Abstract
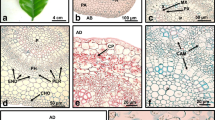
In vitro regeneration and histological analysis of somatic embryogenesis derived from in vitro leaves were assessed in Phlox paniculata L. The explants were incubated on Murashige and Skoog medium containing 2.26–4.52 μM 2,4-dichlorophenoxyacetic acid (2,4-D) with or without 0.89 μM benzyladenine supplemented with 3% or 9% sucrose and vitamins. The intact leaf blades were cultured in the dark at temperature 22 ± 2◦C. Two Phlox cultivars showed significantly higher regeneration capacity in the in vitro conditions when the cultivation media have been supplemented with 2,4-D at concentrations of 4.52 μM and 3% sucrose. The effect of 2,4-D on somatic embryo induction has been enhanced in presence of cytokinin benzyladenine. In cultivars, the embryogenic structures have been created on calluses derived from leaf sections within 6–9 weeks in culture. Meristematic zones with forming embryogenic structures were mostly localized adjacent to de novo forming tracheal elements in calluses in our experiments. In the absence of tracheal elements, the embryogenic structures have been surrounded by several layers of isodiametric cells. The embryo proper and the suspensor part could be clearly distinguished on the embryogenic structures. The embryogenic potential of cultures has been very high, so that even secondary and tertiary somatic embryos were formed. Within 6–9 weeks in culture, well differentiated embryogenic structures up to torpedo stages were developed. Cultivar ‘Fuji’ exhibited slightly higher embryogenic response in culture than cultivar ‘Starfire’. The cultures maintained their embryogenic potential for about 18 months. Embryogenic structures have been able to convert to complete plants within 3 months.
Similar content being viewed by others
Abbreviations
- 2,4-D:
-
2,4-dichlorophenoxyacetic acid
- BA:
-
benzyladenine
References
Chen S., Liang M., Li Q. & Xu M. 2008. Study of rapid propagation of Phlox paniculata. Plant Cell Tiss. Org.95: 245–250.
Declerck V. & Korban S.S. 1995. Shoot regeneration from leaf tissues of Phlox paniculata L. J. Plant Physiol. 147: 441–446.
Dedičová B., Obert B., Hricová A., Šamaj J., Bobák M. & Preťová A. 2000. Shoot and embryo-like structures regenerated from from cultured flax (Linum usitatissimum L.) hypocotyl segments. J. Plant Physiol.157: 327–334.
Fraga M., Alonso M. & Borja M. 2004. Shoot regeneration rates of perennial phlox are dependent on cultivar and explant type. Hort. Sci.39: 1373–1377.
Jain A., Rout G.R. & Raina S.N. 2002. Somatic embryogenesis and plant regeneration from callus cultures of Phlox paniculata Linn. Sci. Hort.94: 137–143.
Mackay W.A., Sankhla D. & Sankhla N. 1999. Micropropagation of superior selections of perennial phlox. In Vitro Cell. Dev. Biol.–Plant35: 49–53.
Matiska P. & Vejsadová H. 2010. Polyploidy induction in Phlox paniculata L. under in vitro conditions. Acta Univ. Agric. Silvic. Mendelianae Brun.58: 101–106.
Matiska P., Vejsadova H. & Pretova A. 2006. Induction of morphogenesis in Phlox paniculata L, p. 58. In: XXII EUCARPIA Symposium (Section Ornamentals) “Breeding for Beauty”, Book of Abstracts, 11–15 September 2006, Sanremo, Italy.
Murashige T. & Skoog F. 1962. A revised medium for rapid growth and bioassays with tobacco tissue cultures. Physiol. Plant.15: 473–497.
Nau J. 1996. Ball Perennial Manual: Propagation and Production. Ball Publishing, Batavia, IL, USA, 487 pp.
Olensen M.N. & Fonnesbech M. 1975. Phlox plants from shoot tips. Acta Hort.54: 95–99.
Preťová A. & Williams E.G. 1986a. Direct somatic embryogenesis from immature zygotic embryos of flax Linum usitatissimum L. J. Plant Physiol.126: 155–161.
Preťová A. & Williams E.G. 1986b. Zygotic embryo cloning in oil seed rape (Brassica napus L.). Plant Sci.47: 195–198.
Raghavan V. 2004. Role of 2,4-dichlorophenoxyacetic acid (2,4-D) in somatic embryogenesis on cultured zygotic embryos of Arabidopsis: cell expansion, cell cycling, and morphogenesis during continuous exposure of embryos to 2,4-D. Am. J. Bot.91: 1743–1756.
Šamaj J., Baluška F., Bobak M. & Volkmann D. 1999. Extracellular matrix surface network of embryogenic units of friable maize callus contains arabinogalactan-proteins recognized by monoclonal antibody JIM 4. Plant Cell Rep.18: 369–374.
Schnabelrauch L.S. & Sink K.C. 1979. In vitro propagation of Phlox subulata and Phlox paniculata. Hort. Sci.14: 607–608.
Vejsadová H. & Preťová A. 2000. Cytological and histological study of morphogenesis in Rhododendron L. Adv. Hort. Sci.14: 87–91.
Vejsadová H. & Preťová A. 2003. Somatic embryogenesis in Rhododendron catawbiense ‘Grandiflorum’. Acta Hort.616: 467–470.
Wherry E.T. 1955. The genus Phlox. Morris Arboretum Monographs III, Philadelphia, PA, USA.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Vejsadová, H., Matiska, P., Obert, B. et al. Somatic embryogenesis in Phlox paniculata — histological analysis. Biologia 71, 763–768 (2016). https://doi.org/10.1515/biolog-2016-0100
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1515/biolog-2016-0100