Abstract
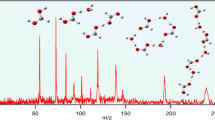
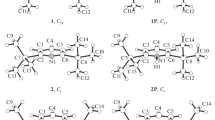
The reduced mobility of protonated pyrazole derivatives was measured by ion mobility spectrometry (IMS) in air, nitrogen, and carbon dioxide, at temperatures between 150 and 250°C. It was found that the mobility of protonated 5-amino-1-phenylpyrazole was higher than that of its 3- and 4-isomers. This was attributed to the fact that in the 5-isomer the preferred site of protonation is on the endocyclic nitrogen, which leads to delocalization of the ionic charge, and thus to a diminished interaction with the drift gas molecules. On the other hand, protonated 5-amino-1-methylpyrazole has a slightly lower mobility than its isomers, which is indicative of a different protonation mechanism.
Similar content being viewed by others
References
Garrone, A.; Fruttero, R.; Tironi, C.; Gasco, A.J. Chem. Soc. Perkin Trans.,1989,2, 1941.
Katritzky, A. R.;Lagowski, J. M.The Principles of Heterocyclic Chemistry; Methuen, London,1967.
Karpas, Z.Anal. Chem.,1989,61, 684.
Karpas, Z.Int. J. Mass Spectrom. Ion Proc.,1989,93, 237.
Karpas, Z.; Berant, Z.; Stimac, R. M.Struc. Chem.,1990,1, 201.
Berant, Z.; Karpas, Z.; Shahal, O.J. Phys. Chem.,1989,93, 7529;
Karpas, Z.; Berant, Z.J. Phys. Chem.,1989,95, 3021.
Cohen, M. J.; Spangler, G. E. InPlasma Chromatography; Carr, T. W., Ed.; Plenum Press, New York,1984,1, 41.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Karpas, Z., Tironi, C. The mobility and ion structure of protonated aminoazoles. Struct Chem 2, 655–659 (1991). https://doi.org/10.1007/BF00676828
Issue Date:
DOI: https://doi.org/10.1007/BF00676828