Summary
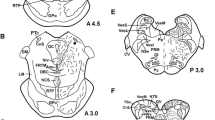
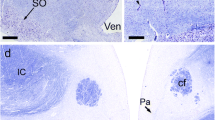
The distributions of ACTH-, αMSH-, βLPH-, and enkephalin-related substances were determined immunohistochemically in the pituitary of the brook lamprey, Lampetra lamotenii. An antiserum directed against the middle region of ACTH reacted chiefly with cells in the proadenohypophysis. An antiserum specific for αMSH reacted with all of the cells of the meta-adenohypophysis, but did not react with any of the middle ACTH-positive cells in the pro-adenohypophysis. Several antisera which crossreact with both βLPH and β-endorphin did not react with any region of the lamprey pituitary. However, an antiserum directed against γLPH did react with a small population of cells in the meso-adenohypophysis. This reactivity could be blocked following pre-absorption with mouse βLPH but was not blocked by synthetic β-endorphin(1–31).
Antisera directed against either met-enkephalin or leuenkephalin reacted with fibers in the anterior neurohypophysis, cells in the pro-adenohypophysis, and all the cells of the meta-adenohypophysis. This crossreactivity could be blocked following pre-absorption with the appropriate enkephalin, but not by pre-absorption with synthetic β-endorphin(1–31) or dynorphin(1–13). In addition, the enkephalin-like reactivity in the adenohypophysis of the lamprey was coincident with middle ACTH-like immunoreactivity in the pro-adenohypophysis and with αMSH-like immunoreactivity in the meta-adenohypophysis.
The absence of βLPH/β-endorphin immunoreactivity coincident with ACTH immunoreactivity, and the presence of enkephalin-like material in the adenohypophysis are unique to the lamprey.
Similar content being viewed by others
References
Adams JC (1981) Heavy metal intensification of DAB-based HRP reaction product. J Histochem Cytochem 29:775
Baker B (1980) The evolution of ACTH, MSH, and LPH-structure, function, and development. In: Barrington EJW (ed) Hormone evolution. Academic Press, London, Vol 2:643–722
Berod A, Hartman BK, Pujol JF (1981) Importance of fixation in immunohistochemistry: use of formaldehyde solutions at variable pH for the localization of tyrosine hydroxylase. J Histochem Cytochem 29:844–850
Browne CA, Bennet HPJ, Solomon S (1981) The isolation and characterization of γ 3-melanotropin from the neurointermediate lobe of the rat pituitary. Biochem Biophys Res Commun 100:336–343
Dores RM (1982a) Localization of multiple forms of ACTH and β-endorphin-related substances in the pituitary of the reptile, Anolis carolinensis. Peptides 3:913–924
Dores RM (1982b) Evidence for a common precursor for αMSH and β-endorphin in the intermediate lobe of the pituitary of the reptile Anolis carolinensis. Peptides 3:925–935
Dores RM, Ringer TE, Gold MR (1982) Immunohistochemical localization of enkephalin-, and ACTH-related substances in the adenohypophysis of the lamprey. Soc Neurosci Abstr 8:98
Eastman JT, Portanova R (1982) ACTH activity in the pituitary and brain of the least brook lamprey, Lampetra aepyptera. Gen Comp Endocrinol 47:346–350
Eipper BA, Mains RE (1978) Existence of a common precursor to ACTH and endorphin in the anterior and intermediate lobes of the rat pituitary. J Supramol Struct 8:247–262
Eipper BA, Mains RE (1980) Structure and function of pro-adrenocorticotropin/endorphin and related peptides. Endocr Rev 1:247–262
Eipper BA, Mains RE (1981) Further analysis of post-translational processing of β-endorphin in rat intermediate pituitary. J Biol Chem 256:5689–5695
Eipper BA, Glembotski CC, Mains RE (1983) Selective loss of α-melanotropin amidating activity in primary cultures of rat intermediate pituitary cells. J Biol Chem: in press
