Summary
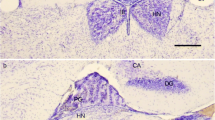
Although some embryological and morphological features speak in favour of a neuronal character of rodent pinealocytes, histochemistry and ultrastructure let this issue appear controversial. Using antibodies to different neurofilaments, the neural adhesion molecule L1, synaptophysin and tubulin as neuronal markers, the pineal glands of rat and guinea-pig were studied by means of immunfluorescence. Neurofilament-immunoreactivity was present in some rat pineal nerve fibers and in the majority of guinea-pig pinealocytes, L1 decorated rat intrapineal nerve fibers, synaptophysin was almost ubiquitously distributed in the pineal of both species, while tubulin-immunofluorescence was seen in nerve fibers of rat and guinea-pig pineal and in some pinealocytes of the latter. These findings speak in favour of the neuronal character of guinea-pig pinealocytes. The lack of neurofilament- and tubulin-immunoreactivity in rat pinealocytes might be attributable to very low concentrations of these proteins or species differences as to their expression. Further studies including in situhybridisation of relevant mRNAs will be necessary to answer these questions definitely.
Similar content being viewed by others
References
Ariëns Kappers J (1960) The development, topographical relations and innervation of the epiphysis cerebri in the albino rat. Z Zellforsch Mikrosk Anat 52:163–215
Bowers CW, Dahm LM, Zigmond RE (1984) The number and distribution of sympathetic neurons that innervate the rat pineal gland. Neuroscience 13:87–96
Cardinali DP, Freire F (1975) Melatonin effects on brain: Interaction with microtubule protein, inhibition of fast axoplasmic flow and induction of crystaloid and tubular formations in the hypothalamus. Mol Cell Endocrinol 2:317–330
Cumming R, Burgoyne RD, Lytton NA (1983) Axonal sub-populations in the central nervous system demonstrated using monoclonal antibodies against α-tubulin. Eur J Cell Biol 31:241–248
Dahl D, Bignami A (1977) Preparation of antisera to neurofilament protein from chicken brain and human sciatic nerve. J Comp Neurol 176:645–657
Dahl D, Bignami A (1985) Intermediate filaments in nervous tissue. In: Shaw JW, (ed) Cell and muscle motility, vol. 6. Plenum Publishing Corporation, New York, pp 75–96
Dahl D, Grossi M, Bignami A (1984) Masking of epitopes in tissue sections. A study of glial fibrillary acidic (GFA) protein with antisera and monoclonal antibodies. Histochemistry 81:525–531
Dahl D, Crosby CJ, Bignami A (1986) Neurofilament proteins in fish: a study with monoclonal antibodies reacting with mammalian NF 150K and NF 200K. J Comp Neurol 250:399–402
Debus E, Weber K, Osborn M (1983) Monoclonal antibodies specific for glial fibrillary acidic (GFA) protein and for each of the neurofilament triplet polypeptides. Differentiation 25:193–203
Draeger UC, Edwards DL, Kleinschmidt J (1983) Neurofilaments contain α-melanocyte-stimulating hormone (α-MSH)-like immunoreactivity. Proc Natl Acad Sci USA 80:6408–6412
Freire F, Cardinali DP (1975) Effects of melatonin treatment and environmental lighting on the ultrastructural appearance, melatonin synthesis, norepinephrine turnover and microtubule protein content of the rat pineal gland. J Neural Transm 37:237–257
Gozes I (1982) Tubulin in the nervous system. Neurochem Int 4:101–120
Groeschel-Stewart U, Mayer VW, Taylor-Mayer RE, Zimmermann FK (1985) Aprotic polar solvents inducing chromosomal malsegregation in yeast interfere with the assembly of porcine brain tubulin in vitro. Mutation Res 149:333–338
Hickey WF, Lee V, Trojanowski JQ, McMillan LJ, McKearn T, Gonatas J, Gonatas NK (1983) Immunohistochemical application of monoclonal antibodies against myelin basic protein subsunits; advantages over antisera and technical limitations. J Histochem Cytochem 31:1126–1135
