Summary
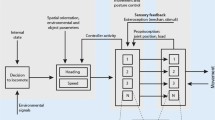
In locusts (Locusta migratoria) walking on a treadwheel, afferents of tarsal hair sensilla were stimulated via chronically implanted hook electrodes (Fig. 1). Stimuli applied to the middle leg tarsus elicited avoidance reflexes (Fig. 2). In quiescent animals, the leg was lifted off the ground and the femur adducted. In walking locusts, the response was phase-dependent. During the stance phase, no reaction was observed except occasional, premature triggering of swing movements; stimuli applied near the end of the swing phase were able to elicit an additional, short leg protraction.
Central nervous correlates of phase-dependent reflex modulation were observed by recording intracellularly from motoneuron somata in walking animals. As a rule, motoneurons recruited during the swing phase showed excitatory stimulus-related responses around the end of the swing movement, correlated to the triggering of additional leg protractions (Figs. 3, 4, 5). Motoneurons active during the stance phase were often inhibited by tarsal stimulation, some showed only weak responses (Figs. 8, 9, 10). Common inhibitory motoneuron 1 was excited by tarsal stimulation during all phases of the leg movement (Figs. 6, 7). In one type of flexor tibiae motoneuron, a complex response pattern was observed, involving the inversion of stimulus-related synaptic potentials from excitatory, recorded during rest, to inhibitory, observed during long-lasting stance phases (Figs. 11, 12).
The results demonstrate how reflex modulation is represented on the level of synaptic input to motoneurons. They further suggest independent gain control in parallel, antagonistic pathways converging onto the same motoneuron as a mechanism for reflex reversal during locomotion.
Similar content being viewed by others
Abbreviations
- CI 1 :
-
common inhibitory motoneuron (1)
- EMG :
-
electromyogram
- Feti :
-
fast extensor muscle of the tibia
References
Andersson O, Forssberg H, Grillner S, Lindquist M (1978) Phasic gain control of the transmission in cutaneous reflex pathways to motoneurones during ‘fictive’ locomotion. Brain Res 149:503–507
Bässler U (1972) Der “Kniesehnenreflex” bei Carausius morosus: Übergangsfunktion und Frequenzgang. Kybernetik 11:32–50
Bässler U (1977) Sense organs in the femur of the stick insect and their relevance to the control of position of the femur-tibia-joint. J Comp Physiol 121:99–113
Bässler U (1983) Neural basis of elementary behavior in stick insects. Braitenberg V (ed) Springer Berlin Heidelberg New York, pp 1–169
Bässler U (1985) Proprioceptive control of stick insect walking. In: Gewecke M, Wendler G (eds) Insect locomotion. Parey, Hamburg, pp 43–48
Bässler U (1986) Afferent control of walking movements in the stick insect Cuniculina impigra. II. Reflex reversal and the release of the swing phase in the restrained foreleg. J Comp Physiol A 158:351–361
Bässler U (1988) Functional principles of pattern generation for walking movements of stick insect forelegs: the role of the femoral chordotonal organ afferences. J Exp Biol 136:125–147
Bässler U, Büschges A (1990) Interneurones participating in the “active reaction” in stick insects. Biol Cybern 62:529–538
Bässler U, Koch UT (1989) Modelling of the active reaction of stick insects by a network of neuromimes. Biol Cybern 62:141–150
Bässler U, Hofmann T, Schuch U (1986) Assisting components within a resistance reflex of the stick insect, Cuniculina impigra. Physiol Entomol 11:359–366
Burns MD (1973) The control of walking in Orthoptera. I. Leg movements in normal walking. J Exp Biol 58:45–58
Burns MD, Usherwood PNR (1979) The control of walking in Orthoptera. II. Motor neurone activity in normal free-walking animals. J Exp Biol 79:69–98
Burrows M (1989) Processing of mechanosensory signals in local reflex pathways of the locust. J Exp Biol 146:209–227
Büschges A (1990) Nonspiking pathways in a joint-control loop of the stick insect Carausius morosus. J Exp Biol 151:133–160
Büschges A, Schmitz J (1991) Nonspiking pathways antagonize the resistance reflex in the thoraco-coxal joint of stick insects. J Neurobiol 22: 224–237
Campbell JI (1961) The anatomy of the nervous system of the mesothorax of Locusta migratoria migratorioides R. and F. Proc R Zool Soc London 137:403–432
Cruse H (1976) The function of the legs in the free walking stick insect, Carausius morosus. J Comp Physiol 112:235–262
Cruse H (1985) The influence of load, position and velocity on the control of leg movement of a walking insect. In: Gewecke M, Wendler G (eds) Insect locomotion. Parey, Hamburg, pp 19–26
