Summary
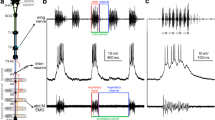
Initiation and modulation of fictive feeding by cerebral to buccal interneurons (CBs) was examined in an isolated CNS preparation of Limax maximus. Three CBs which are phasically active during fictive feeding, CB1, CB3 and CB4, will reliably trigger bouts of fictive feeding when activated alone or in pairs. Another phasic CB, CBEC, is not effective for triggering feeding. One CB which is tonically active during fictive feeding, CBST, drives fictive feeding in 50% of preparations when activated alone and enhances triggering of feeding when co-activated with phasic CBs. The metacerebral giant cell (MGC) was found to be capable of triggering fictive feeding in preparations with an intact subcerebral commissure. The MGC was especially effective at increasing the effectiveness of other CBs for initiation of feeding. Short high-frequency bursts of phasic CB or MGC action potentials are capable of resetting ongoing fictive feeding. Resetting effects of CB action potentials are relatively independent of the phase of the bite-cycle in which they are activated. CB4 phase-advances the bite-cycle while the other phasic CBs phase-delay the bite cycle. Moderate frequency stimulation of CB4 speeds up the bite rate while moderate frequency stimulation of CB3 slows biting. All CBs, except the tonic CB, CBDL, increase the intensity of buccal motor neuron bursting during feeding. The excitatory effects of phasic CBs and the tonic CB, CBEPSP, on fictive feeding persist for many seconds after the offset of stimulation. CBs form both monosynaptic excitatory and monosynaptic inhibitory connections with different BG motor neurons.
Similar content being viewed by others
Abbreviations
- BG :
-
buccal ganglion
- BR :
-
buccal root
- CB :
-
cerebral-buccal interneuron
- CBC :
-
cerebral-buccal connective
- CPG :
-
central pattern generator
- FB :
-
fast burster neuron
- FMP :
-
feeding motor program
- IBI :
-
interbite interval
- MGC :
-
metacerebral giant cell
References
Brodfuehrer PD, Friesen WO (1986) Initiation of swimming activity by trigger neurons in the leech subesophageal ganglion. J Comp Physiol A 159:489–502
Davis WJ, Kennedy D (1972) Command neurons controlling swimmeret movements in the lobster. I. Types of effects on motorneurons. J Neurophysiol 35:20–29
Davis WJ, Kovac MP, Croll RP, Matera E (1984) Brain oscillator(s) underlying rhythmic cerebral and buccal motor output in the mollusc, Pleurobranchaea californica. J Exp Biol 110:1–15
Delaney K, Gelperin A (1985) Cerebral to buccal interneurons which initiate fictive feeding in Limax maximus. Soc Neurosci Abstr 11:509
Delaney K, Gelperin A (1986a) Post-ingestive food-aversion learning to amino acid deficient diets in the terrestrial slug Limax maximus. J Comp Physiol A 159:281–295
Delaney K, Gelperin A (1986b) Cerebral to buccal interneurons controlling initiation and modulation of feeding motor program in Limax maximus. Soc Neurosci Abstr 12:39
Delaney K, Gelperin A (1987) Central correlates of taste discrimination in the slug Limax maximus. Soc Neurosci Abstr 13:388
Delaney K, Gelperin A (1990a) Cerebral interneurons controlling fictive feeding in Limax maximus. I. Anatomy and criteria for re-identification. J Comp Physiol A 166:297–310
Delaney K, Gelperin A (1990b) Cerebral interneurons controlling fictive feeding in Limax maximus. III. Integration of sensory inputs. J Comp Physiol A 166:327–343
Delcomyn F (1980) Neural basis of rhythmic behavior in animals. Science 210:492–498
Friesen WO, Poon M, Stent GS (1978) Neuronal control of swimming in the medicinal leech IV. Identification of a network of oscillatory interneurones. J Exp Biol 75:25–43
Gelperin A (1981) Synaptic modulation by identified serotonin neurons. In: Jacobs BL, Gelperin A (eds) Serotonin neurotransmission and behavior, Chap 10. MIT Press, Cambridge, pp 288–301
