Abstract
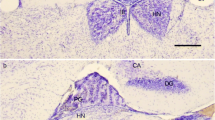
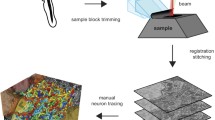
The morphology of the deep pineal gland of the Sprague Dawley rat was investigated by serial block face scanning electron microscopy. Cells were three-dimensionally (3-D) reconstructed using the software Fiji TrackEM. The deep pineal gland consisted of 2–5 layers of electron-lucent pinealocytes, with a euchromatic nucleus, endowed with one or two processes. Laterally, the deep pineal merged with the habenula and the stria medullaris thalami, via an intermediate area containing cells with more electron-dense cytoplasm and an indented nucleus with heterochromatin. Neither nerve terminals nor capillaries were observed in the deep pineal itself but present in the intermediate parts of the gland. The deep pineal was in contact with the third ventricle via the pineal and suprahabenular recesses. The ependymal lining in these recesses was an epithelium connected by tight junctions between their lateral cell membranes. Several intraventricular nerve terminals were in contact with the ependyma. 3-D reconstructions showed the ependymal cells endowed with long slender process penetrating the underlying pineal parenchyma. Few “tanocyte-like” ependymal cells, endowed with a process, reaching the subarachnoid space on the inferior surface of the deep pineal were observed. In addition, pinealocyte and astrocyte processes, often connected by gap junctions, bordered the inferior surface. In summary, the rat deep pineal gland is a neuroendocrine structure connected to the habenula. We here report specialized ependymal cells that might transmit signals from the cerebrospinal fluid to the deep pineal parenchyma and a “trans-pineal tanocyte-like cell” that connects the ventricular system with the subarachnoid space.









Similar content being viewed by others
References
Björklund A, Owman Ch, West KA (1972) Peripheral sympathetic innervation and serotonin cells in the habenular region of the rat brain. Z Zellforsch 127:570–579
Boeckmann D (1980) Morphological investigation of the deep pineal of the rat. Cell Tissue Res 210:283–294. https://doi.org/10.1007/BF00237616 (PMID: 7407872)
Brednow K, Korf HW (1998) Morphological and immunocytochemical features of the pineal organ of C3H and C57BL mice at different stages of postnatal development. Cell Tissue Res 292:521–530. https://doi.org/10.1007/s004410051081 (PMID: 9582409)
Bucana CD, Nadakavukaren MJ, Frehn JL (1971) Annulate lamellae in hamster pineal gland. Tissue Cell 3:405–411. https://doi.org/10.1016/s0040-8166(71)80042-3 (PMID: 18631562)
Dafny N (1983) Evidence that the rat pineal has neuronal connections via the pineal stalk. Exp Neurol 79:858-861. https://doi.org/10.1016/0014-4886(83)90048-1
David GFX, Herbert J (1973) Experimental evidence for a synaptic connection between habenula and pineal ganglion in the ferret. Brain Res 64:327–343
Deerinck TJ, Bushong EA, Thor A, Ellisman MH (2010) NCMIR methods for 3D EM: a new protocol for preparation of biological specimens for serial blockface scanning electron microscopy. Center for Research in Biological Systems and the National Center for Microscopy and Imaging Research, University of California, San Diego, La Jolla, CA, USA https://ncmir.ucsd.edu/sbem-protocol
Denk W, Horstmann H (2004) Serial block-face scanning electron microscopy to reconstruct three-dimensional tissue nanostructure. PLoS Biol 2:e329. https://doi.org/10.1371/journal.pbio.0020329
