Abstract
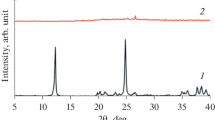
Highly crystalline ZSM-5 zeolites are successfully prepared from kaolinite as low cost precursor. The effects of SiO2/Al2O3 molar ratio, the presence of tetrapropylammonium bromide (TPABr) and the initial precursor on the textural properties of the final products have been investigated. Crystallized samples have been characterized by scanning electron microscopy and nitrogen adsorption in addition to X-ray diffraction. The results show that getting pure ZSM-5 zeolite with a high crystallinity degree is directly dependent on the starting precursor as well as on the presence of the organic template highlighting the role of SiO2/Al2O3 molar ratio. The directed-template ZSM-5 sample prepared frommetakaolinite with the smallest particles (crystal size of 700 nm) and a maximum crystallinity of 98% is obtained at a SiO2/Al2O3 molar ratio of 31.69. Increasing the SiO2/Al2O3 molar ratio to 41.13 isrequired to prepare an organic-template free ZSM-5 from metakaolinite with a relative crystallinity of 81%. In order to synthesize ZSM-5 zeolite from the acid-activated metakaolinite as the only silica and alumina sources, a SiO2/Al2O3 molar ratio of 76.19 is used, the maximal crystallinity degree is 79%, with the largest ZSM crystals of about 3000 nm.
Similar content being viewed by others
References
Cundy, C.S. and Cox, P.A., Chem. Rev., 2003, vol. 103, no. 3, pp. 663–702.
Tosheva, L. and Valtchev, V.P., Chem. Mater., 2005, vol. 17, no. 10, pp. 2494–2513.
Bhat, Y.S., Das, J., Rao, K.V., and Halgeri, A.B. J. Catal., 1996, vol. 159, no. 2, pp. 368–374.
Ren, N., Yang, Z.J., Lv, X.C., Shi, J., et al., Micropor. Mesopor. Mater., 2010, vol. 131, nos. 1–3, pp. 103–114.
Armarolia, T., Simona, L.J., Dignea, M., Montanarib, T., et al., Appl. Catal., A, 2006, vol. 306, pp. 78–84.
Barbera, K., Bonino, F., Bordiga, S., Janssens, et al., J. Catal., 2011, vol. 280, no. 2, pp. 196–205.
Herrmann, C., Haas, J., and Fetting, F., Appl. Catal., 1987, vol. 35, no. 2, pp. 299–310.
Takamitsu, Y., Yamamoto, K., Yoshida, S., and Ogawa, H., J. Porous Mater., 2014, vol. 21, no. 4, pp. 433–440.
Pan, F., Lu, X., Wang, Y., Chen, S., et al., Mater. Lett., 2014, vol. 115, pp. 5–8.
Fouad, O.A., Mohamed, R.M., Hassan, M.S., and Ibrahim, I.A., Catal. Today, 2006, vol. 116, no. 2, pp. 82–87.
Sang, S., Chang, F., Liu, Z., He, C., et al., Catal. Today, 2004, vols. 93–95, pp. 729–734.
Soltanali, S., Halladj, R., Rashidi, A., and Bazmi, M., ADV Powder Technol., 2014, vol. 25, pp. 1767–1771.
Pan, F., Lu, X., Zhu, Q., Zhang, Z., et al., J. Mater. Chem. A, 2014, vol. 2, pp. 20667–20675.
Xianliang, H., Rongrong, Z., and Zhengbao, W., J. Catal., 2012, vol. 33, nos. 7–8, pp. 1290–1298.
Yu, Q., Zhang, Q., Liu, J., Li, C., et al., Cryst. Eng. Commun., 2013, vol. 15, no. 38, pp. 7680–7687.
Kim, S.D., Noh, S.H., Park, J.W., and Kim, W.J., Micropor. Mesopor. Mater., 2006, vol. 92, nos. 1–3, pp. 181–188.
Abdmeziem, K. and Siffert, B., Appl. Clay Sci., 1994, vol. 8, no. 6, pp. 437–447.
Kim, D.J. and Chung, H.S., Appl. Clay Sci., 2003, vol. 24, no. 12, pp. 69–77.
Othman Ali, I., Mostafa Hassan, A., Mohamed Shaaban, S., and Seifelnasser Soliman, K., Sep. Purif. Technol., 2011, vol. 83, pp. 38–44.
Kovo, A.S., Hernandez, O., and Holmes, S.M., J. Mater. Chem., 2009, vol. 19, no. 1, pp. 6207–6212.
Wang, P., Shen, B., Shen, D., Peng, T., and Gao, J., Catal. Commun., 2007, vol. 8, no. 10, pp. 1452–1456.
Ye, L., Xianbo, Y., Lei, Q., Jingdai, W., et al., China Petrol. Process. Petrochem. Technol., 2010, vol. 12, no. 1, pp. 23–28.
Hui, F., Chunyi, L., and Honghong, S., Appl. Clay Sci., 2009, vol. 42, nos. 1–3, pp. 439–445.
Lafi, A.A.F., Matam, S.K., and Hodali, H.A., Ind. Eng. Chem. Res., 2015, vol. 54, no. 15, pp. 3754–3760.
Goodyear, J. and Duffin, W.J., Miner. Mag., 1961, vol. 32, no. 254, pp. 902–907.
Murray, H.H., Appl. Clay Sci., 2000, vol. 17, nos. 5–6, pp. 207–221.
Shirazi, L., Jamshidi, E., and Ghasemi, M.R., Cryst. Res. Technol., 2008, vol. 43, no. 12, pp. 1300–1306.
Persson, A.E., Schoeman, B.J., Sterte, J., and Otterstedt, J.E., Zeolites, 1995, vol. 15, no. 7, pp. 611–619.
Author information
Authors and Affiliations
Corresponding author
Additional information
The article is published in the original.
About this article
Cite this article
Ghrib, Y., Frini-Srasra, N. & Srasra, E. Synthesis of ZSM-5 zeolite from metakaolinite: Effects of the SiO2/Al2O3 molar ratio, the initial precursor and the presence of organic template agent. Surf. Engin. Appl.Electrochem. 53, 64–70 (2017). https://doi.org/10.3103/S1068375517010057
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.3103/S1068375517010057