Gelperin A, Hopfield JF (1989) Differential conditioning to a compound stimulus and its components in the terrestrial mollusc Limax maximus. Behav Neurosci (in press)
Gelperin A, Chang JJ, Reingold SC (1978) Feeding motor program in Limax. I: Neuromuscular correlates and control by chemosensory input. J Neurobiol 9:285–300
Gelperin A, Tank DW, Tesauro J (1989) Olfactory processing and associative memory: Cellular and modeling studies. In: Byrne JH, Berry WO (eds) Neural models of plasticity: theoretical and empirical approaches. Academic Press, New York (in press)
Getting PA (1983) Neural control of swimming in Tritonia. In: Roberts A, Roberts BL (eds) Neural origin of rhythmic movements. Cambridge Univ Press, New York, pp 89–128
Getting PA (1985) Neural control of behavior in gastropods. In: Willows AOD (ed) The Mollusca, vol. 8, neurobiology and behavior, part 1. Academic Press, New York, pp 269–334
Getting PA, Dekin MS (1985) Tritonia swimming, a model system for integration within rhythmic motor systems. In: Selverston AI (ed) Model neural networks and behavior. Plenum Press, New York, pp 3–20
Gillette R, Kovac MP, Davis WJ (1978) Command neurons in Pleurobranchaea receive synaptic feedback from the motor network they excite. Science 199:798–801
Gillette R, Kovac MP, Davis WJ (1982) Control of feeding motor output by paracerebral neurons in brain of Pleurobranchaea californica. J Neurophysiol 47:885–908
King MS, Delaney K, Gelperin A (1987) Acetylcholine activates cerebral interneurons and feeding motor program in Limax. J Neurobiol 18:509–530
Kirk MD (1989) Premotor neurons in the feeding system of Aplysia californica. J Neurobiol 20:497–512
Kovac MP, Davis WJ, Matera E, Gillette R (1982) Functional and structural correlates of cell size in neurons of Pleurobranchaea californica. J Neurophysiol 47:909–927
Kupfermann I, Weiss KR (1978) The command neuron concept. Behav Brain Sci 1:3–39
London JA, Zecević D, Cohen LB (1987) Simultaneous optical recording of activity from many neurons during feeding in Navanax. J Neurosci 7:649–661
McCrohan CR (1984a) Initiation of feeding motor output by an identified interneurone in the snail, Lymnaea stagnalis. J Exp Biol 113:351–366
McCrohan CR (1984b) Properties of ventral cerebral neurons involved in the feeding system of the snail, Lymnaea stagnalis. J Exp Biol 108:257–272
Nusbaum MP, Kristan WB (1986a) Swim initiation in the leech by serotonin-containing interneurons, cells 21 and 61. J Exp Biol 122:277–302
Nusbaum MP, Kristan WB (1986b) Synaptic basis of swim initiation in the leech. III. Synaptic effects of serotonin-containing interneurons (cells 21 and 61) on swim CPG neurons (cells 18 and 208). J Exp Biol 122:303–321
Reingold SC, Gelperin A (1980) Feeding motor program in Limax. II. Modulation by sensory inputs in intact animals and isolated central nervous system. J Exp Biol 85:1–19
Rosen SC, Miller MW, Weiss KR, Kupfermann I (1987) Control of buccal motor programs in Aplysia by identified neurons in the cerebral ganglion. Soc Neurosci Abstr 13:1061
Rovainen CM (1979) Neurobiology of lampreys. Physiol Rev 59:1007–1077
Runham NW, Hunter PJ (1970) Terrestrial slugs. Hutchinson Univ Library, London
Tuersley MD, McCrohan CR (1988) Serotonergic modulation of patterned motor output in Lymnaea stagnalis. J Exp Biol 135:473–486
Weeks JC (1982) Segmental specialization of a leech swim-initiating interneuron, cell 205. J Neurosci 2:972–985
Wieland S, Gelperin A (1983) Dopamine elicits feeding motor program in Limax maximus. J Neurosci 3:1735–1745
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Delaney, K., Gelperin, A. Cerebral interneurons controlling fictive feeding in Limax maximus . J Comp Physiol A 166, 311–326 (1990). https://doi.org/10.1007/BF00204805
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00204805