Del Río-Hortega P (1922) Constitución histologica de la glandula pineal. Arch Neurol 3:359–389
Falck B, Hillarp N-Å, Thieme G, Torp A (1962) Fluoresence of catecholamines and related compounds with formaldehyde. J Histochem Cytochem 10:348–354. https://doi.org/10.1177/10.3.348
Haldar-Misra C, Pévet P (1983) Influence of melatonin on the process of protein and/or peptide secretion in the pineal gland of the rat and hamster. An in vitro study. Cell Tissue Res 231:73–82. https://doi.org/10.1007/BF00215775
Hewing M (1978) A liquor contacting area in the pineal recess of the golden hamster (Mesocricetus auratus). Anat Embryol (berl) 153:295–304. https://doi.org/10.1007/BF00315932
Hewing M (1980) Cerebrospinal fluid-contacting area in the pineal recess of the vole (Microtus agrestis), guinea pig (Cauia cobaya), and rhesus monkey (Macaca mulatta). Cell Tissue Res 209:473–484. https://doi.org/10.1007/BF00234759
Hewing M (1982) Pinealocytes contacting the cerebrospinal fluid of the suprapineal recess in the Mongolian gerbil (Meriones unguiculatus). Cell Tissue Res 222:177–185. https://doi.org/10.1007/BF00218298
Jastrow H, Von Mach M-A, Vollrath L (1997) The shape of synaptic ribbons in the rat pineal gland. Cell Tissue Res 287:255–261. https://doi.org/10.1007/s004410050750
Juillard M-T (1979) The proteinaceous content and possible physiological significance of dense-cored vesicles in hamster and mouse pinealocytes. Ann Biol Anim Biochim Biophys 19:413–428
Kappers JA (1960) The development, topographical relations and innervation of the epiphysis cerebri in the albino rat. Zeitschrift ZellforschMikrosk Anat 52:163–221. https://doi.org/10.1007/BF00338980
Karasek M (1992) Ultrastructure of the mammalian pinealocyte under natural and experimental conditions: quantitative aspects. Micr Res Tech 21:116–123. https://doi.org/10.1002/jemt.1070210204
Karlsen AS, Rath MF, Rohde K, Toft T, Møller M (2013) Developmental and diurnal expression of the synaptosomal-associated protein 25 (Snap25) in the rat pineal gland. Neurochem Res 38:1219–1228. https://doi.org/10.1007/s11064-012-0918-7
King TS, Dougherty WJ (1982) Effect of denervation on “synaptic” ribbon populations in the rat pineal gland. J Neurocytol 1:19–22. https://doi.org/10.1007/bf01258002
Klein DC, Coon SL, Roseboom PH, Weller JL, Bernard M, Gastel JA, Zatz M, Iuvone PM, Rodriguez IR, Begay V, Falcon J, Cahill GM, Cassone VM, Baler R (1997) The melatonin rhythm generating enzyme: molecular regulation of serotonin N-acetyltransferase in the pineal gland. Recent Prog Horm Res 52:307–357 (PMID: 9238858)
Klein D, Sugden D, Weller JL (1983) Postsynaptic a-adrenergic receptors potentiate the padrenergic stimulation of pineal serotonin N-acetyltransferase Proc Natl Acad Sci USA 80599–80603 https://doi.org/10.1073/pnas.80.2.599
Korf HW, Wagner U (1980) Evidence for a nervous connection between the brain and the pineal organ in the guinea pig. Cell Tissue Res 209:505–510. https://doi.org/10.1007/BF00234762
Korf HW, Møller M (2021) The median eminence and infundibular nucleus; window for endocrine and metabolic communication. Handbook of clinical neurology, Vol.180 (3rd series). The human hypothalamus: middle and posterior region (DF Swaab, PJ Lucassen, F Krei, R Buijs, and A Saleh, Editors), pp. 227–251 Elsevier, BV