Korf H-W, Møller M (1984) The innervation of the mammalian pineal gland with special reference to central pinealopetal projections. In: Reiter RJ (ed) Pineal research reviews 2. Alan R. Liss, New York, pp 41–86
Liesi P, Julien J-P, Vilja P, Grosveld F, Rechardt L (1986) Specific detection of neuronal cell bodies: In situ hybridization with a biotin-labeled neurofilament cDNA probe. J Histochem Cytochem 34:923–926
Martini R, Schachner M (1986) Immunoelectron microscopic localization of neural cell adhesion molecules (L1, N-CAM, and MAG) and their shared carbohydrate epitope and myelin basic protein in developing sciatic nerve. J Cell Biol 103:2439–2448
Peters A, Palay SL, Webster H DeF (1976) The fine structure of the nervous system: The neuron and supporting cells. Sanders, Philadelphia, USA
Rathjen FG, Schachner M (1984) Immunocytochemical and biochemical characterization of a new neuronal cell surface component (L1 antigen) which is involved in cell adhesion. EMBO J 3:1–10
Reuss S (1987) Electrical activity of the mammalian pineal gland. In: Reiter RJ (ed) Pineal research reviews 5. Alan R. Liss, New York, pp 153–190
Schröder H (1986) Immunofluorescence assays of catecholamines and related compounds. In: Krstulovic A (ed) Quantitative analysis of catecholamines and related compounds. Horwood, Chichester, pp 316–339
Schröder H (1987) Aminergic innervation pattern of the rodent pineal gland no apparent influence of time of day. Acta Anat 129:22–26
Schröder H, Vollrath L (1985) Distribution of dopamine-beta-hydroxylase-like immunoreactivity in the rat pineal organ. Histochemistry 83:375–380
Schröder H, Fujisawa H, Vollrath L (1987) Immunohistochemical characterization of rat pineal dopamine-beta-hydroxylase (DBH)-containing structures: use of a homologous monoclonal antibody. J Pineal Res 4:221–232
Schröder H, Weihe E, Nohr D, Vollrath L (1988) Immunohistochemical evidence for the presence of peptides derived from proenkephalin, prodynorphin and proopiomelanocortin in the guinea-pig pineal gland. Histochemistry 88:333–341
Shiotani Y, Yamano M, Shiosaka S, Emson PC, Hillyard CJ, Girgis S, Macintyre I (1986) Distribution and origins of substance P (SP)-, calcitonin gene-related peptide (CGRP)-, vasoactive intestinal polypeptide (VIP)-, and neuropeptide Y (NPY)-containing nerve fibers in the pineal gland of gerbils. Neurosci Lett 70:187–192
Vollrath L (1981) The pineal organ. In: Oksche A, Vollrath L (eds) Handbuch der mikroskopischen Anatomie des Menschen, VI/7. Springer, Berlin Heidelberg New York, pp 1–665
Vollrath L, Schröder H (1987) Neuronal properties of mammalian pinealocytes? In: Trentini GP, De Gaetani C, Pévet P (eds) Fundamentals and clinics in pineal research. Raven Press, New York, pp 13–23
Wiche G, Briones E, Koszka Ch, Artlieb U, Krepler R (1984) Widespread occurence of polypeptides related to neurotubule-associated proteins (MAP-1 and MAP-2) in non-neuronal cells and tissues. EMBO J 3:991–998
Wiedenmann B, Franke WW (1985) Identification and localization of synaptophysin, an integral membrane glycoprotein of Mr 38000 characteristic of presynaptic vesicles. Cell 41:1017–1028
Author information
Authors and Affiliations
Additional information
Supported by the Deutsche Forschungsgemeinschaft, grant Schr 283/2-1
Rights and permissions
About this article
Cite this article
Schröder, H., Bendig, A., Dahl, D. et al. Neuronal markers in the rodent pineal gland — an immunohistochemical investigation. Histochemistry 94, 309–314 (1990). https://doi.org/10.1007/BF00266633
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00266633