Estivariz FE, Iturriza F, McLean C, Hope J, Lowry PJ (1982) Stimulation of adrenal mitogenesis by N-terminal proopiocortin peptides. Nature 297:419–422
Gianoulakis C, Seidah NG, Routhier R, Chretien M (1979) Biosynthesis and characterizaton of adrenocorticotropic hormone, α-melanocyte-stimulating hormone, and an NH2-terminal fragment of the adrenocorticotropic/β-lipotropin precursor from rat pars intermedia. J Biol Chem 254:11903–11906
Glembotski CC (1982) Acetylation of α-melanotropin and β-endorphin in the rat intermediate pituitary. J Biol Chem 257:20493–20500
Gold MR, Finger TE (1982) Localization of enkephalin-like immunoreactivity in the brain of the lamprey. Soc Neurosci Abstr 7:85
Goossens N, Dierickx K, Vandesande F (1977) Immunocytochemical demonstration of the hypothalamo-hypophysial vasotocinergic system of Lampetra fluviatilis. Cell Tissue Res 177:317–323
Gorbman A (1965) Vascular relation between the neurohypophysis and adenohypophysis of cyclostomas and the problem of hypothalamic neuroendocrine control. Archs Anat Microsc Morphol Exp 54:163–194
Hardisty MW (1981) Biology of the cyclostomes. Chapman and Hall, London 198–223
Herbert E, Budarf M, Phillips M, Rosa P, Policastro P, Oates E, Roberts JL, Seidah NG, Chretien M (1980) The presence of a presequence in the common precursor to ACTH and endorphin and the role of glycosylation in the processing of the precursor and secretion of ACTH and endorphin. Ann NY Acad Sci 343:79–93
Holmes RL, Ball JN (1974) The pituitary gland: a comparative account. Cambridge Press, Cambridge 170–220
Kawauchi H, Muramoto K (1979) Isolation and primary structure of melanotropins from salmon pituitary glands. Int J Pept Protein Res 14:373–374
Kawauchi H, Adachi Y, Tsubokawa M (1980a) Occurrence of a new melanocyte stimulating hormone in the salmon pituitary gland. Biochem Biophys Res Commun 96:1508–1517
Kawauchi H, Adachi Y, Ishizuka B (1980b) Isolation and structure of another β-melanotropin from salmon pituitary glands. Int J Pept Protein Res 16:79–82
Kawauchi H, Tsubokawa M, Kanezawa A, Kitagawa K (1980c) Occurrence of two different endorphins in the salmon pituitary. Biochem Biophys Res Commun 92:1278–1288
Kawauchi H, Akiyoshi T, Ken-Ihi A (1982) Gamma-melanotropin is not present in an N-terminal peptide of salmon pro-opiocortin. Int J Pept Protein Res 18:223–227
Krieger DT, Liotta AS, Brownstein MJ, Zimmerman EA (1980) ACTH, β-lipotropin and related peptides in brain, pituitary, and blood. Recent Prog Horm Res 36:277–345
Lanzing WJR (1954) The occurrence of a water balance, a melanophore expanding and an oxytocic principle in the pituitary gland of the river lamprey Lampetra fluviatilis. Acta Endocrinol 16:277–291
Loh YP (1979) Immunological evidence for two common precursors to corticotropins, endorphins, and melanotropin in the neurointermediate lobe of the toad pituitary. Proc Natl Acad Sci USA 76:796–800
Loh YP, Gainer H (1977) Biosynthesis, processing, and control of release of melanotropic peptides in the neurointermediate lobe of Xenopus laevis. J Gen Physiol 70:37–58
Lowry PJ, Scott AP (1975) The evolution of vertebrate corticotrophin and melanocyte stimulating hormone. Gen Comp Endocrinol 26:16–23
Mains RE, Eipper BA (1978) Coordinate synthesis of corticotropins and endorphins by mouse pituitary tumor cells. J Biol Chem 253:651–656
Mains RE, Eipper BA (1979) Synthesis and secretion of corticotropins, melanotropins, and endorphins by rat intermediate pituitary cells. J Biol Chem 254:7885–7894
Mains RE, Eipper BA (1981) Differences in the post-translational processing of β-endorphin in rat anterior and intermediate pituitary. J Biol Chem 256:5683–5688
Mains RE, Eipper BA, Ling N (1978) Common precursor to corticotropins and endorphins. Proc Natl Acad Sci USA 75:3014–3018
