Summary
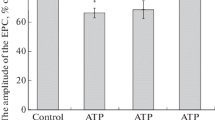
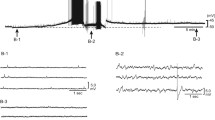
The effect of bath-applied adenosine on transmission in the isolated superior cervical ganglion of the rat was investigated. The compound post ganglionic action potential was recorded as an index of ganglionic transmission. Adenosine and 2-chloroadenosine were equipotent in producing a dose-dependent inhibition of the amplitude of the compound action potential. At the highest concentration tested (1 mM) adenosine and 2-chloroadenosine produced about 30% decrease in the amplitude of the compound action potential. This inhibitory effect was antagonized by theophylline (1 and 100 μM) which by itself had no significant effect on ganglionic transmission. The adenosine uptake blocker dipyridamole (1 and 100 μM) failed to potentiate the inhibitory action of adenosine. Both 4-aminopyridine (20 μM) and high frequencies of stimulation (3, 10 and 20 Hz) were effective in nearly completely abolishing the inhibitory effect of adenosine on ganglionic transmission.
The results suggest that the inhibitory effect of adenosine on ganglionic transmission may be the result of activation of presynaptic adenosine receptors in the ganglion.
Similar content being viewed by others
References
Akasu T, Hirai K, Koketsu K (1983) Modulatory actions of ATP on membrane potential of bullfrog sympathetic ganglion cells. Brain Research 258:313–317
Alkadhi KA, Sabouni M (1983) Effect of adenosine on ganglionic transmission. Fed Proc 42:499
Branisteanu DD, Haulica ID, Nhue BG (1979) Adenosine effects upon transmitter release parameters in the Mg2+-paralyzed neuromuscular junction of frog. Naunyn-Schmiedeberg's Arch Pharmacol 301:273–779
Brown DA, Caulfield MP, Kirby PJ (1979) Relationship between catecholamine-induced cyclic AMP changes and hyperpolarization in isolated rat sympathetic ganglia. J Physiol 290:441–451
Clanachan AS (1979) Modification of release by adenosine and adenine nucleotides. In Paton DM (ed) The release of catecholamines from adrenergic neurons. Pergamon Press, Oxford pp 263–273
Daly JW, Burns RF, Snyder SH (1981) Adenosine receptors in the central nervous system: Relationship to the central actions of methylxanthines. Life Sciences 28:2083–2097
Edstrom JP, Phillis JW (1976) The effects of AMP on the potential of rat cerebral cortical neurons. Can J Physiol Pharmac 54: 787–790
Fredholm BB, Hedqvist P (1980) Modulation of neurotransmission by purine nucleotides and nucleosides. Biochem Pharmac 29:1635–1643
Ginsborg BL, Hirst GDS (1972) The effect of adenosine on the release of the transmitter from the phrenic nerve of the rat. J Physiol 224:629–645
Gustafsson L, Hedqvist P, Fredholm BB (1978) Inhibition of acetylcholine release by adenosine. Acta Physiol Scand 104: 469–478
Harms HH, Wardeh G, Mulder AH (1979) Effects of adenosine on depolarization-induced release of various radiolabelled neurotransmitters from slices of rat corpus striatum. Neuropharmacology 19:577–580
Nayashi E, Kunitomo M, Mori M, Shinozuka K, Yamada S (1978) The development of tachyphylaxis to electrical stimulation in guinea-pig ileal longitudinal muscle and the possible participation of adenosine nucleotides. Br J Pharmac 62:457–464
Henon BK, McAfee DA (1983) The ionic basis of adenosine receptor actions on postganglionic neurones in the rat. J Physiol 336:607–620
Hollins C, Stone TW (1980) Adenosine inhibition of γ-aminobutyric acid release from slices of rat cerebral cortex. Br J Pharmac 69:107–112
Jhamandas K, Sawynok J (1976) Methylxanthine antagonism of opiate and purine effects on the release of acetylcholine. In Kosterlitz HW (ed) Opiates and Endogenous Opioid Peptides. Elsevier-North Holland, Amsterdam pp 161–168
Perkins MN, Stone TW (1980) 4-Aminopyridine blockade of neuronal depressant responses to ATP. Br J Pharmac 70: 425–428
Phillis JW, Wu PH (1982) The effect of various centrally active drugs on adenosine uptake by the central nervous system. Comp Biochem Physiol 77C:179–187
Robeiro JA, Sa-Almeida AM, Namorado JM (1979) Adenosine and adenosine triphosphate decrease 45Ca uptake by synaptosomes stimulated by potassium. Biochem Pharmac 28: 1297–1300
Siggins GR, Gruol DL, Padjen AL, Forman DS (1977) Purine and pyrimidine mononucleotides depolarize neurones of explanted amphibian sympathetic ganglia. Nature 270:263–265
Silinsky EM (1984) On the mechanism by which adenosine receptor activation inhibits the release of acetylcholine from motor nerve ending. J Physiol 346:243–256
Stone TW (1981) Physiological roles for adenosine and adenosine 5′-triphosphate in the nervous system. Neuroscience 6: 523–555
Thesleff S (1980) Aminopyridines and synaptic transmission. Neuroscience 5:1413–1419
Vizi ES (1979) Presynaptic modulation of neurochemical transmission. Progress in Neurobiol 12:181–290
Wu PH, Phillis JW, Thierry DL (1982) Adenosine receptor agonists inhibit K+-evoked Ca2+ uptake by rat brain cortical synaptosomes. J Neurochem 39:700–708
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Alkadhi, K.A., Brown, T.R. & Sabouni, M.H. Inhibitory effect of adenosine on transmission in sympathetic ganglia. Naunyn-Schmiedeberg's Arch. Pharmacol. 328, 16–19 (1984). https://doi.org/10.1007/BF00496098
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00496098