A.T. and Fernandez, M.C., Development of exine and apertures in Fumaria densiflora DC. from the tetrad stage to maturity, in Pollen and Spores: Morphology and Biology, Harley, M.M., Morton, C.M., and Blackmore, S., Eds., 2000, pp. 45–56.
Rowley, J.R. and Skvarla, J.J., Pollen development in Epilobium (Onagraceae): Early microspore stages, Rev. Palaeobiol. Palyn., 2004, vol. 132, pp. 237–260.
Rowley, J.R. and Skvarla, J.J., Pollen development in Epilobium (Onagraceae): From microspore mitosis to formation of the intine, Grana, 2007, vol. 46, pp. 130–139.
Rowly, J.R., Skvarla, J.J., and Gabaraeva, N.I., Exine development in Borago (Boraginaceae): 2. Free microspore stages, Taiwania, 1999, vol. 44, no. 2, pp. 212–229.
Saad-Limam, S.B., Nabli, M.A., and Rowley, J.R., Exine ontogeny in Borago officinalis pollen, Grana, 2002, vol. 41, pp. 216–226.
Stone, D.E., Sellers, S.C., and Kress, W.J., Ontogeny of exineless pollen in Heliconia, a banana relative, Ann. Miss. Bot. Gard., 1979, vol. 66, pp. 701–730.
Takahashi, M., Development of omniaperturate pollen in Trillium kamtschaticum (Liliaceae), Am. J. Bot., 1987, vol. 74, no. 12, pp. 1842–1852.
Takahashi, M., Development of the echinate pollen wall in Farfugium japonicum, Bot. Mag. Tokyo, 1989a, vol. 102, pp. 219–234.
Takahashi, M., Pattern determination of the exine in Caesalpinia japonica (Leguminosae: Caesalpinioideae), Am. J. Bot., 1989b, vol. 76, no. 11, pp. 1615–1626.
Takahashi, M. and Kouchi, J., Ontogenetic development of spinous exine in Hibiscus syriacus (Malvaceae), Am. J. Bot., 1988, vol. 75, no. 10, pp. 1549–1558.
Taylor, M.L. and Osborn, J.M., Pollen ontogeny in Brasenia (Cabombaceae, Nymphaeales), Am. J. Bot., 2006, vol. 93, no. 3, pp. 344–356.
Volkova, O.A., Severova, E.E., and Polevova, S.V., Development of sporoderm of pollen grains of Lythrum salicaria (Lythraceae), Byull. Mosk. Obshchest. Ispyt. Prir. Otd. Biol., 2012, vol. 117, no. 5, pp. 57–63.
Weber, M. and Ulrich, S., The endexine: A frequently overlooked pollen wall layer and a simple method for detection, Grana, 2010, vol. 49, pp. 83–90.
Zavada, M.S. and Anderson, G.J., The wall and aperture development of pollen from dioecious Solanum appendiculatum: What is inaperturate pollen?, Grana, 1997, vol. 36, pp. 129–134.
Zolala, H. and Polevova, S., Sporoderm development in Pratia begonifolia Lindl. (Lobeliaceae, Asterales), Wulfenia, 2009, vol. 16, pp. 51–60.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Polevova, S.V. Important stages in the development of different layers and areas of sporoderm in angiosperms. Paleontol. J. 48, 1324–1329 (2014). https://doi.org/10.1134/S0031030114120077
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0031030114120077