Abstract
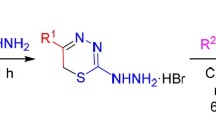
The reaction of dimeric nitroso chlorides of olefins with potassium dithiocarbazate was used to synthesize 3-amino-4-hydroxylaminothiazolidine-2-thiones, which undergo rearrangement to 2-mercapto-1,3,4-thiadiazines on heating and react with butane-2,3-dione to give 2,3-dimethyl-4a,5-dihydro-7-thioxothiazolo[3,4-b]-1,2,4-triazine 4-oxides. The latter are reduced by sodium borohydride to the corresponding 3,4,4a,5-tetrahydro-7-thioxo derivatives.
Similar content being viewed by others
Literature Cited
I. Beger and P. D. Thong, East German Patent No. 127811; Chem. Abstr., 88, 121250 (1978).
T. I. Orlova, S. P. épshtein, V. P. Tashchi, A. F. Rukasov, L. Ya. Bogel'fer, and Yu. G. Putsykin, Khim. Geterotsikl. Soedin., No. 4, 549 (1986).
Author information
Authors and Affiliations
Additional information
Translated from Khimiya Geterotsiklicheskikh Soedinenii, No. 4, pp. 554–556, April, 1987.
Rights and permissions
About this article
Cite this article
épshtein, S.P., Orlova, T.I., Rukasov, A.F. et al. Synthesis of 3-amino-4-hydroxylaminothiazolidine-2-thiones and 2,3-dimethyl-4a,5-dihydro-7-thioxothiazolo[3,4-b]-1,2,4-triazines. Chem Heterocycl Compd 23, 463–464 (1987). https://doi.org/10.1007/BF00546749
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00546749