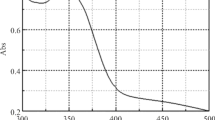
The complexation reactions of Fe3+, Cu2+ and Pd2+ ions with tetracycline HCl (TC) in water or DMF at 15, 25, 35 and 45 ±0.1°C were studied by the spectrophotometric method. The complexation process was optimized in terms of pH, temperature, and time. The stoichiometry (metal ion/ligand ratio) of the complexes was found to be 1 : 2 for Pd(II) – TC and Fe(III) – TC and 1 : 1 for Cu(II) – TC. The formation constants of the obtained complexes were determined from computer fitting of the absorbance – molar ratio data and emphasized by the KINFIT program. The values of the thermodynamic parameters for complexation reactions were estimated from the temperature dependence of the stability constants. TC could be determined by measuring the absorbance of each complex at its characteristic wavelength λmax.









Similar content being viewed by others
References
S. Harbarth, W. Albrich, D. A. Goldmann, and J. Huebner, Lancet Infect. Dis., 1, 251 (2001).
L. A. Mitscher, S. P. Pillai, E. J. Gentry, and D. M. Shankel, Med. Res. Rev., 19, 477 (1999).
I. Berber, C. Cokmus, and E. Atalan, Mikrobiologia, 72, 54 (2003).
R. L. Lintvedt and B. C. Glick, Inorg. Chem., 15, 1633 (1976).
D. E. Fenton and J. R. Tate, Inorg. Chim. Acta, 23, 83 (1984).
W. A. Baker and P. M. Brown, J. Am. Chem. Soc., 88, 1314 (1966).
S. M. Sultan, F. O. Suliman, S. O. Duffuaa, and I. I. Abu-Abdoun, Analyst, 117, 1179 (1966).
H. Poiger and Ch. Schlatter, Analyst, 101, 808 (1976).
Z. Gong and Z. Zhang, Anal. Chim. Acta, 351, 205 (1997).
A. Regos, G. Zuk, and J. D. Lukasiak, Sci. Pharm., 46, 249 (1978).
M. A. Ghandour and A. M. M. Ali, Anal. Lett., 24, 2171 (1991).
C. M. Couto, J. L. F. C. Lima, M. Conceição, et al., J. Pharm. Biomed. Anal., 18, 527 (1998).
T. Charoenraks, S. Chuanuwatanakul, K. Honda, et al., Anal. Sci., 21, 241 (2005).
M. S. Mahrous and M. M. Abdel-Khalek, Talanta, 31, 289 (1984).
U. Saha, A. K. Sen, T. K. Das, and S. K. Bhowal, Talanta, 37, 1193 (1990).
V. A. Nicely and J. L. Dye, J. Chem. Educ., 48, 443 (1990).
G. W. Everett, J. Gulbis, and J. Shaw, J. Am. Chem. Soc., 104, 445 (1982).
J. Shaw and G. W. Everett, Inorg. Biochem., 17, 305 (1982).
T. Sawal and A. Yamaguchi, J. Biol. Chem., 265, 4809 (1990).
L. Lambs and M. Brion, Inorg. Chim. Acta, 106, 151 (1983).
M. Brion and G. Berton, Inorg. Chim. Acta, 55, 47 (1981).
J. Rokos and M. Burger, Nature, 181, 1201 (1958).
L. Laco and J. Korlnek, Clin. Chim. Acta, 4, 800 (1959).
D.P. Rall and T. L. Loo, J. Natl. Cancer Inst., 19, 79 (1957).
W. E. Wenthworth, J. Chem. Educ., 42, 96 (1962).
M. J. D. Powell, Comput. J., 7, 155 (1964).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Gh., A.B. Thermodynamic Studies of Metal Complexes of Tetracycline and their Application in Drug Analysis. Pharm Chem J 48, 483–487 (2014). https://doi.org/10.1007/s11094-014-1136-1
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11094-014-1136-1