Summary
-
1.
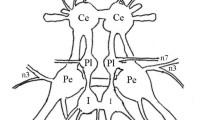
Electrical stimulation of the hindwing tegula inSchistocerca gregaria can elicit synchronous firing in tegula afferents. Recordings from the metathoracic wing sensory nerve, 1C1, showed that both the exact position of the stimulus wires in the tegula and the stimulus voltage determined which of two groups of tegula afferents were predominantly activated (Fig. 2).
-
2.
Recordings were made from metathoracic nerve 1 and flight motoneuron (MN) cell bodies during tegula stimulation. It was found that the faster conducting fibre group from the tegula elicited an EPSP in a wing depressor motoneuron (127) and an IPSP in a wing elevator motoneuron (113). The slightly slower conducting fibre group produced the opposite effects, i.e., an IPSP in the depressor (127) and an EPSP in the elevator motoneuron (113). Latency measurements and responses to high frequency stimulation indicated that the excitatory connections are monosynaptic whereas the inhibitory connections are probably mediated by nonspiking interneurons (Figs. 4–7).
-
3.
Stimulation of the hindwing tegula produced PSP's of the same sign in a contralateral motoneuron as in its ipsilateral homologue. The contralaterally evoked EPSP's were mediated by a polysynaptic pathway. These EPSP's often potentiated with repetitive stimulation whereas the ipsilateral responses did not (Fig. 9).
-
4.
Stimulating both hindwing tegulae caused interactions at the motoneurons. Stimulation of the contralateral hindwing tegula potentiated the response of MN 113 to an ipsilateral stimulus given up to 30–40 ms later. Ipsilateral stimulation did not potentiate the contralateral response (Fig. 10). This interaction between the two inputs probably occurs at the tegula-to-motoneuron synapse.
-
5.
Simultaneous stimulation of the fore- and hindwing tegulae at flight frequency could evoke a larger EPSP in MN 113 than when the hindwing alone was stimulated (Fig. 11).
-
6.
These interactions between tegulae result in a considerable amplification of their individual inputs to motoneurons during flight.
Similar content being viewed by others
Abbreviations
- EPSP :
-
excitatory postsynaptic potential
- IPSP :
-
inhibitory postsynaptic potential
- MN :
-
flight motoneuron
References
Altman, J.S.: Changes in the flight motor pattern during development of the Australian plague locust,Choroicetes terminifera. J. Comp. Physiol.97, 127–142 (1975)
Altman, J.S., Tyrer, N.M.: The locust wing hinge stretch receptors I. Primary sensory neurones with enormous central arborisations. J. Comp. Neurol.172, 409–430 (1977)
Altman, J.S., Anselment, E., Kutsch, W.: Postembryonic development of an insect sensory system: ingrowth of axons from hindwing sense organs inLocusta migratoria. Proc. R. Soc. London (Biol.)202, 497–516 (1978)
Altman, J.S., Shaw, M.K., Tyrer, N.M.: Electron microscopy of a cobalt-filled sensory neurone in a locust reveals both pre- and postsynaptic contacts. (in preparation) (1979)
Burrows, M.: Monosynaptic connexions between wing stretch receptors and flight motoneurons of the locust. J. Exp. Biol.62, 189–219 (1975)
Burrows, M.: The influence of sensory inflow on the flight system of the locust. Perspect. Exp. Biol.1, 399–409 (1976)
Campbell, J.I.: The anatomy of the nervous system of the mesothorax ofLocusta migratoria migratorioides R. & F. Proc. Zool. Soc. London137, 403–432 (1961)
Kandel, E.R.: Cellular basis of behavior. San Francisco: Freeman 1976
Kien, J.: Variability of locust motoneuron responses to sensory stimulation: A possible substrate for motor flexibility (in press) (1979a)
Kien, J.: Morphology of locust neck muscle motoneurons and functionally distinct inputs. (submitted) (1979b)
Klein, U.: Zur Funktion und Struktur der Palpen und ihres Sensillenfeldes beiGryllus maculatus de Geer. Doctoral Dissertation, Universität München (1979)
Möhl, B., Nachtigall, W.: Proprioceptive input on the locust flight motor revealed by muscle stimulation. J. Comp. Physiol.128, 57–65 (1978)
Pearson, K.G., Stein, R.B., Malhotra, S.K.: Properties of action potentials from insect motor nerve fibres. J. Exp. Biol.53, 299–316 (1970)
Pearson, K.G. Wong, R.K.S., Fourtner, C.R.: Connexions between hair-plate afferents and motoneurones in the cockroach leg. J. Exp. Biol.64, 251–256 (1976)
Snodgrass, R.E.: The thoracic mechanism of a grasshopper and its antecedents. Smithson. Misc. Collect.82 (2), 1–11 (1929)
Tyrer, N.M., Altman, J.S.: Motor and sensory flight neurones in a locust demonstrated using cobalt chloride. J. Comp. Neurol.157, 117–138 (1974)
Usherwood, P.N.R., Grundfest, H.: Peripheral inhibition in skeletal muscle of insects. J. Neurophysiol.28, 497–518 (1965)
Wendler, G.: The influence of proprioceptive feedback on locust flight co-ordination. J. Comp. Physiol.88, 173–200 (1974)
Wendler, G.: The possible role of fast wing reflexes in locust flight. Naturwissenschaften65, 65 (1978)
Wilson, D.M.: The central nervous control of flight in a locust. J. Exp. Biol.38, 471–490 (1961)
Wilson, D.M., Gettrup, E.: A stretch reflex controlling wingbeat frequency in grasshoppers. J. Exp. Biol.40, 171–185 (1963)
Author information
Authors and Affiliations
Additional information
Supported by DFG grant ‘Neural Mechanisms of Behaviour’ (Hu 35/17).
Supported by the British Science Research Council
Rights and permissions
About this article
Cite this article
Kien, J., Altman, J.S. Connections of the locust wing tegulae with metathoracic flight motoneurons. J. Comp. Physiol. 133, 299–310 (1979). https://doi.org/10.1007/BF00661132
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00661132