Summary
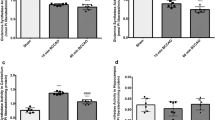
The time course of changes in monoamine metabolism in ischemic striatum was assessed by measurement of levels of dopamine (DA), dihydroxyphenylacetic acid (DOPAC), homovanillic acid (HVA), serotonin (5-HT) and 5-hydroxy-indole-acetic acid (5-HIAA) 2, 4, 7 and 16 hours after irreversible unilateral carotid ligation in Mongolian gerbils with stroke. DA was reduced to 30% of the level in the contralateral non-ischemic striata by 2 hours after stroke, but DOPAC was significantly elevated (p < 0.01) to 227%, while HVA remained equal to control. At 4 hours after stroke, DOPAC was 86% of the contralateral non-ischemic striata but HVA had risen to 130%. At 7 hours after stroke, DOPAC in the ischemic striata was 148% of control, while HVA remained at 133%. By 16 hours after stroke, DA, DOPAC and HVA were depleted from the ischemic striata, corresponding to the time course for irreversible damage to the neurotransmitter uptake function of nerve terminals. 5-HT levels in the ischemic striata were 30% of control at 2 hours, 46% at 4 hours, 30% at 7 hours and 21% at 16 hours, while 5-HIAA remained equal to control throughout the time course. These studies indicate that monoamine metabolism continues in ischemic striatum for up to 8 hours after the onset of stroke following irreversible unilateral carotid ligation in the Mongolian gerbil, but metabolism of DA is disrupted by 16 hours after stroke while metabolism of 5-HT continues.
Similar content being viewed by others
References
Ahagon A, Ishikawa M, Nanda H (1980) Histochemical changes of brain dopamine in acute stage of cerebral ischemia in gerbils. Stroke 11: 622–628
Astrup J, Siesjo BK, Symon L (1981) Thresholds in cerebral ischemia—the ischemic penumbra. Stroke 12: 723–725
Berry K, Wisniewski HM, Svarzebein L, Baez S (1975) On the relationship of brain vasculature to production of neurological deficit and morphological changes following acute unilateral common carotid artery ligation in gerbils. J Neurol Sci 25: 75–92
Brannan T, Weinberger J, Knott P, Taff I, Kaufmann H, Togasaki D, Nieves-Rosa J, Maker H (1987) Direct evidence of acute, massive striatal dopamine release in gerbils with unilateral strokes. Stroke 18: 108–110
Brown AW, Brierley JB (1975) The nature, distribution and earliest stages of anoxic-ischaemic nerve cell damage in the rat brain as defined by the optical microscope. Br J Exp Pathol 49: 87–106
Elchisak MA, Maas JW, Roth RH (1977) Dihydroxyphenylacetic acid conjugate: Natural occurence and demonstration of probenecid-induced accumulation in rat striatum, olfactory tubercles and frontal cortex. Eur J Pharmacol 41: 361–378
Ferguson GA (1971) Statistical analysis in psychology and education, 3rd edn. McGraw-Hill, New York, pp 492
Gaudet R, Welch KMA, Chabi E, Wang TP (1978) Effect of transient ischemia on monoamine levels in the cerebral cortex of gerbils. J Neurochem 30: 751–757
Harik SI, Yoshida S, Busto R, Ginsberg MD (1986) Monoamine neurotransmitters in diffuse reversible forebrain ischemia and early recirculation: Increased dopaminergic activity. Neurology 36: 971–976
Ito U, Spatz M, Walker JT Jr, Klatzo I (1975) Experimental cerebral ischemia in Mongolian gerbils. I. Light microscopic observations. Acta Neuropathol 32: 209–233
Kahn K (1972) The natural course of experimental cerebral infarction in the gerbil. Neurology 22: 510–514
Lavyne MH, Moskowitz MA, Larin F, Zervas NT, Wurtman RJ (1975) Brain3H-catecholamine metabolism in experimental cerebral infarction. Neurology 25: 483–485
Levine S, Payan H (1966) Effects of ischemia and other procedures on the brain and retina of the gerbil (Meriones unguiculatus). Exp Neurol 16: 255–262
Levy DE, Brierley JB (1979) Delayed pentobarbital administration limits ischemic brain damage in gerbils. Ann Neurol 5: 59–64
Lust WD, Mrsulja BB, Mrsulja BJ, Passoneau JV, Klatzo I (1975) Putative neurotransmitters and cyclic nucleotides in prolonged ischemia of the cerebral cortex. Brain Res 98: 394–399
Mrsulja BB, Mrsulja BJ, Spatz M, Klatzo I (1975) Action of cerebral ischemia on decreased levels of 3-methyl-4-hydroxy-phenylethglycol sulfate, homovanillic acid and 5-hydroxy-indoleacetic acid produced by pargyline. Brain Res 98: 388–393
Mrsulja BB, Mrsulja BJ, Spatz M, Ito U, Walker JT Jr, Klatzo I (1976) Experimental cerebral ischemia in mongolian gerbils, IV. Behavior of biogenic amines. Acta Neuropath 36: 1–8
Nakai K, Welch KMA, Meyer JS (1977) Critical cerebral blood flow for production of hemiparesis after unilateral carotid occlusion in the gerbil. J Neurol Neurosurg Psychiatry 40: 595–599
Silverstein F, Johnston MV (1984) Effects of hypoxia-ischemia on mono-amine metabolism in the immature brain. Ann Neurol 15: 342–347
Wagner J, Palfreyman M, Zraika M (1979) Determination of DOPA, dopamine, DOPAC, epinephrine, norepinephrine, alpha-monofluoromethyldopa and alpha-difluoro methyldopa in various tissues of mice and rats using reversed-phase ion-pair liquid chromatography with electrochemical detection. J Chromatogr 164: 41–54
Weinberger J, Cohen G (1982) The differential effect of ischemia on the active uptake of dopamine, gamma-aminobutyric acid and glutamate by brain synaptosomes. J Neurochem 38: 963–968
Weinberger J, Cohen G, Nieves-Rosa J (1983) Nerve terminal damage in cerebral ischemia: greater susceptibility of catecholamine nerve terminals relative to serotonin nerve terminals. Stroke 14: 986–989
Zervas NT, Hori H, Negora M, Wurtman RJ, Larin F, Lavyne MH (1974) Reduction of brain dopamine following experimental cerebral ischemia. Nature 274: 283–284
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Weinberger, J., Nieves-Rosa, J. Metabolism of monoamine neurotransmitters in the evolution of infarction in ischemic striatum. J. Neural Transmission 69, 265–275 (1987). https://doi.org/10.1007/BF01244347
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF01244347