McLean C, Lowry PJ (1981) Natural occurrence but lack of melanotrophic activity of MSH in fish. Nature 290:341–343
McLean IW, Nakane PK (1974) Peroxidase-lysine-paraformaldehyde fixative. A new fixative for immunoelectron microscopy. J Histochem Cytochem 22:1077–1083
Miller RJ, Chang K-J, Cooper B, Cuatrecasas P (1978) Radioimmunoassay and characterization of enkephalins in rat tissues. J Biol Chem 253:531–538
Nakanishi S, Inoue A, Kita T, Nakamura M, Chang ACY, Cohen SN, Numa S (1979) Nucleotide sequence of cloned cDNA for bovine corticotropin-β-lipotropin precursor. Nature 278:423–427
Nozaki M, Gorbman A (1983) Immunocytochemical localization of somatostatin and vasotocin in the brain of the Pacific hagfish, Eptatretus stouti. Cell Tissue Res 229:541–550
Pederson RC, Brownie AC (1980) Adreno-cortical response to corticotropin is potentiated by part of the amino-terminal region of procorticotropin/endorphin. Proc Natl Acad Sci USA 77:2239–2243
Pelletier G, Leclerc R, Labrie F, Cote J, Chretian M, Lis M (1977) Immunohistochemical localization of β-lipotropic hormone in the pituitary gland. Endocrinology 100:770–776
Pezella PD, Seidah NG, Bonjannet P, Crine M, Lis M, Chretien M (1978) Biosynthesis of beta-endorphin, beta-lipotropin, and the putative ACTH-LPH precursor in the frog pars intermedia. Life Sci 23:2281–2292
Roberts JL, Herbert E (1977) Characterization of a common precursor and identification of corticotropin peptides in the molecules. Proc Natl Acad Sci USA 74:4826–4830
Rossier J, Vargo TM, Minick S, Ling N, Bloom FE, Guillemin R (1977) Regional dissociation of β-endorphin and enkephalin contents in rat brain and pituitary. Proc Natl Acad Sci USA 74:5162–5165
Rudman D, Chawla RK, Hollins BM (1979) N, O-diacetylserine α-melanocyte-stimulating hormone, a naturally occurring melanotropic peptide. J Biol Chem 254:10102–10108
Rurak DW, Perks AM (1976) The neurohypophysial principles of the Western Brook lamprey, Lampetra richardsoni: studies in the adult. Gen Comp Endocrinol 29:301–312
Rurak DW, Perks AM (1977) The neurohypophysial principle of the Western Brook lamprey, Lampetra richardsoni: studies in the ammocoete larvae. Gen Comp Endocrinol 31:91–100
Schwyzer R, Schiller P, Seelig S, Sayer G (1971) Isolated adrenal cells: log dose response curve for steroidogenesis induced by ACTH(1–24), ACTH(1–10), ACTH(4–10) and ACTH(5–10). FEBS Lett 19:229–231
Scott A, Besser GM, Ratchiffe JG (1971) A phylogenetic study of pituitary corticotrophic activity. J Endocrinol 51: i-ii
Simantov R, Goodman R, Aposhian D, Snyder SH (1976) Phylogenetic distribution of a morphine-like peptide ‘enkephalin’. Brain Res 111:204–211
Sternberger LA (1979) Immunocytochemistry, 2nd Edition, J Wiley and Sons, New York, New York
Tsuneki K, Gorbman A (1975a) Ultrastructure of anterior neurohypophysis and the pars distalis of the lamprey, Lampetra tridentata. Gen Comp Endocrinol 25:487–508
Tsuneki K, Gorbman A (1975b) Ultrastructure of pars nervosa and pars intermedia of the lamprey, Lampetra tridentata. Cell Tissue Res 157:165–184
Watson SJ, Akil H, Sullivan SO, Barchas JD (1977) Immunocytochemical localization of methionine-enkephalin: Preliminary observations. Life Sci 25:733–738
Watson SJ, Akil H, Richard CW, Barchas JD (1978) Evidence for two separate opiate peptide neuronal systems. Nature 274:226–228
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Dores, R.M., Finger, T.E. & Gold, M.R. Immunohistochemical localization of enkephalin and ACTH-related substances in the pituitary of the lamprey. Cell Tissue Res. 235, 107–115 (1984). https://doi.org/10.1007/BF00213730
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00213730