Summary
An antibody to opsin isolated from rod outer segments of the frog retina was applied in light- and electron-microscopic immunocytochemical studies to the pineal organ of various vertebrates (Cyprinus carpio, Carassius auratus, Rana esculenta, Emys orbicularis, Pseudemys scripta elegans, Lacerta agilis and viridis, Gallus domesticus, Columba livia, Melopsittacus undulatus, Serinus canaria, Taeniopyga punctata, Didelphis virginiana, Erinaceus roumanicus, Myotis myotis, rabbit, rat, cat).
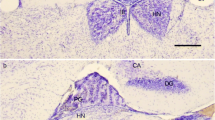
A strong immunoreaction was visible in the outer segments of the pinealocytes of one anuran and several teleost species. The outer segments of pinealocytes in the chelonian reptiles and birds also contained immunoreactive opsin. Ultrastructurally, PAP complexes were localized to the photoreceptor membranes of the outer segments. Immunoreactivity for opsin could not be demonstrated in the lacertilian parietal eye and pineal organ.
In the opossum (Marsupialia), pinealocytes remain in contact with the intrapineal invaginations of the pineal recess. In insectivores, the cilia of the pinealocytes exhibit a relation to glial cells similar to that between the outer segments of retinal photoreceptors and the pigment cells. The cilia of mammalian pinealocytes did not show a clear-cut immunoreactivity to opsin with the electron-microscopic technique employed.
Similar content being viewed by others
References
Clabough JW (1973) Cytological aspects of pineal development in rats and hamsters. Am J Anat 137:215–230
Collin JP (1971) Differentiation and regression of the cells of the sensory line in the epiphysis cerebri. In: Wolstenholme GEW, Knight J (eds) The pineal gland. Ciba Foundation Symposium. Churchill Livingstone, Edinburgh London, pp. 79–125
Dodt E (1973) The parietal eye (pineal and parapineal organs) of lower vertebrates. In: Jung R (ed) Handbook of sensory physiology VII-3B. Springer, Berlin Heidelberg New York, pp 113–140
Erlandsen SL, Parsons JA, Burke JP, Redick JA, Van Orden DE, Van Orden LS (1975) A modification of the unlabeled antibody enzyme method using heterologous antisera for the light microscopic and ultrastructural localization of insulin, glucagon and growth hormone. J Histochem Cytochem 23:666–677
Hamasaki DI, Dodt E (1969) Light sensitivity of the lizard's epiphysis cerebri. Pflüger's Arch ges Physiol 313:19–29
Hartwig H-G (1975) Neurobiologische Studien an photoneuroendokrinen Systemen. Habil.-Schrift, Fachbereich Humanmedizin, Giessen
Hartwig H-G, Baumann Ch (1974) Evidence for photosensitive pigments in the pineal complex of the frog. Vision Res 14:597–598
Hewing M (1978) A liquor contacting area in the pineal recess of the golden hamster (Mesocricetus auratus). Anat Embryol 153:295–304
Hewing M (1980) Cerebrospinal fluid-contacting area in the pineal recess of the vole (Microtus agrestis), guinea pig (Cavia cobaya) and rhesus monkey (Macaca mulatta). Cell Tissue Res 209:473–484
Hisano N, Cardinali DP, Rosner JM, Nagle CA, Tramezzani JH (1972) Pineal role in the duck extraretinal photoreception. Endocrinology 91:1318–1322
Hofer HO, Merker G, Oksche A (1976) Atypische Formen des Pinealorgans der Säugetiere. Verh Anat Ges 70:97–102
Marucci AA, Dougherty RM (1975) Use of the unlabeled antibody histochemical technique for the detection of human antibody. J Histochem Cytochem 23:618–623
McNulty JA, Hazlett JC (1980) The pineal region in the opossum, Didelphis virginiana. I. Ultrastructural observations. Cell Tissue Res 207:109–121
Morita Y (1966) Absence of electrical activity of the pigeon's pineal organ in response to light. Experientia 22:402
Morita Y, Bergmann G (1971) Physiologische Untersuchungen und weitere Bemerkungen zur Struktur des lichtempfindlichen Pinealorgans von Pterophyllum scalare Cuv et Val (Cichlidae, Teleostei). Z Zellforsch 119:289–294
Oksche A (1970) Differentiation of sensory and secretory structures in the central nervous system. Proc. 64th Congr German Zool Soc. Gustav Fischer, Stuttgart, pp 72–79
