Abstract
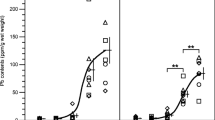
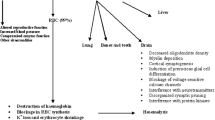
The urinary excretion of δ-aminolevulinic acid (ALA) and coproporphyrin (CP) isomers (I and III) was investigated in mice exposed to lead in drinking water (200 and 500 ppm) for 14 or 30 days. Furthermore, the inhibition of pyrimidine 5′-nucleotidase (P5N) and δ-aminolevulinic acid dehydratase (ALAD) was studied in the same mice. In the lead-exposed mice, urinary excretion of ALA was significantly elevated (12-times control), while that of CP remained unchanged. This discrepancy on the urinary excretion may be of interest. The P5N activity in both erythrocytes and bone marrow cells was significantly inhibited by exposure to lead. However, the degree of inhibition was greater in the erythrocytes (45% inhibition) than in the bone marrow cells (25% inhibition). The inhibition of ALAD was much higher in erythrocytes (90% inhibition) than in liver (20–40% inhibition). The increase in erythrocyte protoporphyrin and the decrease in both hemoglobin and hematocrit were not observed in mice exposed to lead under the above conditions.
Similar content being viewed by others
References
Alessio L, Bertazzi PA, Monelli O, Foa V (1976) Free erythrocyte protoporphyrin as an indicator of the biological effect of lead in adult males. II. Comparison between erythrocyte protoporphyrin and other indicators of effect. Int Arch Occup Environ Health 37: 89–105
Angle C, McIntire MS (1978) Low level lead and inhibition of erythrocyte pyrimidine nucleotidase. Environ Res 17: 296–302
Buc HA, Kaplan JC (1978) Red-cell pyrimidine 5′-nucleotidase and lead poisoning. Clin Chim Acta 87: 49–55
Cook LR, Angle CR, Kubitschek CE, Stohs SJ (1987) Prediction of blood lead by HPLC assay of erythrocyte pyrimidine 5′-nucleotidase. J Anal Toxicol 11: 39–42
Dieter MP, Finley MT (1979) Delta-aminolevulinic acid dehydratase enzyme activity in blood, brain, and liver of lead-dosed ducks. Environ Res 19: 127–135
Hammond PB (1977) Exposure of humans to lead. Ann Rev Pharmacol Toxicol 17: 197–214
Horiguchi S, Teramoto K, Kiyota I, Shinagawa K, Nakano H, Karai I, Matsuda F (1981) Relationships among the parameters of lead absorption and lead effects especially on the hematopoetic system. Osaka City Med J 27: 35–45
Ichiba M, Tomokuni K, Sugimoto K (1987) Erythrocyte pyrimidine 5′-nucleotidase test for occupational lead exposure. Ind Health 25: 195–204
Kondo M, Shimizu Y (1986) The effects of ethanol, estrogen, and hexachlorobenzene on the activities of hepatic δ-aminolevulinate synthetase, δ-aminolevulinate dehydratase, and uroporphyrinogen decarboxylase in male rats. Arch Toxicol 59: 141–145
Lowry OH, Rosebrough NJ, Farr AL, Randall RJ (1951) Protein measurement with the Folin-phenol reagent. J Biol Chem 193: 265–271
Mohammed-Brahim B, Buchet JP, Lauwerys R (1985) Erythrocyte pyrimidine 5′-nucleotidase activity in workers exposed to lead, mercury or cadmium. Int Arch Occup Environ Health 55: 247–252
Nikkanen J, Hernberg S, Tola S (1972) Modifications of the deltaaminolevulinic acid dehydratase test and their significance for assessing different intensities of lead exposure. Work Environ Health 9: 46–52
Ohmori S, Harada K, Miura H (1986) Behavior of biological parameters for lead exposure in Japanese male workers. I. Actual levels of parameters in different lead exposure. Kumamoto Med J 39: 187–199
Paglia DE, Valentine WN, Dahlgren JG (1975) Effects of low-level lead exposure on pyrimidine 5′-nucleotidase and other erythrocyte enzymes. Possible role of pyrimidine 5′-nucleotidase in the pathogenesis of lead-induced anemia. J Clin Invest 56: 1164–1169
Piomelli S (1973) A micromethod for erythrocyte porphyrins; The FEP test. J Lab Clin Med 81: 932–940
Sakai T, Ushio K (1986) A simplified method for determining erythrocyte pyrimidine 5′-nucleotidase (P5N) activity by HPLC and its value in monitoring lead exposure. Br J Ind Med 43: 839–844
Sassa S (1978) Toxic effects of lead, with particular reference to porphyrin and heme metabolism. In: Handbook of experimental pharmacology, Vol 44. Springer-Verlag, Berlin
Sato Y, Sasaki T, Taniguchi N, Saito K (1981) Normal pyrimidine 5′-nucleotidase activity level of Japanese subjects and a significance as a marker for low concentration of lead in blood (in Japanese). Jpn J Hyg 36: 518–525
Tola S, Hernberg S, Asp S, Nikkanen J (1973) Parameters indicative of absorption and biological effect in new lead exposure. A prospective study. Br J Ind Med 30: 134–141
Tomokuni K, Ogata M (1976) Relationship between lead concentration in blood and biological response for porphyrin metabolism in workers occupationally exposed to lead. Arch Toxicol 35: 239–246
Tomokuni K, Hirai Y (1986) Simple liquid-chromatographic determination of urinary coproporphyrin in workers exposed to lead. Clin Chem 32: 872–873
Tomokuni K, Ichiba M (1986) Simple determination of erythrocyte pyrimidine 5′-nucleotidase activity in human blood by high-performance liquid chromatography. Ind Health 24: 227–233
Tomokuni K, Ichiba M, Hirai Y, Hasegawa T (1987) Optimized liquid-chromatographic method for fluorometric determination of urinary δ-aminolevulinic acid in workers exposed to lead. Clin Chem 33: 1665–1667
Tomokuni K, Ichiba M, Hirai Y (1988a) Species difference of urinary excretion of δ-aminolevulinic acid and coproporphyrin in mice and rats exposed to lead. Toxicol Lett 41: 255–259
Tomokuni K, Ichiba M, Hirai Y, Hasegawa T, Sugimoto K (1988b) Relationship between inhibition of erythrocyte pyrimidine 5′-nucleotidase activity and biological response for porphyrin metabolism in workers occupationally exposed to lead. Int Arch Occup Environ Health 60: 431–436
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Tomokuni, K., Ichiba, M. & Hirai, Y. Effect of lead exposure on some biological indices related to porphyrin metabolism and the activity of erythrocyte pyrimidine 5′-nucleotidase in the mice. Arch Toxicol 63, 23–28 (1989). https://doi.org/10.1007/BF00334629
Received:
Revised:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00334629