Abstract
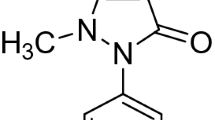
The chelation behaviour of complexes of La(III), Pr(III), Nd(III), Sm(III), Y(III), Tb(III), Dy(III), Ho(III) with ω-benzoyl-2-hydroxy-4-methoxy-3-methyl-acetophenone has been studied potentiometrically in 75% (v/v) aqueous alcohol medium at various ionic strengths. The method ofBjerrum andCalvin, as modified byIrving andRossotti, has been used to find values of\(\bar n\) andpL. The stability constants and the values ofS min have been calculated. The order of stability constants was found to be: La<Pr<Nd<Sm<Y<Tb<Dy<Ho.
Zusammenfassung
Das Chelationsverhalten von La(III), Pr(III), Nd(III), Sm(III), Y(III), Tb(III), Dy(III) und Ho(III) mit ω-Benzoyl-2-hydroxy-4-methoxy-3-methyl-acetophenon wurde in 75% (v/v) wä\rigem Alkohol bei verschiedenen Ionenstärken potentiometrisch untersucht. Mittels der Methode vonBjerrum undCalvin, in der Modifikation nachIrving undRossotti, wurden Werte für\(\bar n\) undpL bestimmt. Die Stabilitätskonstanten und die Werte fürS min wurden berechnet. Die Ordnung der Stabilitätskonstante war: La<Pr<Nd<Sm<Y<Tb<Dy<Ho.
Similar content being viewed by others
References
Irving HM,Rossotti HS (1954) J Chem Soc: 2904
Sullivan JC, Rydberg J, Miller WF (1959) Acta Chem Scand 13: 2023
Rydberg J, Sullivan JC (1959) Acta Chem Scand 13: 2059
Martell AE, Smith RM (1974) Critical stability constant, vol 1. Plenum, New York
Nair PKR, Srinivasulu K (1979) J Inorg Nucl Chem 41: 251
Nasanen R, Ekman A (1952) Acta Chem Scand 6: 1389
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Dixit, R., Garg, B.S. Potentiometric studies on the chelation behaviour of ω-benzoyl-2-hydroxy-4-methoxy-3-methyl-acetophenone (BHMMA) with lanthanons. Monatsh Chem 118, 1113–1117 (1987). https://doi.org/10.1007/BF00811284
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00811284