Abstract
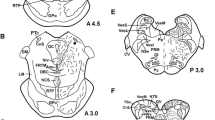
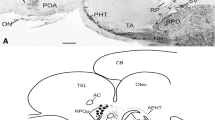
The morphology and neurophysin expression of the magnocellular accessory neuroendocrine system located in the rostral human hypothalamus is investigated in a series of brains obtained at autopsy. The hypothalami were fixed in formalin and embedded in paraffin, or after cryoprotection, frozen for cryostat sectioning. Paraffin sections were either stained with Luxol Fast blue or immunoreacted for neurophysin I or neurophysin II, the precursor molecule for oxytocin and vasopressin. Further, 50-μm-thick serial cryostat sections were immunoreacted with the same antibodies. Both the paraventricular and supraoptic nuclei as well as the hypothalamo-hypophysial tracts exhibited strong immunoreactivity for the neurophysin antibodies. In addition, large collections of immunoreactive accessory magnocellular nuclei and single scattered neurophysin-positive neurons were located in the preoptic region between the paraventricular and supraoptic nucleus among the hypothalamo-hypophysial nerve fibers. In addition, smaller collections of neurophysin-immunoreactive neurons were located in the basal part of this region. Among the accessory magnocellular nuclei, the classical circular nucleus was identified. Accessory magnocellular neurons were often located along the blood vessels and projections of some of these neurons penetrated the vascular endothelium. The accessory magnocellular cell bodies expressed either neurophysin I or neurophysin II immunoreactivity. Summarizing, the accessory magnocellular system in the human brain is large and differs in morphology compared to the system seen in other vertebrates. The neurons of this system contain both vasopressin and oxytocin. Some neurons of the accessory neuronal systems might secrete vasopressin or oxytocin directly into the blood stream.






Similar content being viewed by others
References
Acher R, Chauvet J (1988) Structure, processing and evolution of the neurohypophysial hormone-neurophysin precursors. Biochimie 70:1197–1207
Antunes JL, Zimmerman EA (1978) The hypothalamic magnocellular system of the rhesus monkey: an immunocytochemical study. J Comp Neur 181:539–566
Badiu C, Badiu L, Coculescu M, Vilhardt H, Møller M (2001) Presence of oxytocinergic neuronal-like cells in the bovine pineal gland; an immunocytochemical and in situ hybridization study. J Pineal Res 31:273–280
Bandaranayake RC (1971) Morphology of the accessory neurosecretory nuclei and of the retrochiasmatic part of the supraoptic nucleus of the rat. Acta Anat 80:14–22
Bargmann W (1949) Über die neurosekretorische Verknüpfung von hypothalamus und neurohypophyse. Z Zellforsch 34:610–634
Bodian D, Maren TH (1951) The effect of neuro- and adenohypophysectomy on retrograde degeneration in hypothalamic nuclei of the rat. J Comp Neurol 94:485–511
Braak H, Braak E (1987) The hypothalamus of the human adult: chiasmatic region. Anat Embryol 176:315–330
Breslow E (1993) Structure and folding properties of neurophysin and its peptide complexes: biological implications. Regul Pept 45:15–19
Brockhaus H (1942) Beitrag zur normalen anatomie des hypothalamus und der zona incerta beim Menschen. J Psychol Neurol 51:96–196
Burford GD, Dyball RE, Moss RL, Pickering BT (1974) Synthesis of both neurohypophysial hormones in both the paraventricular and supraoptic nuclei of the rat. J Anat 117:261–269
Castel M, Morris JF (1988) The neurophysing-containing innervation of the forebrain of the mouse. Neuroscience 24:931–966
Diepen R (1962) Der hypothalamus. In: Bargmann W (ed) Handbuch der Mikroskopischen Anatomie des Menschen, vol IV/7. Springer, Berlin-Heidelberg, pp 1–525