Korf HW, Oksche A, Ekström P, Gery I, Zigler JS Jr (1986) Klein DC (1986) Pinealocyte projections into the mammalian brain revealed with S-antigen antiserum. Science 231:735–737. https://doi.org/10.1126/science.3454660
Korf HW, Sato T, Oksche A (1990) Complex relationships between the pineal organ and the medial habenular nucleus-pretectal region of the mouse as revealed by S-antigen immunocytochemistry. Cell Tissue Res 261:493–500. https://doi.org/10.1007/BF00313528
Larsen PJ, Enquist LW, Card JP (1998) Characterization of the multisynaptic neuronal control of the rat pineal gland using viral transneuronal tracing. Eur J Neurosci 10:128–145. https://doi.org/10.1046/j.1460-9568.1998.00003.x
Liu W, Møller M (2000) Innervation of the rat pineal gland by PACAP-immunoreactive nerve fibers originating in the trigeminal ganglion: a degeneration study. Cell Tissue Res 301:369–373. https://doi.org/10.1007/s004410000251
Matsushima S, Sakai Y, Hira Y (1999) Peptidergic peripheral nervous systems in the mammalian pineal gland. Microsc Res Tech 46:265–280. https://doi.org/10.1002/(SICI)1097-0029(19990815/01)46:4/5%3c265::AID-JEMT4%3e3.0.CO;2-S
Mcnulty JA, Fox LM (1992) Pinealocyte synaptic ribbons and neuroendocrine function. Microsc Res Techn 21:175–187. https://doi.org/10.1002/jemt.1070210302
Mikkelsen JD, Møller M (1999) Neuropeptide Y in the mammalian Pineal gland. Microsc Res Techniq 46:239–256. https://doi.org/10.1002/(SICI)1097-0029(19990815/01)46:4/5%3c239::AID-JEMT2%3e3.0.CO;2-2
Moore RY (1975) Indolamine metabolism in the intact and denervated pineal, pineal stalk and habenula. Neuroendocrinol 19:323–330. https://doi.org/10.1159/000122453 (PMID: 1241605)
Møller M (1981) The ultrastructure of the deep pineal gland of the Mongolian gerbil and mouse: granular vesicle localization and innervation. In: Matthews CD, RF seemark, (eds) Pineal Function. Elsevier, Amsterdam, pp 257–266
Møller M (1992) Fine structure of the pinealopetal innervation of the mammalian pineal gland. Microsc Res Techn 21:188–204. https://doi.org/10.1002/jemt.1070210303
Møller M (1999) Introduction to mammalian pineal innervation. Microsc Res Techniq 46:235–238. https://doi.org/10.1002/(SICI)1097-0029(19990815/01)46:4/5%3c235::AID-JEMT1%3e3.0.CO;2-9
Møller M, Korf HW (1983a) The origin of central pinealopetal nerve fibers in the Mongolian gerbil as demonstrated by the retrograde transport of horseradish peroxidase. Cell Tissue Res 230:273–287. https://doi.org/10.1007/bf00213805
Møller M, Korf HW (1983b) Central innervation of the pineal organ of the Mongolian gerbil. A histochemical and lesion study. Cell Tissue Res 230:259–272. https://doi.org/10.1007/BF00213804
Møller M, Liu W (1999) Innervation of the rat pineal gland by nerve fibres originating in the sphenopalatine, otic and trigeminal ganglia. A retrograde in vivo neuronal tracing study. Reprod Nutr Dev 39:345–353. https://doi.org/10.1051/rnd:19990307
Møller M, Baeres FMM (2002) The anatomy and innervation of the mammalian pineal gland. Cell Tissue Res 309:139–150. https://doi.org/10.1007/s00441-002-0580-5
Møller M, Cozzi B, Schröder H, Mikkelsen JD (1987) The peptidergic innervation of the mammalian pineal gland. In: Fundamentals and clinics in pineal research (GP Trentini, C de Gaetani, P Pévet, eds.). Serono Symposia Publication nr. 44, pp. 71 77. New York: Raven Press 1987
Olcese J (1991) Neuropeptide Y: an endogenous inhibitor of norepinephrine-stimulated melatonin secretion in the rat pineal gland. J Neurochem 57:943–947. https://doi.org/10.1111/j.1471-4159.1991.tb08241.x
