Abstract
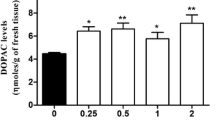
Effects of DSP-4 on noradrenaline (NA), 3-methoxy-4-hydroxyphenyl glycol (MHPG), serotonin (5-HT) and 5-hydroxyindole acetic acid (5-HIAA) levels and on beta adrenoceptor binding kinetics (Bmax and KD) in rat hippocampus, cortex and hypothalamus were studied between 24 hours and 14 days after systemic administration. Beta adrenoceptor numbers in hippocampus and cortex, but not in hypothalamus, were significantly increased after DSP-4. No significant changes in KD values were observed in hypothalamus, but significant increases in this parameter were measured in hippocampus and cortex. NA and MHPG levels were significantly decreased in all three brain regions, but MHPG/NA ratios were increased in hippocampus, decreased in cortex and unchanged in hypothalamus. Very prominent increases in 5-HIAA levels were observed in all three brain regions, but only at one day after DSP-4. The greatest increases in 5-HIAA levels occurred in the hippocampus, but this effect of DPS-4 appeared to be slightly diminished by pre-treatment with fluoxetine. In cortex and hippocampus 5-HT levels were slightly, but significantly decreased after DSP-4.
Similar content being viewed by others
References
Jonsson, G., Hallman, H., Ponzio, F. and Ross, S. 1981. DSP-4(N-(2-chloroethyl)-N-ethyl-2-bromobenzylamine) a useful denervation tool for central and peripheral noradrenaline neurons. Eur. J. Pharmacol. 72:173–188.
Dudley, M. W., Howard, B. D., and Cho, A. K. 1990. The interaction of the beta-haloethyl benzylamines, xylamine and DSP-4 with catecholaminergic neurons. Ann. Rev. Pharmacol. Toxicol. 30:387–403.
Grzanna, R., Berger, U., Fritschy, J., and Gefford, M. 1989. Acute action of DSP-4 on central norepinephrine axons: Biochemical and immunohistochemical evidence for differential effects. J. Histochem. Cytochem. 37:1435–1442.
Lookingland, K. J., Chapin, D. S., McKay, D. W., and Moore, K. E. 1986. Comparative effects of the neurotoxins N-chloroethyl-N-ethyl-2-bromobenzylamine hydrochloride (DSP-4) and 6-hydroxydopamine on hypothalamic noradrenergic, dopaminergic and 5-hydroxytrypraminergic neurons in the male rat. Brain Res. 365:228–234.
Hallman, H., Sundström, E., and Jonsson, G. 1984. Effects of the noradrenaline neurotoxin DSP-4 on monoamine neurons and their transmitter turnover in rat CNS. J. Neural Transm. 60:89–102.
Eison, A. S., Eison, M. S., Yocca, F. D., and Gianutose, G. 1989. Effects of imipramine and serotonin-2 agonists and antagonist on serotonin-2 and beta-adrenergic receptors following noradrenergic or serotonergic denervation. Life Sci. 44:1419–1427.
Zahniser, N. R., Weiner, G. R., Worth, T. R., Philpott, K., Yasuda, R. P., Jonsson, G., and Dunwiddie, T. V. 1986. DSP-4 induced noradrenergic lesions increase β-adrenergic receptors and hippocampal electrophysiological responsiveness. Pharmacol. Biochem. Behav. 24:1397–1402.
Mogilnicka, E. 1986. Increase in β- and α1-adrenoceptor binding sites in the rat brain and in the α1-adrenoceptor functional sensitivity after DSP-4-induced noradrenergic denervation. Pharmacol. Biochem. Behav. 25:743–746.
Logue, M. P., Growdon, J. H., Coviella, I. L. G., and Wurtman, R. J. 1985. Differential effects of DSP-4 administration on regional brain norepinephrine turnover in rats. Life Sci. 37:403–409.
Hutson, P. H., and Curzon, G. 1986. Dopamine metabolites in rat cisternal cerebrospinal fluid: Major contribution from extrastriatal dopamine neurones. J. Neurochem. 46:186–190.
