Summary
Coxal hair-plate sensilla in the spider Cupiennius salei are described with respect to their innervation, central projection pattern, electrical response to mechanical stimulation, and putative behavioral function.
-
1.
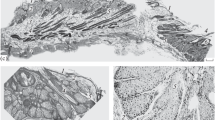
Hair plates, each comprising 25–70 hairs, are situated on the ventrolateral leg coxae close to the prosomal joint; during coxal movements they are deflected by the bulging joint membrane. Each plate hair is innervated by one sensory cell.
-
2.
Threshold sensitivity lies at 0.5° to 1° of hair deflection. Only distalward deflection excites. During maintained deflections the spike rate declines slowly. These hairs differ from hair sensilla of insects in that we measure no ‘standing potential’, nor do we measure a ‘receptor potential’ accompanying a mechanical stimulus.
-
3.
The central projection areas of both hair plates are limited to neuropil of the ipsilateral neuromere.
-
4.
Natural stimulus situation and the spike response to maintained deflection suggest that these hairs are used in proprioception and graviception. Yet behavioral changes following selective hair-plate ablations are not very pronounced. Unilateral removal of hair-plates produced significant increases in average body height in 7 of 10 animals, while the angular orientation of the long body axis with respect to gravity remained unchanged after hair-plate removal.
Similar content being viewed by others
Abbreviations
- CNS :
-
central nervous system
- TEP :
-
transepithelial potential difference
References
Babu KS, Barth FG (1989) Central nervous projections of mechanoreceptors in the spider Cupiennius salei Keys. Cell Tissue Res 258:69–82
Bacon JP, Altman JS (1977) A silver intensification method for cobalt-filled neurones in wholemount preparations. Brain Res 138:359–363
Barth FG (1971) Der sensorische Apparat der Spaltsinnesorgane (Cupiennius salei Keys., Araneae). Z Zellforsch 112:212–246
Barth FG (1985) Slit sensilla and the measurement of cuticular strains. In: Barth FG (ed) Neurobiology of arachnids. Springer, Berlin Heidelberg New York, pp 162–188
Bässler U (1983) Neural basis of elementary behavior in stick insects. Studies of brain function, vol. 10. Springer, Berlin Heidelberg New York
Bohnenberger J (1981) Matched transfer characteristics of single units in a compound slit sense organ. J Comp Physiol 142:391–402
Eckweiler W, Seyfarth E-A (1988) Tactile hairs and the adjustment of body height in wandering spiders: behavior, leg reflexes, and afferent projections in the leg ganglia. J Comp Physiol A 162:611–621
Eckweiler W, Hammer K, Seyfarth E-A (1989) Long, smooth hair sensilla on the spider leg coxa: sensory physiology, central projection pattern, and proprioceptive function (Arachnida, Araneida). Zoomorphology 109:97–102
Foelix RF (1985) Mechano- and chemoreceptive sensilla. In: Barth FG (ed) Neurobiology of arachnids. Springer, Berlin Heidelberg New York, pp 118–137
French AS (1988) Transduction mechanisms of mechanosensilla. Annu Rev Entomol 33:39–58
Gaffal KP, Theiss J (1978) The tibial thread-hairs of Acheta domesticus L. (Saltatoria, Gryllidae). The dependence of stimulus transmission and mechanical properties on the anatomical characteristics of the socket apparatus. Zoomorphologie 90:41–51
Gnatzy W (1982) ‘Campaniforme’ Spaltsinnesorgane auf den Beinen von Weberknechten (Opiliones, Arachnida). Verh Dtsch Zool Ges 75:248
Gnatzy W, Tautz J (1980) Ultrastructure and mechanical properties of an insect mechanoreceptor: stimulus-transmitting structures and sensory apparatus of the cereal filiform hairs of Gryllus. Cell Tissue Res 13:441–463
Gnatzy W, Mohren W, Steinbrecht RA (1984) Pheromone receptors in Bombyx mori and Antherae pernyi. II. Morphometric analysis. Cell Tissue Res 25:35–42
Grünert U, Gnatzy W (1987) K + and Ca++ in the receptor lymph of arthropod cuticular mechanoreceptors. J Comp Physiol A 161:329–333
Hackney CM, Altman JS (1982) Cobalt mapping of the nervous system: how to avoid artifacts. J Neurobiol 13:403–411
