Abstract
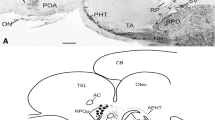
Gonadotropin-inhibitory hormone (GnIH) is a newly discovered hypothalamic RFamide peptide that influences reproduction by regulating brain and pituitary neuroendocrine functions in vertebrates. We report here for the first time, the ontogenetic description of GnIH-like immunoreactivity in the brain, olfactory system, and pituitary of the frog, Pelophylax esculentus. GnIH-like immunoreactive (GnIH-ir) elements were first observed in larvae at stage 24 in the olfactory mucosa, ventral telencephalon, and diencephalon. GnIH-ir-positive staining progressively increased in frequency and intensity during larval growth and other ir perikarya appeared in the medial septum, anterior commissure, dorsal hypothalamus, and posterior tuberculum. A decline in GnIH-ir neurons was seen along the olfactory/vomeronasal/terminal nerve complex in the stages following the pre- and prometamorphosis, while other GnIH-ir neurons showed positivity in the ventromedial surface of the olfactory bulbs and into the habenular nuclei, but the latter are no longer observed in the following stages of development. The anterior-posterior axis in several brain areas, along with the median eminence and pars intermedia of the hypophysis had the appearance of GnIH-ir fibers from early stages, with a progressive increase in the number till metamorphosis in all major subdivisions of the brain. After premetamorphosis, GnIH-ir fibers arising from labeled neurons in the suprachiasmatic nucleus could be seen contacting the ventricular lumen. The transient appearance of GnIH-ir elements in the olfactory system may hint at an olfactory placode origin in the extracranial region. The distribution of GnIH in several brain regions throughout development suggests important involvement of GnIH in multiple brain functions during development.








Similar content being viewed by others
References
Amano M, Moriyama S, Ligo M et al (2006) Novel fish hypothalamic neuropeptides stimulate the release of gonadotrophins and growth hormone from the pituitary of sockeye salmon. J Endocrinol 188:417–423. https://doi.org/10.1677/joe.1.06494
Anjum S, Krishna A, Sridaran R, Tsutsui K (2012) Localization of gonadotropin-releasing hormone (GnRH), gonadotropin-inhibitory hormone (GnIH), kisspeptin and GnRH receptor and their possible roles in testicular activities from birth to senescence in mice. J Exp Zool Part A Ecol Genet Physiol 317:630–644. https://doi.org/10.1002/jez.1765
Biswas S, Jadhao AG, Pinelli C et al (2015) GnIH and GnRH expressions in the central nervous system and pituitary of Indian major carp, Labeo rohita during ontogeny: an immunocytochemical study. Gen Comp Endocrinol 220:88–92. https://doi.org/10.1016/j.ygcen.2014.06.005
Calisi RM (2014) An integrative overview of the role of gonadotropin-inhibitory hormone in behavior: applying Tinbergen’s four questions. Gen Comp Endocrinol 203:95–105. https://doi.org/10.1016/j.ygcen.2014.03.028
Chartrel N, Dujardin C, Leprince J et al (2002) Isolation, characterization, and distribution of a novel neuropeptide, Rana RFamide (R-RFa), in the brain of the European green frog Rana esculenta. J Comp Neurol 448:111–127. https://doi.org/10.1002/cne.10253
Chartrel N, Bruzzone F, Leprince J et al (2006) Structure and functions of the novel hypothalamic RFamide neuropeptides R-RFa and 26RFa in vertebrates. Peptides 27:1110–1120
Chowdhury VS, Ubuka T, Osugi T et al (2011) Identification, localization and expression of LPXRFamide peptides, and melatonin-dependent induction of their precursor mRNA in the newt brain. J Endocrinol 209:211–220. https://doi.org/10.1530/JOE-10-0494
D’Aniello B, Fiorentino M, Pinelli C et al (1996) Distribution of FMRFamide-like immunoreactivity in the brain and pituitary of Rana esculenta during development. Dev Brain Res 95:194–204. https://doi.org/10.1016/0165-3806(96)00088-0
