Summary
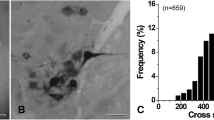
Using the method of transganglionic transport of horseradish peroxidase (HRP) according to Mesulam (1978), the course and central terminations of the afferents from the longus capitis muscle were studied in the rat. Application of HRP to the cut muscle nerves was followed by heavy labeling of a considerable number of cell bodies of different sizes in the spinal ganglia C2 and C3. The labeled central processes follow two distinct main routes: one to the external cuneate nucleus, which is known to project ipsilaterally to the cerebellum, the other to the central cervical nucleus (CCN) of the spinal cord. The latter nucleus contains dense arborizations and terminals of muscle afferents in close relationship to medium sized cells which project contralaterally to the cerebellum. This could be shown in double labeling experiments with HRP as a tracer for primary afferents and Nuclear yellow for the cerebellar pathway. The labeled area of CCN extends from the fourth cervical segment up to the medulla oblongata where it lies laterally adjacent to the hypoglossal nucleus, though clearly separated from the latter.
In the cervical part of CCN, dendrites of neck muscle motoneurons arborize within the area of afferent terminals.
Besides the external cuneate and the central cervical nuclei, afferent projections were seen in lamina X, partly in close contact with the central canal, and in more lateral areas of lamina VII. In contrast to results from studies on other neck muscles, no reaction product was observed in dorsal horn laminae I–VI.
Similar content being viewed by others
References
Abrahams VC (1981) Sensory and motor specialization in some muscles of the neck. Trends in Neuroscience 4:24–27
Brodal A (1941) Die Verbindungen des Nucleus cuneatus externus mit dem Kleinhirn beim Kaninchen und bei der Katze. Experimentelle Untersuchungen. Z ges Neurol Psychiat 171:167–199
Brodal A (1975) Neurological anatomy in relation to clinical medicine. 2nd ed. The Oxford University Press, London, Toronto
Cervero F, Iggo A, Ogawa H (1976) Nociceptor-driven dorsal horn neurones in the lumbar spinal cord of the cat. Pain 2:5–24
Cooke JD, Larson B, Oscarsson O, Sjölund B (1971) Origin and termination of cuneocerebellar tract. Exp Brain Res 13:339–358
Corbin KB, Hinsey JC (1935) Intramedullary course of the dorsal root fibers of each of the first four cervical nerves. J Comp Neurol 63:119–126
Cummings JF, Petras JM (1977) The origin of spinocerebellar pathways. 1. The nucleus cervicalis centralis of the cranial cervical spinal cord. J Comp Neurol 173:655–692
Eccles JC, Fatt P, Landgren S, Winsbury GJ (1954) Spinal cord potentials, generated by volleys in the large muscle afferents. J Physiol Lond 125:590–606
Ferraro A, Barrera SE (1935) The nuclei of the posterior funiculi in Macacus rhesus. An anatomic and experimental investigation. Arch Neurol Psychiat (Chicago) 33:262–275
Gottschall J, Neuhuber W, Müntener M, Mysicka A (1980) The ansa Embryol 159:59–69
Gottschall J, Zenker W, Neuhuber W, Mysicka A, Müntener M (1980) The sternomastoid muscle of the rat and its innervation. Muscle fiber composition, perikarya and axons of efferent and afferent neurons. Anat Embryol 160:285–300
Grant G (1962) Projection of the external cuneate nucleus onto the cerebellum in the cat: An experimental study using silver methods. Exp Neurol 5:179–195
Grant G, Arvidsson J, Robertson B, Ygge J (1979) Transganglionic transport of horseradish peroxidase in primary sensory neurons. Neuroscience Letters 12:23–28
Heckmann Th, Bourassa ChM (1981) Lesions of the dorsal column nuclei or medial lemniscus of the cat: effect on motor performance. Brain Res 224:405–411
Hirai N, Hongo T, Sasaki S (1978) Cerebellar projection and input organizations of the spinocerebellar tract arising from the central cervical nucleus of the cat. Brain Res 157:341–345
Ishizuka N, Mannen H, Hongo T, Sasaki S (1979) Trajectory of group Ia afferent fibers stained with horseradish peroxidase in the lumbosacral cord of the cat: three dimensional reconstructions from serial sections. J Comp Neurol 186:189–212
Hohnson JL jr, Welker WI, Pubols BH jr (1968) Somatotopic organization of raccoon dorsal column nuclei. J Comp Neurol 132:1–44
Katan S, Gottschall J, Neuhuber W (1982) Simultaneous visualization of horseradish peroxidase and nuclear yellow in tissue sections for neuronal double labeling. Neuroscience Lett 28:121–126
Kerr FWL (1961) Structural relation of the trigeminal spinal tract to upper cervical roots and the solitary nucleus of the cat. Exp Neurol 4:134–148
