Summary
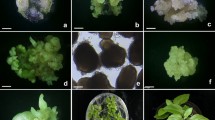
Immature embryo explants taken 8 days after anthesis were used to establish callus cultures of spring barley. Two types of calli were observed. A soft, watery callus produced a limited number of shoots and a harder, more compact, yellowish callus gave rise to numerous green primordia and shoots. Gamborg's B5 basal medium supplemented with either 2,4-D (2,4-dichlorophenoxyacetic acid) or Cl3 POP (2,4,5-trichlorophenoxypropionic acid) was found to give good callus growth and shoot initiation. Media containing 2,4-D at 1.0 mg L−1 or Cl3 POP at 5.0 mg L−1 produced numerous cultures resulting in regeneration of plants. Plantlets developed roots on basal medium with Cl3 POP at 1.0 mg L−1 or on auxin-free medium. Twenty genetically diverse genotypes were screened to determine if these techniques were suitable for a wide range of spring barley cultivars. Regeneration of plantlets was obtained for 19 of the 20 genotypes approximately 4 months after culture initiation. Lines differed in the ability to develop vigorously growing calli and in the ability of calli to develop large numbers of shoots and regenerated plantlets.
Similar content being viewed by others
References
Ahloowalia BS (1982) Plant regeneration from callus culture in wheat. Crop Sci 22:405–410
Bayliss MW, Dunn S (1979) Factors effecting callus formation from embryos of barley (Hordeum vulgare). Plant Sci Lett 14:311–316
Cheng TY, Smith HH (1975) Organogenesis from callus cultures of Hordeum vulgare. Planta 123:307–310
Dale PJ, Deambrogio E (1979) A comparison of callus induction and plant regeneration from different explants of Hordeum vulgare. Z Pflanzenphysiol 94:65–77
Deambrogio E, Dale PJ (1980) Effect of 2,4-D on the frequency of regenerated plants in barley and on genetic variability between them. Cereal Res Commun 8:417–423
Eapen S, Rao PS (1982 a) Plant regeneration from callus cultures of ‘Durum’ and ‘Emmer’ wheat. Plant Cell Rep 1:215–218
Eapen S, Rao PS (1982 b) Callus induction and plant regeneration from immature embryos of rye and triticale. Plant Cell Tissue Organ Culture 1:221–227
Gamborg OL, Wetter LR (1975) Plant tissue culture methods. Published by the National Research Council of Canada, Prarie Regional Laboratory, Saskatoon, Saskatchewan, Canada
Heyser JW, Nabors MW (1982 a) Regeneration of proso millet from embryogenic calli derived from various plant parts. Crop Sci 22:1070–1074
Heyser JW, Nabors MW (1982 b) Long term plant regeneration, somatic embryogenesis and green spot formation in secondary oat (Avena sativa) callus. Z Pflanzenphysiol 107:153–160
Jelaska S, Rengel Z, Cesar V (1984) Plant regeneration from mesocotyl callus of Hordeum vulgare L. Plant Cell Rep 3:125–129
Larkin PJ, Ryan SA, Bretell RIS, Scowcroft WR (1984) Heritable somaclonal variation in wheat. Theor Appl Genet 67:443–455
Nabors MW, Heyser JW, Dykes TA, DeMott KJ (1983) Long-duration, high-frequency plant regeneration from cereal tissue cultures. Planta 157:385–391
Orton T (1979) A quantitative analysis of growth and regeneration from tissue cultures of Hordeum vulgare, H. jubatum, and their interspecific hybrid. Environ Exp Bot 19:319–335
Rogers SO, Quatrano RS (1983) Morphological staging of wheat caryopsis development. Am J Bot 70:308–311
Saalbach G, Koblitz H (1977) Karyological instabilities in callus cultures from haploid barley plants. Biochem Physiol Pflanz 171:469–473
Scheunert EU, Shamina ZB, Koblitz H (1977) Studies on barley calluses cultured in vitro. 1. Establishment, maintenance and growth of two different tissue strains. Plant Sci Lett 10:313–318
Sears RG, Deckard EL (1982) Tissue culture variability in wheat: callus induction and plant regeneration. Crop Sci 22:546–550
Author information
Authors and Affiliations
Additional information
Communicated by G. Melchers
Contribution from Department of Crop Science, Oregon State University, Corvallis, OR 97331. Oregon Agric. Exp. Stn. No. 7582
Rights and permissions
About this article
Cite this article
Goldstein, C.S., Kronstad, W.E. Tissue culture and plant regeneration from immature embryo explants of Barley, Hordeum vulgare . Theoret. Appl. Genetics 71, 631–636 (1986). https://doi.org/10.1007/BF00264267
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00264267