D’Aniello B, Polese G, Luongo L, Scandurra A, Magliozzi L, Aria M, Pinelli C (2016) Neuroanatomical relationships between FMRFamide-immunoreactive components of the nervus terminalis and the topology of olfactory bulbs in teleost fish. Cell and Tissue Research 364(1):43–57
Di Yorio MP, Sallemi JE, Toledo Solís FJ, Pérez Sirkin DI, Delgadin TH, Tsutsui K, Vissio PG (2018) Ontogeny of gonadotrophin-inhibitory hormone in the cichlid fish. Journal of Neuroendocrinology 30(6):e12608
Fiorentino M, Pinelli C, D’Aniello B, Iela L, di Meglio M, Rastogi RK (2001) Development and distribution of FMRFamide-like immunoreactivity in the toad (Bufo bufo) brain. J Chem Neuroanat 21:201–213. https://doi.org/10.1016/s0891-0618(01)00110-7
Jadhao AG, Pinelli C, D’Aniello B, Tsutsui K (2017) Gonadotropin-inhibitory hormone (GnIH) in the amphibian brain and its relationship with the gonadotropin releasing hormone (GnRH) system: an overview. Gen Comp Endocrinol 240:69–76. https://doi.org/10.1016/j.ygcen.2016.09.006
Kanetoh T, Sugikawa T, Sasaki I et al (2003) Identification of a novel frog RFamide and its effect on the latency of the tail-flick response of the newt. Comp Biochem Physiol - C Toxicol Pharmacol 134:259–266. https://doi.org/10.1016/S1532-0456(02)00277-6
Kemali M, Braitenberg V (1969) Atlas of the frog’s brain. Berlin. Springer-Verlag
Koda A, Ukena K, Teranishi H et al (2002) A novel amphibian hypothalamic neuropeptide: isolation, localization, and biological activity. Endocrinology 143:411–419. https://doi.org/10.1210/endo.143.2.8630
Krajniak KG (2013) Invertebrate FMRFamide related peptides. Protein Pept Lett 20:647–670. https://doi.org/10.2174/0929866511320060005
Kriegsfeld LJ, Silver R, Mason AO et al (2006) Identification and characterization of a gonadotropin-inhibitory system in the brains of mammals. Proc Natl Acad Sci 103:2410–2415. https://doi.org/10.1073/pnas.0511003103
Kriegsfeld LJ, Ubuka T, Bentley GE, Tsutsui K (2015) Seasonal control of gonadotropin-inhibitory hormone (GnIH) in birds and mammals. Front Neuroendocrinol 37:65–75
Làzàr G, Losonczy A. (1999) NADPH-diaphorase-positive neurons and pathways in the brain of the frog Rana esculenta. Anat Embryol 199:185–198. https://doi.org/10.1007/s004290050219
Legagneux K, Bernard-Franchi G, Poncet F, La Roche A, Colard C, Fellmann D, Pralong F, Risold PY (2009)Distribution and genesis of the RFRP-producing neurons in the rat brain: comparison with melanin-concentrating hormone and hypocretin-containing neurons. Neuropeptides 43:13–19. https://doi.org/10.1016/j.npep.2008.11.001
Moussavi M, Wlasichuk M, Chang JP, Habibi HR (2013) Seasonal effect of gonadotrophin inhibitory hormone on gonadotrophin-releasing hormone-induced gonadotroph functions in the goldfish pituitary. J Neuroendocrinol 25:506–516. https://doi.org/10.1111/jne.12024
Nieuwenhuys R, Donkelaar H, Nicholson C (1998) The central nervous system of vertebrates. Springer-Verlag, Heideleberg
Northcutt RG, Muske LE (1994) Multiple embryonic origins of gonadotropin-releasing hormone (GnRH) immunoreactive neurons. Dev Brain Res 78:279–290. https://doi.org/10.1016/0165-3806(94)90037-x
Okubo K, Nagahama Y (2008) Structural and functional evolution of gonadotropin-releasing hormone in vertebrates. Acta Physiol 193:3–15. https://doi.org/10.1111/j.1748-1716.2008.01832.x
Osugi T, Ukena K, Bentley GE et al (2004) Gonadotropin-inhibitory hormone in Gambel’s white-crowned sparrow (Zonotrichia leucophrys gambelii): cDNA identification, transcript localization and functional effects in laboratory and field experiments. J Endocrinol 182:33–42. https://doi.org/10.1677/joe.0.1820033
Osugi T, Ubuka T, Tsutsui K (2014) Review: evolution of GnIH and related peptides structure and function in the chordates. Front Neurosci 8:1–11. https://doi.org/10.3389/fnins.2014.00255