Liu CN (1956) Afferent nerves to Clarke's and lateral cuneate nuclei in the cat. Arch Neurol Psychiat (Chicago) 75:67–77
Lucas E, Willis WD (1974) Identification of muscle afferents which activate interneurons in the intermediate nucleus. J Neurophysiol 37:282–293
Matsushita M, Ikeda M (1975) The central cervical nucleus as cell origin of a spinocerebellar tract arising from the cervical cord: a study in the cat using horseradish peroxidase. Brain Res 100:412–419
Matsushita M, Hosoya Y (1979) Cells of origin of the spinocerebellar tract in the rat, studied with the method of retrograde transport of horseradish peroxidase. Brain Res 173:185–200
Mesulam MM (1978) Tetramethylbenzidine for horseradish peroxidase neurohistochemistry: a non-carcinogenic blue reaction product with superior sensitivity for visualizing neural afferents and efferents. J Histochem Cytochem 26:106–117
Mesulam MM, Brushart TM (1979) Transganglionic and anterograde transport of horseradish peroxidase across dorsal root ganglia: A tetramethylbenzidine method for tracing central sensory connections of muscles and peripheral nerves. Neuroscience 4:1107–1117
Müntener M, Gottschall J, Neuhuber W, Mysicka A (1980) The ansa cervicalis and the infrahyoid muscles of the rat. I. Anatomy; distribution, number and diameter of fiber types; motor units. Anat Embryol 159:49–57
Mysicka A, Zenker W (1981) Central projections of muscle afferents from the sternomastoid nerve in the rat. Brain Res 211:257–265
Oscarsson O (1965) Functional organization of the spino- and cuneocerebellar tracts. Physiol Rev 45:495–522
Petras JM, Cummings JF (1975) Cervical spinocerebellar projections in the dog. Anat Rec 181:448
Ramon y Cajal S (1909) Histologie du système nerveux de l'homme et des vertébrés. Vol I, Maloine, Paris
Ranson SW, Davenport HK, Doles EA (1932) Intramedullary course of the dorsal root fibers of the first three cervical nerves. J Comp Neurol 54:1–12
Rexed B (1952) The cytoarchitectonic organization of the spinal cord in the cat. J comp Neurol 96:415–495
Rexed B (1954) A cytoarchitectonic atlas of the spinal cord of the cat. J Comp Neurol 100:297–378
Richmond FJR, Scott DA, Abrahams VC (1978) Distribution of motoneurons to neck muscles biventer cervicis, splenius and complexus in cat. J Comp Neurol 181:451–463
Rinvik E, Walberg F (1975) Studies on the cerebellar projections from the main and external cuneate nuclei in the cat by means of retrograde axonal transport of horseradish peroxidase. Brain Res 95:371–381
Rosén I (1969) Localization in caudal brain stem and cervical spinal cord of neurones activated from forelimb group I afferents in the cat. Brain Res 16:55–71
Shriver JE, Stein BM, Carpenter MB (1968) Central projections of spinal dorsal roots in monkey. I. Cervical and upper thoracic dorsal roots. Am J Anat 123:27–74
Stacey MJ (1969) Free endings on skeletal muscle of the cat. J Anat (London) 105:231–254
Stillhard G (1981) Musculi longus capitis et splenius der Ratte und innervierende Motoneurone. Acta Neuropathol 53:267–274
Wiksten B (1975) The central cervical nucleus — a source of spinocerebellar fibers, demonstrated by retrograde transport of horseradish peroxidase. Neuroscience Lett 1:81–84
Wiksten B (1979) The central cervical nucleus in the cat. I. A Golgi study. Exp Brain Res 36:143–154
Wiksten B (1979) The central cervical nucleus in the cat. II. The cerebellar connections studied with retrograde transport of horseradish peroxidase. Exp Brain Res 36:155–173
Wiksten B (1979) The central cervical nucleus in the cat. III. The cerebellar connections studied with anterograde transport of 3H-Leucine. Exp Brain Res 36:175–189
Willis WD, Coggeshall RE (1975) Sensory mechanisms of the spinal cord. New York London
Yee J, Corbin KB (1939) The intramedullary course of the upper five, cervical dorsal root fibers in the rabbit. J Comp Neurol 70:297–304
Zenker W, Mysicka A, Neuhuber W (1980) Dynamics of the transganglionic movement of horseradish peroxidase in primary sensory neurons. Cell Tissue Res 207:479–489
Author information
Authors and Affiliations
Additional information
This investigation was supported by the EMDO-Stiftung Zürich
Rights and permissions
About this article
Cite this article
Ammann, B., Gottschall, J. & Zenker, W. Afferent projections from the rat longus capitis muscle studied by transganglionic transport of HRP. Anat Embryol 166, 275–289 (1983). https://doi.org/10.1007/BF00305088
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00305088