Hergenröder R, Barth FG (1983) The release of attack and escape behavior by vibratory stimuli in a wandering spider (Cupiennius salei Keys.). J Comp Physiol 152:347–358
Kaestner A (1924) Beiträge zur Kenntnis der Lokomotion der Arachniden. I. Araneae. Arch Naturgesch 90A:1–19
Markl H (1962) Borstenfelder an den Gelenken als Schweresinnesorgane bei Ameisen und anderen Hymenopteren. Z Vergl Physiol 45:475–569
McIver SB (1985) Mechanoreception. In: Kerkut GA, Gilbert LI (eds) Comprehensive insect physiology, biochemistry, and pharmacology, vol 6. Pergamon Press, Oxford, pp 71–132
Melchers M (1963) Zur Biologie und zum Verhalten von Cupiennius salei (Keyserling), einer amerikanischen Ctenide. Zool Jb Syst 91:1–90
Milde JJ, Seyfarth E-A (1988) Tactile hairs and leg reflexes in wandering spiders: physiological and anatomical correlates of reflex activity in the leg ganglia. J Comp Physiol A 162:623–631
Pringle JWS (1938) Proprioception in insects. III. The function of the hair sensilla at the joints. J Exp Biol 15:467–473
Reissland A, Görner P (1985) Trichobothria. In: Barth FG (ed) Neurobiology of arachnids. Springer, Berlin Heidelberg New York, pp 138–161
Schmidt JM, Smith JJB (1987) The external sensory morphology of the legs and hairplate system of female Trichogramma minutum Riley (Hymenoptera: Trichogrammatidae). Proc R Soc Lond B 232:323–366
Seyfarth E-A (1980) Daily patterns of locomotor activity in a wandering spider. Physiol Entomol 5:199–206
Seyfarth E-A (1985) Spider proprioception: receptors, reflexes, and control of locomotion. In: Barth FG (ed) Neurobiology of arachnids. Springer, Berlin Heidelberg New York, pp 230–248
Seyfarth E-A, Hammer K (1988) Central projections of cuticular mechanoreceptors in spiders: the specificity of proximal leg sensilla. In: Haupt J (ed) Comptes rendus du XIème colloque d'arachnologie. TUB-Dokumentation 38. Berlin, pp 23–28
Seyfarth E-A, Bohnenberger J, Thorson J (1982) Electrical and mechanical stimulation of a spider slit sensillum: outward current excites. J Comp Physiol 147:423–432
Seyfarth E-A, Eckweiler W, Hammer K (1985) Proprioceptors and sensory nerves in the legs of a spider, Cupiennius salei (Arachnida, Araneida). Zoomorphology 105:190–196
Thorson J, Biederman-Thorson M (1974) Distributed relaxation processes in sensory adaptation. Science 183:161–172
Thurm U (1962) Ableitung der Rezeptorpotentiale und Nervenimpulse einzelner Cuticula-Sensillen bei Insekten. Z Naturforsch 17:285–286
Thurm U (1963) Die Beziehungen zwischen mechanischen Reizgrößen und stationären Erregungszuständen bei Borstenfeld-Sensillen von Bienen. Z Vergl Physiol 46:351–382
Thurm U (1984) Beiträge der Ultrastrukturforschung zur Aufklärung sensorischer Mechanismen. Verh Dtsch Zool Ges 77:89–103
Thurm U, Küppers J (1980) Epithelial physiology of insect sensilla. In: Locke M, Smith D (eds) Insect biology in the future. Academic Press, New York, pp 735–763
Thurm U, Wessel G (1979) Metabolism-dependent transepithelial potential differences at epidermal receptors of arthropods. I. Comparative data. J Comp Physiol 134:119–130
Tyrer NM, Bacon JP, Davies CA (1979) Sensory projections from the wind-sensitive head hairs of the locust Schistocerca gregaria. Cell Tissue Res 203:79–92
Vedel JP (1986) Morphology and physiology of a hair plate sensory organ located on the antenna of the rock lobster Palinurus vulgaris. J Neurobiol 17:65–76
Wendler G (1964) Laufen und Stehen der Stabheuschrecke Carausius morosus: Sinnesborstenfelder in den Beingelenken als Glieder von Regelkreisen. Z Vergl Physiol 48:198–250
Wong RKS, Pearson KG (1976) Properties of the trochanteral hair plate and its function in the control of walking in the cockroach. J Exp Biol 64:233–249
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Seyfarth, EA., Gnatzy, W. & Hammer, K. Coxal hair plates in spiders: physiology, fine structure, and specific central projections. J Comp Physiol A 166, 633–642 (1990). https://doi.org/10.1007/BF00240013
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00240013