Parhar I, Ogawa S, Kitahashi T (2012) RFamide peptides as mediators in environmental control of GnRH neurons. Prog Neurobiol 98:176–196. https://doi.org/10.1016/j.pneurobio.2012.05.011
Paullada-Salmerón JA, Loentgen GH, Cowan M et al (2017) Developmental changes and day-night expression of the gonadotropin-inhibitory hormone system in the European sea bass: effects of rearing temperature. Comp Biochem Physiol-Part A Mol Integr Physiol 206:54–62. https://doi.org/10.1016/j.cbpa.2017.01.009
Pinelli C, D’Aniello B, Fiorentino M et al (1999) Distribution of FMRFamide-like immunoreactivity in the amphibian brain: comparative analysis. J Comp Neurol 414:275–305. https://doi.org/10.1002/(SICI)1096-9861(19991122)414:3<275::AID-CNE1>3.0.CO;2-1
Pinelli C, Rastogi RK, Scandurra A et al (2014) A comparative cluster analysis of nicotinamide adenine dinucleotide phosphate (NADPH)-diaphorase histochemistry in the brains of amphibians. J Comp Neurol 522:1–24. https://doi.org/10.1002/cne.23561
Pinelli C, Jadhao AG, Biswas SP et al (2015) Neuroanatomical organization of the brain gonadotropin-inhibitory hormone and gonadotropin-releasing hormone systems in the frog pelophylax esculentus. Brain Behav Evol 85:15–28. https://doi.org/10.1159/000368594
Rastogi RK, D’Aniello B, Pinelli C et al (2001) FMRFamide in the amphibian brain: a comprehensive survey. Microsc Res Tech 54:158–172. https://doi.org/10.1002/jemt.1130
Rastogi RK, Iela L, di Meglio M et al (2005) Hormonal regulation of reproductive cycles in amphibians. In: Heatwole H (ed) Amphibian biology. Volume 6, Endocrinology. Surrey Beatty & Sons, Sydney, pp 2045–2177
Rastogi RK, Pinelli C, Polese G, et al (2011) Hormones and reproductive cycles in anuran amphibians. In Norris DO & Lopez KH (Eds) Hormones and Reproduction of Vertebrates 2:171–186. San Diego: Elsevier
Sandvik GK, Hodne K, Haug TM et al (2014) RFamide peptides in early vertebrate development. Front Endocrinol (Lausanne) 5. https://doi.org/10.3389/fendo.2014.00203
Tobari Y, Son YL, Ubuka T et al (2014) A new pathway mediating social effects on the endocrine system: female presence acting via norepinephrine release stimulates gonadotropin-inhibitory hormone in the Paraventricular nucleus and suppresses luteinizing hormone in quail. J Neurosci 34:9803–9811. https://doi.org/10.1523/JNEUROSCI.3706-13.2014
Tsutsui K (2009) A new key neurohormone controlling reproduction, gonadotropin-inhibitory hormone (GnIH): biosynthesis, mode of action and functional significance. Prog Neurobiol 88:76–88. https://doi.org/10.1016/j.pneurobio.2009.02.003
Tsutsui K (2016) How to contribute to the progress of neuroendocrinology: new insights from discovering novel neuropeptides and neurosteroids regulating pituitary and brain functions. Gen Comp Endocrinol 227:3–15. https://doi.org/10.1016/j.ygcen.2015.05.019
Tsutsui K, Ubuka T (2018) How to contribute to the progress of neuroendocrinology: discovery of GnIH and progress of GnIH research. Front Endocrinol (Lausanne) 9. https://doi.org/10.3389/fendo.2018.00662
Tsutsui K, Ukena K (2006) Hypothalamic LPXRF-amide peptides in vertebrates: identification, localization and hypophysiotropic activity. Peptides 27:1121–1129. https://doi.org/10.1016/j.peptides.2005.06.036
Tsutsui K, Saigoh E, Ukena K et al (2000) A novel avian hypothalamic peptide inhibiting gonadotropin release. Biochem Biophys Res Commun 275:661–667. https://doi.org/10.1006/bbrc.2000.3350
Tsutsui K, Bentley GE, Ubuka T et al (2007) The general and comparative biology of gonadotropin-inhibitory hormone (GnIH). Gen Comp Endocrinol 153:365–370. https://doi.org/10.1016/j.ygcen.2006.10.005
Tsutsui K, Bentley GE, Bedecarrats G et al (2010a) Gonadotropin-inhibitory hormone (GnIH) and its control of central and peripheral reproductive function. Front Neuroendocrinol 31:284–295. https://doi.org/10.1016/j.yfrne.2010.03.001