Riva, M. A., and Creese, I. 1989. Comparison of two putatively selective radioligands for labelling central nervous system β-adrenergic receptors: Inadequacy of [3H] dihydroalprenolol. Mol. Pharmacol. 36:201–210.
Tiong, A. H. K., and Richardson, J. S. 1990. Characterization of rat cerebral cortical beta adrenoceptor subtypes using [125I]-Iodocyanopindolol. J. Recept. Res. 9:495–508.
Munson, P. J., and Rodbard, D. 1980. Ligand: A versatile computerized approach for characterization of ligand-binding systems. Anal. Biochem. 107:220–239.
Lowry, O. H., Rosebrough, N. J., Farr, A. L., and Randall, R. J. 1951. Protein measurements with the Folin phenol reagent. J. Biol. Chem. 193:265–275.
Allin, R., Russell, V. A., Lamm, M. C. L., and Taljaard, J. J. F. 1988. Regional distribution of monoamines in the nucleus accumbens of the rat. Neurochem. Res. 13:937–942.
De Villiers, A. S., Russell, V. A., and Taljaard, J. J. F. 1992. Effect of corticosterone on noradrenergic nuclei in the pons-medulla and [3H] NA release from terminals in hippocampal slices. Neurochem. Res. 17:273–280.
Dooley, D. J., Heal, D. J., and Goodwin, G. M. 1986. Repeated electroconvulsive shock prevents increased neocortical β1-adrenoceptor binding after DSP-4 treatment in rats. Eur. J. Pharmacol. 134:333–337.
Swann, A. C. 1984. Brain (Na+, K+)-ATPase and noradrenergic function; recovery of enzyme activity after norepinephrine depletion. Brain Res. 321:323–326.
Johnson, M., Hanson, G. R., and Gibb, J. W. 1991. Norepinephrine does not contribute to metamphetamine-induced changes in hippocampal serotonergic system. Neuropharmacol. 30:617–622.
Dudley, M. W., Siegel, B. W., Ogden, A. M., and McCarty, D. R. 1988. A low dose of xylamine produces sustained and selective decreases in rat brain norepinephrine without evidence of neuronal degeneration. J. Pharmacol. Exp. Ther. 247:174–179.
Johnson, G. A., Kim, E. G. and Boukma, S. J. 1972. 5-Hydroxyindole levels in rat brain after inhibition of dopamine β-hydroxylase. J. Pharmacol. Exp. Ther. 180:539–546.
Frankhuijzen, A. L., and Mulder, A. H. 1980. Noradrenaline inhibits [3H]serotonin release from slices of rat hippocampus. Eur. J. Pharmacol. 63:179–182.
Raiteri, M., Maura, G., Folghera, S., Cavazzani, P., Andrioli, G. C., Schlicker, E., Schalmus, R., and Göthert, M. 1990. Modulation of 5-hydroxytryptamine release by presynaptic inhibitory α2-adrenoceptors in the human cerebral cortex. Naunyn-Schmied. Arch. Pharmacol. 342:508–512.
Frankhuijzen, A. L., and Mulder, A. H. 1982. Pharmacological characterization of presynaptic α-adrenoceptors modulating [3H] noradrenaline and [3H] 5-hydroxytryptamine release from slices of the hippocampus of the rat. Eur. J. Pharmacol. 81:97–106.
Benkirane, S., Arbilla, S., and Langer, S. Z. 1985. Supersensitivity of α2-adrenoceptors modulating [3H]5-HT release after noradrenergic denervation with DSP-4. Eur. J. Pharmacol. 119:131–133.
Kuhn, D. M., Wolf, W. A., and Youdin, B. H. 1986. Serotonin neurochemistry revisited: A new look at some old axioms. Neurochem. Int. 8:141–154.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Theron, C.N., de Villers, A.S. & Taljaard, J.J.F. Effects of DSP-4 on monoamine and monoamine metabolite levels and on beta adrenoceptor binding kinetics in rat brain at different times after administration. Neurochem Res 18, 1321–1327 (1993). https://doi.org/10.1007/BF00975054
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00975054