Oksche A, Hartwig H-G (1979) Pineal sense organs — components of photoneuroendocrine systems. Prog Brain Res 52:113–130
Papermaster DS, Schneider BG, Zorn MA, Kraehenbuhl JP (1977) Quantitative immunocytochemical analysis of membrane protein antigen distribution in photoreceptor cell outer segments. J Cell Biol 75:210a
Papermaster DS, Schneider BG, Zorn MA, Kraehenbuhl JP (1978) Immunocytochemical localization of opsin in outer segments and Golgi zones of frog photoreceptor cells. An electron microscope analysis of cross-linked albumin-embedded retinas. J Cell Biol 77:196–210
Pévet P, Kappers JA, Voûte AM (1977) Morphologic evidence for differentiation of pinealocytes from photoreceptor cells in the adult noctule bat (Nyctalus noctula Schreber). Cell Tissue Res 182:99–109
Ralph CL, Dawson DC (1968) Failure of the pineal body of two species of birds (Coturnix coturnix japonica and Passer domesticus) to show electrical responses to illumination. Experientia 24:147–148
Röhlich P (1976) Photoreceptor membrane carbohydrate on the intradiscal surface of retinal rod disks. Nature 263; No 5580:780–791
Rosner JM, Declercq de Perez Bedes G, Cardinali DP (1971) Direct effect of light on duck pineal explants. Life Sci 10:1065–1069
Sternberger LA (1979) Immunocytochemistry, 2nd ed. Wiley-Sons, New York
Ueck M (1970) Weitere Untersuchungen zur Feinstruktur und Innervation des Pinealorgans von Passer domesticus L. Z Zellforsch 105:276–302
Vigh B, Vigh-Teichmann I (1975) Vergleich der Ultrastruktur der Liquorkontaktneurone und Pinealozyten der Säugetiere. Verh Anat Ges 69:453–461
Vigh B, Vigh-Teichmann I (1981) Immunocytochemical demonstration of rhodopsin in the pinealocytes of various vertebrates. 2nd Colloquium of the European Pineal Study Group, Giessen, FRG, June 30 to July 5. Abstract EPSG Newsletter, Suppl 3, p 56
Vigh B, Vigh-Teichmann I, Aros B (1975) Comparative ultrastructure of cerebrospinal fluid-contacting neurons and pinealocytes. Cell Tissue Res 158:409–424
Vigh B, Vigh-Teichmann I, Röhlich P (1981a) The special axonic and dendritic terminals of neurosecretory cells. Verh Anat Ges 75:785–787
Vigh B, Vigh-Teichmann I, Röhlich P, Aros B (1981b) Immunoreactive opsin in the pineal organ of reptiles and birds. Z mikr-anat Forsch (in press)
Vigh-Teichmann I, Vigh B (1977) Zilientragende Perikaryen im Diencephalon. Verh Anat Ges 71:989–995
Vigh-Teichmann I, Vigh B (1979) A comparison of epithalamic, hypothalamic and spinal neurosecretory terminals. Acta biol Acad Sci hung 30:1–39
Vigh-Teichmann I, Vigh B, Röhlich P, Olsson R (1980a) Phylogenetic aspects of the sensory neurons of the wall of the diencephalon. In: Spatz M, Mrsulja BB, Rakic LjM, Lust WD (eds) Circulatory and developmental aspects of brain metabolism. Plenum Press, New York, pp 415–428
Vigh-Teichmann I, Röhlich P, Vigh B, Aros B (1980b) Comparison of the pineal complex, retina and cerebrospinal fluid contacting neurons by immunocytochemical antirhodopsin reaction. Z mikranat Forsch 94:623–640
Vollrath L (1981) The pineal organ. Handbuch der mikrokopischen Anatomie des Menschen, Bd 6 Teil 7, Oksche A, Vollrath L (eds) Springer, Berlin Heidelberg New York
Vollrath L, Boeckmann D (1978) Comparative anatomy of the rodent pineal complex. Gen Comp Endocrinol 34; Abstract No 39
Zimmermann BL, Tsou MOM (1975) Morphologic evidence of photoreceptor differentiation of pinealocytes in the neonatal rat. J Cell Biol 66:60–75
Author information
Authors and Affiliations
Additional information
A preliminary report based on a portion of the results was presented at the 2nd Colloquium of the European Pineal Study Group, Giessen, June 30 to July 5, 1981
Rights and permissions
About this article
Cite this article
Vigh, B., Vigh-Teichmann, I. Light- and electron-microscopic demonstration of immunoreactive opsin in the pinealocytes of various vertebrates. Cell Tissue Res. 221, 451–463 (1981). https://doi.org/10.1007/BF00216748
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00216748