Tsutsui K, Bentley GE, Kriegsfeld LJ et al (2010b) Discovery and evolutionary history of gonadotrophin-inhibitory hormone and kisspeptin: new key neuropeptides controlling reproduction. J Neuroendocrinol 22:716–727
Tsutsui K, Ubuka T, Bentley GE, Kriegsfeld LJ (2012) Gonadotropin-inhibitory hormone (GnIH): discovery, progress and prospect. Gen Comp Endocrinol 177:305–314. https://doi.org/10.1016/j.ygcen.2012.02.013
Tsutsui K, Ubuka T, Son YL et al (2015) Contribution of GnIH research to the progress of reproductive neuroendocrinology, Front. Endocrinol. (Lausanne). 6:179
Ubuka T, Ueno M, Ukena K, Tsutsui K (2003) Developmental changes in gonadotropin-inhibitory hormone in the Japanese quail (Coturnix japonica) hypothalamo-hypophysial system. J Endocrinol 178:311–318. https://doi.org/10.1677/joe.0.1780311
Ubuka T, Kim S, Huang YC et al (2008) Gonadotropin-inhibitory hormone neurons interact directly with gonadotropin-releasing hormone-I and -II neurons in European starling brain. Endocrinology 149:268–278. https://doi.org/10.1210/en.2007-0983
Ubuka T, Lai H, Kitani M et al (2009) Gonadotropin-inhibitory hormone identification, cDNA cloning, and distribution in rhesus macaque brain. J Comp Neurol 517:841–855. https://doi.org/10.1002/cne.22191
Ubuka T, Inoue K, Fukuda Y et al (2012a) Identification, expression, and physiological functions of Siberian hamster gonadotropin-inhibitory hormone. Endocrinology 153:373–385. https://doi.org/10.1210/en.2011-1110
Ubuka T, Son YL, Tobari Y, Tsutsui K (2012b) Gonadotropin-inhibitory hormone action in the brain and pituitary. Front Endocrinol (Lausanne) 3:1–13. https://doi.org/10.3389/fendo.2012.00148
Ubuka T, Haraguchi S, Tobari Y et al (2014) Hypothalamic inhibition of socio-sexual behaviour by increasing neuroestrogen synthesis. Nat Commun 5:3061. https://doi.org/10.1038/ncomms4061
Ukena K, Koda A, Yamamoto K et al (2003) Novel neuropeptides related to frog growth hormone-releasing peptide: isolation, sequence, and functional analysis. Endocrinology 144:3879–3884. https://doi.org/10.1210/en.2003-0359
Vigh B, Frank CL, Vincze C et al (2004) The system of cerebrospinal fluid-contacting neurons. Its supposed role in the nonsynaptic signal transmission of the brain. Histol Histopathol 19:607–628. https://doi.org/10.1002/brb3.218
Wada M, Urano A, Gorbman A (1980) A stereotaxic atlas for diencephalic nuclei of the frog, Rana pipiens. Arch Histol Jpn 43:157–173. https://doi.org/10.1679/aohc1950.43.157
Walker RJ, Papaioannou S, Holden-Dye L (2009) A review of FMRFamide- and RFamide-like peptides in metazoa. Invertebr Neurosci 9:111–153. https://doi.org/10.1007/s10158-010-0097-7
Witschi E (1956) Development of vertebrates. Saunders, Philadelphia
Zhang Y, Li S, Liu Y, Lu D, Chen H, Huang X, Liu X, Meng Z, Lin H, Cheng CHK (2010) Structural diversity of the GnIH/GnIH receptor system in teleost: its involvement in early development and the negative control of LH release. Peptides 31:1034–1043. https://doi.org/10.1016/j.peptides.2010.03.003
Funding
This study was supported by the University of Campania “Luigi Vanvitelli” to C.P.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
All procedures performed in studies involving animals were in accordance with the ethical standards of the institution or practice at which the studies were conducted (Ethical Clearance M. D. No. 77-2003A; M. D. No. 103-2005A of the University of Naples Research Ethics Committee).
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Pinelli, C., Jadhao, A.G., Bhoyar, R.C. et al. Distribution of gonadotropin-inhibitory hormone (GnIH)-like immunoreactivity in the brain and pituitary of the frog (Pelophylax esculentus) during development. Cell Tissue Res 380, 115–127 (2020). https://doi.org/10.1007/s00441-019-03139-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00441-019-03139-y