Summary
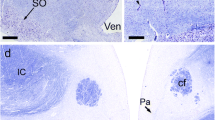
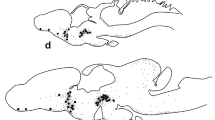
The morphological organization of monoaminergic cells and fibers in the hypothalamus of the lizards Lacerta sicula and Lacerta muralis was investigated by fluorescence histochemistry. An extensive monoaminergic system emanates from the nucleus organi paraventricularis (NOP), a circumventricular organ of the medial and posterior hypothalamus containing numerous monoaminergic perikarya. Fluorescent processes extending from these cells end as intraventricular thickenings. An extensive fiber system presumably arising from the NOP innervates the accompanying nucleus ventromedialis hypothalami (NVH) as well as the nucleus periventricularis hypothalami (NPH) and the median eminence. A monoaminergic fiber path of extrahypothalamic origin enters the preoptic/anterior hypothalamus, terminating in the nucleus paraventricularis (NP). A discrete pathway of catecholaminergic fibers courses through the hypothalamus along the ventral border of the optic tract. Levels of fluorescence intensity are highest in the spring and in castrated animals and lowest in lizards during testicular regression.
Similar content being viewed by others
References
Baumgarten, H.G.: Biogenic monoamines in the cyclostomes and lower vertebrates. Prog. Histochem. Cytochem. 4, 1–90 (1972)
Baumgarten, H.G., Braak, H.: Catecholamine im Gehirn der Eidechse (Lacerta viridis und Lacerta muralis). Z. Zellforsch. 86, 574–602 (1968)
Braak, H., Baumgarten, H.G., Falck, B.: 5-Hydroxytryptamin im Gehirn der Eidechse (Lacerta viridis and Lacerta muralis). Z. Zellforsch. 90, 161–185 (1968)
Brodie, B.B., Bogdanski, D.F., Bonomi, L.: Formation, storage and metabolism of serotonin (5-HT) and catecholamines in lower vertebrates. Comp. Neurochem. Proc. 5th Int. Neurochem. Symp. (Richter, D., ed.) Oxford: Pergamon Press 1964
Campbell, G.T., Wolfson, A.: Hypothalamic norepinephrine, luteinizing hormone releasing factor activity and reproduction in the Japanese quail, Coturnix coturnix japonica. Gen. Comp. Endocrinol. 23, 302–310 (1974)
Corrodi, H., Hillarp, N.-A., Jonsson, G.: Fluorescence methods for the histochemical demonstration of monoamines. 3. Sodium borohydride reduction of the fluorescent compounds as a specificity test. J. Histochem. Cytochem. 12, 582–586 (1964)
Crow, T.J.: Neurotransmitter-related pathways: The structure and function of central monoamine neurons. In: Biochemical correlates of brain structure and function. (Davison, A.N., ed.) New York: Academic Press (1977)
Doshi, E., Huggins, S.E.: Monoamine distribution in the brain of Caiman sclerops. Comp. Biochem. Physiol. [C] 56, 85–88 (1977)
Doshi, E., Huggins, S.E., Fitzgerald, J.M.: Circadian rhythm in the brain serotonin concentrations of the lizard, Anolis carolinensis. Comp. biochem. Physiol. [C] 51, 227–229 (1975)
El Halawani, M.E., Burke, W.H.: Role of catecholamines in photoperiodically-induced gonadal development in Coturnix quail. Biol. Reprod. 13, 603–609 (1975)
Falck, B., Hillarp, N.-A., Thieme, G., Torp, A.: Fluorescence of catecholamines and related compounds condensed with formaldehyde. J. Histochem. Cytochem. 10, 348–354 (1962)
Fischer, K.: Exogene und endogene Steuerung der Fortpflanzungsaktivität bei männlichen Ruineneidechsen (Lacerta sicula campestris Betta). J. Neuro-Visc. Rel. Suppl. X:233 (1971)
Fischer, K.: Die Steuerung der Fortpflanzungszyklen bei männlichen Reptilien. Fortschr. Zool. 22, 362–390 (1974)
Fitch, H.S.: Reproductive cycles in lizards and snakes. Univ. Kans. Mus. Nat. Hist. Misc. Publ. 52 (1970)
Graber, J.W., Nalbandov, A.V.: Relationship of hypothalamic catecholamines and gonadotrophin levels in the chicken. Neuroendocrinology 10, 325–337 (1972)
Hamberger, B., Malmfors, T., Sachs, Ch.: Standardization of paraformaldehyde and of certain procedures for the histochemical demonstration of catecholamines. J. Histochem. Cytochem. 13, 147 (1965)
Harri, M.N.E.: Effect of season and temperature acclimation on the tissue Catecholamine level and utilization in the frog, Rana temporaria. Gen. Pharmacol. 3, 101–112 (1972)