Duan X, Ju G (1998) The organization of chemically characterized afferents to the perivascular neuronal groups of the hypothalamic magnocellular neurosecretory system in the rat. Brain Res Bull 46:409–415
Ettrup KS, Sørensen JC, Bjarkam CR (2010) The anatomy of the Göttingen minipig hypothalamus. J Chem Neuroanat 39:151–165
Fisher AW, Price PG, Burford GD, Lederis K (1979) A 3-dimensional reconstruction of the hypothalamo-neurohypophysial system of the rat. The neurons projecting to the neuro/intermediate lobe and those containing vasopressin and somatostatin. Cell Tissue Res 204:343–354
Gagel O (1928) Zur Topik und feineren Histologie der vegetativen Kerne des Zwischenhirns. Z Anat Entwicki ungs gesch 37:548–584
Ginsberg SD, Price L, Blackstone CD, Backastone LH, RL MLJ (1995) The AMPA glutamate receptor GluR3 is enriched in oxytocinergic magnocellular neurons and is localized at synapses. Neurosc 65:563–575
Grinevich VV, Krasnovskaya IA, Voropanova LS, Polenov AL (1994) Accessory groups of nonapeptidergic cells of the diencephalon in intact and hypophysectomized rats. Bull Exp Biol Med 117:607–610
Grünthal E (1930) Vergleichende anatomische und entwicklungsgeschichtliche Untersuchungen über die Zentren des Hypothalamus der Sauger und des Menschen. Arch Psychiat Nervenkr 90:216–267
Hatton GI (1976) Nucleus circularis: is it an osmoreceptor in the brain? Brain Res Bull 1:123–131
Ichimiya Y, Piers C, Emson PC, Fraser D, Shaw FD (1988) Localization of vasopressin mRNA-containing neurones in the hypothalamus of the monkey. Mol Brain Res 4:81–85
Jirikowski GF, Caldwell JD, Pedersen CA, Stumpf WE (1988) Estradiol influences oxytocin-immunoreactive brain systems. Neuroscience 25:237–248
Ju G, Liu S, Tao J (1986) Projections from the hypothalamus and its adjacent areas to the posterior pituitary in the rat. Neuroscience 19:803–828
Kawata M, Sano Y (1982) Immunohistochemical identification of the oxytocin and vasopressin neurons in the hypothalamus of the monkey (Macaca fuscata). Anat Embryol (Berl) 165:151–167
Kiss A, Bundzikova J, Pirnik Z, Mikkelsen JD (2010) Different antipsychotics elicit different effects on magnocellular oxytocinergic and vasopressinergic neurons as revealed by Fos immunohistochemistry. J Neurosci Res 88:677–685
Klüver H, Barrera E (1953) A method for the combined staining of cells and fibers in the nervous system. J Neuropathol Exp Neurol 12:400–403
Koutcherov Y, Mai JK, Ashwell KW, Paxinos G (2000) Organization of the human paraventricular hypothalamic nucleus. J Comp Neurol 423:299–318
Krolewski DM, Medina A, Kerman IA, Bernard R, Burke S, Thompson RC, Bunney WE Jr, Schatzberg AF, Myers RM, Akil H, Jones EG, Watson SJ (2010) Expression patterns of corticotropin-releasing factor, arginine vasopressin, histidine decarboxylase, melanin-concentrating hormone, and orexin genes in the human hypothalamus. J Comp Neurol 518:4591–4611
Kuhlenbeck H, Haymaker W (1949) The derivatives of the hypothalamus in the human brain; their relation to the extrapyramidal and autonomic systems. Mil Surg 105:26–52
Le Gros Clark WE (1936) The topographies and homologies of the hypothalamic nuclei in man. J Anat (London) 70:203–214
Mai JK, Lensing-Höhn S, Ende AA, Sofroniew MV (1997) Developmental organization of neurophysin neurons in the human brain. J Comp Neurol 385:477–489
Makarenko IG, Ugryumov MV, Kalas A (2002) Involvement of accessory neurosecretory nuclei of hypothalamus in the formation of hypothalamohypohysial system during prenatal and postnatal development in rats. R J develop. Biol 33:37–42
Malone EF (1910) Über die Kerne des menschlichen Diencephalon. Abh Preuss Akad Wiss Ahn, Abh 1:1–32
Morton A (1969) A quantitative analysis of the normal neuron population of the hypothalamic magnocellular nuclei in man and of their projections to the neurohypophysis. J comp Neurol 136:37–158