Phansuwan-Pujito P, Møller M, Govitrapong P (1999) Cholinergic innervation and function in the mammalian pineal gland. Microsc Res Tech 46:281–195. https://doi.org/10.1002/(SICI)1097-0029(19990815/01)46:4/5%3c281::AID-JEMT5%3e3.0.CO;2-N
Rath MF, Coon SL, Amaral FG, Weller JL, Møller M, Klein DC (2016) Melatonin synthesis: acetylserotonin o-methyltransferase (ASMT) is strongly expressed in a subpopulation of pinealocytes in the male rat pineal gland. Endocrinol 157:2028–2040. https://doi.org/10.1210/en.2015-1888
Rath MF, Muñoz E, Ganguly S, Morin F, Shi Q, Klein DC, Møller M (2006) Expression of the Otx2 homeobox gene in the developing mammalian brain: embryonic and adult expression in the pineal gland. J Neurochem 97:556–566. https://doi.org/10.1111/j.1471-4159.2006.03773.x
Rath MF, Rohde K, Klein DC, Møller M (2013) Homeobox genes in the rodent pineal gland: roles in development and phenotype maintenance. Neurochem Res 3:1100–1111. https://doi.org/10.1007/s11064-012-0906-y
Rawe VY, Olmedo SB, Nodar FN, Ponzio R, Sutovsky P (2003) Abnormal assembly of annulate lamellae and nuclear pore complexes coincides with fertilization arrest at the pronuclear stage of human zygotic development. Hum Reprod 28:576–582. https://doi.org/10.1093/humrep/deg114 (PMID: 12615828)
Reuss S (1999) Trigeminal innervation of the mammalian pineal gland. Microsc Res Tech 46:305–309. https://doi.org/10.1002/(SICI)1097-0029(19990815/01)46:4/5%3c305::AID-JEMT7%3e3.0.CO;2-#
Ribas, JL (1977) The Rat Epithalamus I. Correlative Scanning-Transmission Electron Microscopy of Supraependymal Nerves. Cell Tiss. Res. 182, 1-16. https://doi.org/10.1016/0006-8993(77)90631-x
Ribelayga C, Gauer F, Pévet P, Simonneaux V (1998) Distribution of hydroxyindole-O-methyltransferase mRNA in the rat brain: an in situ hybridisation study. Cell Tissue Res 291:415–421.
Romijn HJ (1975) Structure and innervation of the pineal gland of the rabbit, Oryctolagus cuniculus (L.). Cell Tissue Res 157:25–51. https://doi.org/10.1007/bf00223229
Rønnekleiv OK, Møller M (1979) Brain-pineal nervous connections in the rat: an ultrastructure study following habenular lesion. Exp Brain Res 37:551–562. https://doi.org/10.1007/BF00236823
Rønnekleiv OK, Kelly MJ, Wuttke W (1980) Single unit recordings in the rat pineal gland: evidence for habenulo-pineal neural connections. Exp Brain Res 39:187–192. https://doi.org/10.1007/BF00237549
Sakai Y, Hira Y, Oomori Y, Daikoku S, Matsushima S (1994) Immunohistochemical studies on sympathetic and non-sympathetic nerve fibers and neuronal cell bodies in the pineal gland of cotton rats, Sigmodon hispidus. Arch Histol Cytol 57:47–58. https://doi.org/10.1679/aohc.57.47
Scharenberg K, Liss L (1965) The histologic structure of the human pineal body. Prog Brain Res 10:193–217. https://doi.org/10.1016/s0079-6123(08)63452-4 (PMID: 14281603)
Schneider CA, Rasband WS, Eliceiri KW (2012) NIH Image to ImageJ: 25 years of image analysis. Nature Methods 9:671–675. https://doi.org/10.1038/nmeth.2089
Semm P, Schneider T, Vollrath L (1981) Morphological and electrophysiological evidence for habenular influence on the guinea-pig pineal gland. J Neural Transm 150:247–266. https://doi.org/10.1007/BF01249146
Sheridan MN, Rollag MD (1983) Development and melatonin content of the deep pineal gland in the Syrian hamster. Am J Anat 168:145–156. https://doi.org/10.1002/aja.1001680204
