Abstract
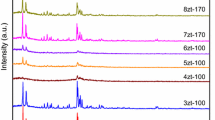
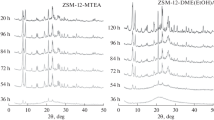
Pentasil zeolites of ZSM-5 type are synthesised hydrothermally using triethyl-n-proplyammonium bromide (TEPA-Br) and triethyl-n-butylammonium bromide (TEBA-Br). The crystallization kinetics, followed by XRD, SEM and thermal analysis, clearly demonstrate the influence of size and molecular weight of the templating quaternary ammonium cation (QAC) species on the rates of nucleation and crystallization. The values of the apparent activation energies for nucleation and crystal growth indicate that both nucleation and crystal growth are faster when TEPA-Br rather than TEBA-Br is used as a template. The quantitative identification of intergrown phases characterizes both the phases to be ZSM-5 zeolite. Thermoanalytical curves for both these zeolites in as-synthesised forms exhibit two-step oxidative decomposition of the occluded organic species. This suggests that the quaternary ammonium cation may be located at two energetically different sites within the zeolite channels. The equilibrium sorption capacity, however, is found to increase in the order of size and molecular weight of the templating species in both the zeolites. The nature of acid site distribution, obtained from the temperature programmed desorption of ammonia is found to be independent of the templating species used during the synthesis.
Similar content being viewed by others
References
R. J. Argauer and G. R. Landolt:U.S. Pat. 3,702,886 (1972)
A. Erdem and L. B. Sand:U.S. Pat. 60, 241 (1979).
S. B. Kulkarni, V. P. Shiralkar, A. N. Kotasthane, R. B. Borade and P. Ratnasamy:Zeolites 2, 313 (1982).
A. N. Kotasthane, V. P. Shiralkar, S. G. Hedge and S. B. Kulkarni:Zeolite 6, 253 (1986).
Z. Gabelica, M. Cavez-Bierman, P. Boudart, A. Gourgue and J. B. Nagy:Zeolite Synthesis, Structure, Technology and Applications, Studies in Surface Science and Catalysis, 24, B. Drzaj, S. Hocevar and S. Pejovnik, Eds., Elsevier, New York, 1985. p. 55.
E. Narita, K. Sato and T. Okabe:Chem. Lett. 1055 (1984).
M. Avrami:J. Chem. Phys. 9, 177 (1941) and B. V. Erofeev, C.R. Acad. Sci., U.S.S.R. 52, 511 (1946).
Z. Gabelica, N. Blom and E. G. Derouane:Appl. Catal. 5, 227 (1983).
B. M. Lok, T. R. Cannan and C. A. Messina:Zeolites 3, 282 (1983).
K. J. Chao, T. C. Tasi, M. S. Chen and I. Wang:J. Chem. Soc. Faraday Trans. 1 77, 547 (1981).
F. Paulik, J. Paulik and L. Erdey:Talanta 13, 1405 (1946).
K. J. Chao:Proc. Natl. Sci. Counc., ROC,3, 233 (1979).
D. M. Biddy, N. B. Milestone and L. P. Aldridge:Nature (London) 285, 30 (1980).
A. N. Kotasthane and V. P. Shiralkar:Thermochim. Acta 102, 37 (1986).
V. S. Nayak and L. Rickert:Acta Phys. Chem. 31, 157 (1985).
R. Mostovich and L. B. Sand:Zeolites 2, 143 (1982).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Joshi, P.N., Rao, G.N., Kotasthane, A.N. et al. Influence of template on crystallization of ZSM-5 zeolites. J Incl Phenom Macrocycl Chem 9, 91–99 (1990). https://doi.org/10.1007/BF01041253
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF01041253