Palkovits M, Zaborszky L, Ambach G (1974) Accessory neurosecretory cell groups in the rat hypothalamus. Acta Morphol Acad Sci Hung 22:21–33
Peterson RP (1966) Magnocellular neurosecretory centers in the rat hypothalamus. J Comp Neurol 128:181–190
Raadsheer FC, Sluiter AA, Ravid R, Tilders FJH, Swaab DF (1993) Localization of corticotropin-releasing hormone (CRH) neurons in the paraventricular nucleus of the human ypothalamus; age-dependent colocalization with vasopressin. Brain Res 615:50–62
Rhodes CH, Morrell JL, Pfaff DW (1981) Immunohistochemical analysis of magnocellular elements in rat hypothalamus: distribution and numbers of cells containing neurophysin, oxytocin, and vasopressin. J Comp Neurol 198:45–64
Rovsing L, Rath MF, Møller M (2013) Hypothalamic neurosecretory and circadian vasopressinergic neuronal systems in the blind cone-rod homeobox knock out mouse (Crx−/−) and the 129sv wild type mouse. J Comp Neurol 521:4061–4074
Ruggeri RM, Ferraù F, Campennì A, Simone A, Barresi V, Giuffrè G, Tuccari G, Baldari S, Trimarchi F (2009) Immunohistochemical localization and functional characterization of somatostatin receptor subtypes in a corticotropin releasing hormone- secreting adrenal phaeochromocytoma: review of the literature and report of a case. Eur J Histochem 53:1–6
Sachs H, Fawcett P, Takabatake Y, Portanova R (1969) Biosynthesis and release of vasopressin and neurophysin. Recent Prog Horm Res 25:447–491
Saper CB (2012) Hypothalamus. In: Mai JK, Paxinos G (eds) The Human nervous system, 2nd edn. Elsevier, Amsterdam, pp 548–583
Saper CB, Lowell BB (2014) The hypothalamus. Curr Biol 24:1111–1116
Scharrer B (1937) Über sekretorisch tätige Nervenzellen bei wirbellosen Tieren. Naturwissenschaften 25:131–138
Sofroniew MV, Glasmann W (1981) Golgi-like immunoperoxidase staining of hypothalamic magnocellular neurons that contain vasopressin, oxytocin or neurophysin in the rat. Neurosc 6:619–643
van den Pol AN (1982) The magnocellular and parvocellular paraventricular nucleus of rat: intrinsic organization. J Comp Neurol 206:317–345
van Eerdenburg FJ, Swaab DF, van Leeuwen FW (1992) Distribution of vasopressin and oxytocin cells and fibres in the hypothalamus of the domestic pig (Sus scrofa). J Comp Neurol 318:138–146
Vierling R (1957) Die anatomische Situation der Hypothalamuskerne Nucleus supraopticus und Nucleus paraventricularis bei Rind und Schaf. Anat Anz 104:157–182
Wahren W (1959) Anatomie des Hypothalamus. In: Einführung in die stereotaktischen Operationen mit einem Atlas des menschlichen Gehirns. Eds.: G Schaltenbrand and P Bailey. Vol.1. Thieme, Stuttgart, pp 118–151
Acknowledgments
We wish to thank Rikke Lundorf for expert technical assistance.
Funding
Grant sponsors: The work was partially funded by the SURVIVE project, Dept. Forensic Medicine, University of Copenhagen, Denmark (to JB), the A.P.Møller foundation for the Advancement of Medical Science, gr.nr. 16-249 (to MM), and the Augustinus Foundation, gr.nr.16-4611 (to MM).
Author information
Authors and Affiliations
Contributions
Study concept and design: MM, JRB, CJ, NL, MFR, JB. Acquisition of materials and data: MM, JRB, SL, CJ. Analysis and interpretation of data. MM, JRB. Drafting of the manuscript: MM. Critical revision of manuscript: MM, MFR, JRB, JB.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflicts of interest.
Rights and permissions
About this article
Cite this article
Møller, M., Busch, J.R., Jacobsen, C. et al. The accessory magnocellular neurosecretory system of the rostral human hypothalamus. Cell Tissue Res 373, 487–498 (2018). https://doi.org/10.1007/s00441-018-2818-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00441-018-2818-x