Hökfelt, T., Ljungdahl, A.: Modification of the Falck-Hillarp formaldehyde fluorescence method using the Vibratome: Simple, rapid and sensitive localization of catecholamines in sections of unfixed or formalin fixed brain tissue. Histochemie 29, 325–339 (1972)
Juorio, A.V.: The distribution of catecholamines in the hypothalamus and other brain areas of some lower vertebrates. J. Neurochem. 20, 641–645 (1973)
Klüver, H., Barrera, E.: A method for the combined staining of cells and fibers in the nervous system. J. Neuropath. Exp. Neurol. 12, 400–403 (1953)
Kopin, I.J., Palkovits, M., Kobayashi, R.M., Jacobowitz, D.M.: Quantitative relationship of catecholamine content and histofluorescence in brain of rats. Brain Res. 80, 229–235 (1974)
Licht, P.: Environmental physiology of reptilian breeding cycles: Role of temperature. Gen. and Comp. Endocrinol. Suppl. 3, 477–488 (1972)
McKenna, O.C., Rosenbluth, J.: Cytological evidence for catecholamine — containing sensory cells bordering the ventricle of the toad hypothalamus. J. Comp. Neurol. 154, 133–148 (1974)
Oksche, A.: The neuroanatomical basis of comparative neuroendocrinology. Gen. Comp. Endocrinol. 29, 225–239 (1976)
Parent, A.: Distribution of monoamine-containing nerve terminals in the brain of the painted turtle, Chrysemys picta. J. Comp. Neurol. 148, 153–166 (1973a)
Parent, A.: Demonstration of a catecholaminergic pathway from the midbrain to the strio-amygdaloid complex in the turtle (Chrysemys picta). J. Anat. (Lond.) 114, 379–387 (1973b)
Parent, A., Poirier, L.J.: Occurrence and distribution of monoamine-containing neurons in the brain of the painted turtle, Chrysemys picta. J. Anat. 110, 81–89 (1971)
Parent, A., Poitras, D.: The origin and distribution of catecholaminergic axon terminals in the cerebral cortex of the turtle (Chrysemys picta). Brain Res. 78, 345–358 (1974a)
Parent, A., Poitras, D.: Morphological organization of monoamine-containing neurons in the hypothalamus of the painted turtle (Chrysemys picta). J. comp. Neurol. 154, 379–394 (1974b)
Quay, W.B., Wilhoft, D.C.: Comparative and regional differences in serotonin content of reptilian brains. J. Neurochem. 11, 805–811 (1964)
Quay, W.B., Kelley, T.D., Stebbins, R.C., Cohen, N.W.: Experimental studies on brain 5-hydroxytryptamine and monoamine oxidase in a field population of the lizard Sceloporus occidentalis. Physiol. Zool. 43, 90–97 (1970)
Rujirekagulwat, T., Huggins, S.E.: Brain serotonin content and concentration in the Siamese crocodile, Crocodylus siamensis. Gen. Pharmacol. 6, 133–140 (1975)
Schipper, J., Tilders, F.J.H., Ploem, J.S.: Microfluorimetric scanning of sympathetic nerve fibers. An improved method to quantitate formaldehyde-induced fluorescence of biogenic amines. J. Histochem. Cytochem. 26, 1057–1066 (1978)
Sharp, P.J., Follett, B.K.: The distribution of monoamines in the hypothalamus of the Japanese quail, Coturnix coturnix japonica. Z. Zellforsch. 90, 245–262 (1968)
Soest, S.W., Farner, D.S., Oksche, A.: Fluorescence microscopy of neurons containing primary catecholamines in the ventral hypothalamus of the White-crowned Sparrow, Zonotrichia leucophrys gambelii. Z. Zellforsch. 141, 1–17 (1973)
Weiss, J., Kabisch, K.: Jahreszyklische Untersuchungen der Katecholamine im Hypothalamus von Triturus vulgaris. (L.). Acta Histochem. (Jena) 46, 319–321 (1973)
Welsh, J.H.: The quantitative distribution of 5-hydroxytryptamine in the nervous system, eyes and other organs of some vertebrates. In: Comparative Neurochem. Proc. 5th Int. Neurochem. Symp. (Richter, D., ed.) Oxford: Pergamon Press (1964)
Yamamoto, K. Tohyama, M., Shimizu, N.: Comparative anatomy of the topography of catecholamine-containing neuron system in the brain from birds to teleosts. J. Hirnforsch. 18, 229–240 (1977)
Author information
Authors and Affiliations
Additional information
Submitted in partial fulfillment of the requirements for the degree of Dr. rer. nat. Present address: Abt. Molekulare Genetik, Max Planck Institut für experimentelle Medizin, 3400 Göttingen, Federal Republic of Germany
Rights and permissions
About this article
Cite this article
Marschall, C. Hypothalamic monoamines in lizards (Lacerta). Cell Tissue Res. 205, 95–105 (1980). https://doi.org/10.1007/BF00234446
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00234446