Shiotani Y, Yamano M, Shiosaka S, Emson PC, Hillyard CJ, Girgis S, MacIntyre I (1986) Distribution and origins of substance P (SP)-, calcitonin gene-related peptide (CGRP)-, vasoactive intestinal polypeptide (VIP)-, and neuropeptide Y (NPY)-containing nerve fibers in the pineal gland of gerbils. Neurosci Lett 70:87–192. https://doi.org/10.1016/0304-3940(86)90461-1
Simonneaux V, Ouichou A, Pévet P (1993) Pituitary adenylate cyclase-activating polypeptide (PACAP) stimulates melatonin synthesis from rat pineal gland. Brain Res 603:148–152. https://doi.org/10.1016/0006-8993(93)91313-h
Tricoire H, Malpaux B, Møller M (2003a) Morphological indications for a direct secretion of melatonin from the pineal gland to the third ventricle of the sheep. A light and electron microscopical study of the pineal recess. J Comp Neurol 456:39–47. https://doi.org/10.1002/cne.10477
Tricoire H, Møller M, Chemineau P, Malpaux B (2003b) Origin of cerebrospinal fluid melatonin and possible function in the integration of photoperiod. Reproduction, Suppl 61:311–321 (PMID: 14635944)
Vollrath L (1973) Synaptic ribbons of a mammalian pineal gland circadian changes. Cell Tissue Res 145:171–183. https://doi.org/10.1007/bf00307386
Vollrath L (1981) The pineal organ. Hdb mikr Anat Mensch, VI/7 (A Oksche, L Vollrath, eds) Springer: Berlin Heidelberg New York.
Welsh MG (1983) CSF-contacting pinealocytes in the pineal recess of the Mongolian gerbil: a correlative scanning and transmission electron microscope study. Am J Anat 166:483–493. https://doi.org/10.1002/aja.1001660408 (PMID: 6858943)
Wolfe DE (1965) The epiphyseal cell: an electron-microscopic study of its intercellular relationship and intracellular morphology in the pineal body of the Albino rat. Prog Brain Res 10:332–386. https://doi.org/10.1016/s0079-6123(08)63460-3 (PMID: 14281612)
Yuwiler A (1983) Vasoactive intestinal peptide stimulation of pineal serotonin-N-acetyltransferase activity: general characteristics. J Neurochem 41:146–153. https://doi.org/10.1111/j.1471-4159.1983.tb11826.x.PMID
Zhang E, Mikkelsen JD, Møller M (1991) Tyrosine hydroxylase-and neuropeptide Y-immunoreactive nerve fibers in the pineal complex of untreated rats and rats following removal of the superior cervical ganglia. Cell Tissue Res 265:63–71. https://doi.org/10.1007/BF00318140
Funding
This work was supported by the Velux foundation, grant no. 37840 to Morten Møller, the Independent Research Fund Denmark, grant no. 8020-00037B (to MFR), and the Lundbeck Foundation, grant no. R344-2020–261 (to MFR).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethics approval
All animal experiments were performed in accordance with the guidelines of EU directive 2010/63/EU. The study was approved by the Danish Animal Experiments Inspectorate (authorization number 2017–15-0201–01190) and by the Faculty of Health and Medical Sciences, University of Copenhagen (authorization number P21-146).
Informed consent
The informed consent of the participants in this study was obtained verbally and confirmed by e-mail.
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Møller, M., Midtgaard, J., Qvortrup, K. et al. An ultrastructural study of the deep pineal gland of the Sprague Dawley rat using transmission and serial block face scanning electron microscopy: cell types, barriers, and innervation. Cell Tissue Res 389, 531–546 (2022). https://doi.org/10.1007/s00441-022-03654-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00441-022-03654-5