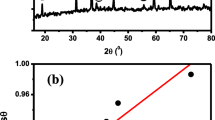
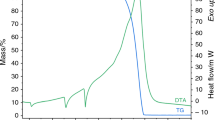
Conclusions
-
1.
The possibility of obtaining tetramethyl- (TMAS) and tetraethylammonium (TEAS) superperoxides with a content of the basic substance of 97 and 98%, respectively, in the exchange of their hydroxides with potassium superperoxide in the solid state, followed by extraction with liquid ammonia, was demonstrated.
-
2.
The TMAS and TEAS obtained were identified by chemical analysis, by the methods of ESR, magnetochemistry, and UV spectroscopy. Their solubility in liquid ammonia was determined.
-
3.
The limits of thermal stability of TMAS and TEAS were determined by the method of differential thermal analysis. The possibility of the formation of tetrabutylammonium superperoxide was demonstrated.
Similar content being viewed by others
Literature cited
A. D. McElroy and J. S. Hashman, Inorgan. Chem.,3, 1798 (1964).
G. Csapski and B. Halperin, Israel J. Chem.,5, 185 (1967).
E. Seyb and J. Kleinberg, Analyt. Chem.,23, 115 (1951).
Author information
Authors and Affiliations
Additional information
Translated from Izvestiya Akademii Nauk SSSR, Seriya Khimicheskaya, No. 8, pp. 1684–1686, August, 1974.
Rights and permissions
About this article
Cite this article
Latysheva, E.I., Cherkasov, É.N., Tokareva, S.A. et al. Synthesis and certain physicochemical properties of tetraalkylammonium superperoxides. Russ Chem Bull 23, 1610–1612 (1974). https://doi.org/10.1007/BF00923173
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00923173