Cruse H, Saxler G (1980a) Oscillations of force in the standing legs of a walking insect (Carausius morosus). Biol Cybern 36:159–163
Cruse H, Saxler G (1980b) The coordination of force oscillations and of leg movement in a walking insect (Carausius morosus). Biol Cybern 36:165–171
El Manira A, Cattaert D, Clarac F (1991) Monosynaptic connections mediate resistance reflex in crayfish (Procambarus clarkii) walking legs. J Comp Physiol A 168:337–349
Forssberg H (1979) Stumbling corrective reaction: A phase-dependent compensatory reaction during locomotion. J Neurophysiol 42:936–953
Forssberg H, Grillner S, Rossignol S (1975) Phase-dependent reflex reversal during walking in chronic spinal cats. Brain Res 85:103–107
Forssberg H, Grillner S, Rossignol S (1977) Phasic gain control of reflexes from the dorsum of the paw during spinal locomotion. Brain Res 132:121–139
Godden DH, Graham D (1984) A preparation of the stick insect Carausius morosus for recording intracellularly from identified neurones during walking. Physiol Entomol 9:275–286
Graham D (1972) A behavioural analysis of the temporal organization of walking movements in the 1st instar and adult stick insect (Carausius morosus). J Comp Physiol 81:23–52
Graham D (1979) Effects of circum-oesophageal lesion on the behaviour of the stick insect Carausius morosus. II. Change in walking co-ordination. Biol Cybern 32:147–152
Graham D, Wendler G (1981) Motor output to the protractor and retractor coxae muscles of Carausius morosus during walking on a treadwheel. Physiol Entomol 6:161–174
Hale JP, Burrows M (1985) Innervation patterns of inhibitory motor neurones in the thorax of the locust. J Exp Biol 117:401–413
Helversen O von, Elsner N (1977) The stridulatory movements of acridid grasshoppers recorded with an opto-electronic device. J Comp Physiol 122:53–64
Holst E von (1954) Relations between the central nervous system and the peripheral organs. Brit J Anim Behav 2:89–94
Kendall MD (1970) The anatomy of the tarsi of Schistocerca gregaria (Forskal). Z Zellforsch 109:112–137
Laurent G (1986) Thoracic intersegmental interneurones in the locust with mechanoreceptive inputs from a leg. J Comp Physiol A 159:171–186
Laurent G (1988) Local circuits underlying excitation and inhibition of intersegmental interneurones in the locust. J Comp Physiol A 162:145–157
Laurent GJ, Burrows M (1988) Direct excitation of nonspiking local interneurones by exteroceptors underlies tactile reflexes in the locust. J Comp Physiol A 162:563–572
Laurent G, Burrows M (1989a) Distribution of intersegmental inputs to nonspiking local interneurons and motor neurons in the locust. J Neurosci 9:3019–3029
Laurent G, Burrows M (1989b) Intersegmental interneurons can control the gain of reflexes in adjacent segments of the locust by their action on nonspiking local interneurons. J Neurosci 9:3030–3039
Laurent G, Hustert R (1988) Motor neuronal receptive fields delimit patterns of motor activity during locomotion of the locust. J Neurosci 8:4349–4366
Libersat F, Zill S, Clarac F (1987) Single-unit responses and reflex effects of force-sensitive mechanoreceptors of the dactyl of the crab. J Neurophysiol 57:1601–1617
Libersat F, Clarac F, Zill S (1987) Force-sensitive mechanoreceptors of the dactyl of the crab: single-unit responses during walking and evaluation of function. J Neurophysiol 57:1618–1637
Müller A, Wolf H, Galler S, Rathmayer W (1992) Correlation of electrophysiological, histochemical, and mechanical properties in fibres of the coxa rotator muscle of the locust, Locusta migratoria. J Comp Physiol B 162:5–15
Müller U, Clarac F (1990a) Dactyl sensory influences on rock lobster locomotion. 1. Intrasegmental and intersegmental leg reflexes during standing and walking. J Exp Biol 148:89–112
Müller U, Clarac F (1990b) Dactyl sensory influences on rock lobster locomotion. 2. Role in interleg coordination. J Exp Biol 148:113–128
Nagayama T, Hisada M (1987) Opposing parallel connections through crayfish local nonspiking interneurons. J Comp Neurol 257:347–358
Newland PL (1991) Physiological properties of afferents from tactile hairs on the hindlegs of the locust. J Exp Biol 155:487–503
Nothof U, Bässler U (1990) The network producing the “active reaction” of stick insects is a functional element of different pattern generating systems. Biol Cybern 62:453–462
Pearson KG, Wolf H (1988) Connections of hindwing tegulae with flight neurones in the locust, Locusta migratoria. J Exp Biol 135:381–409
Phillips CE (1981) Organization of motor neurones to a multiply innervated insect muscle. J Neurobiol 12:269–280
Rathmayer W (1990) nhibition through neurons of the common nhibitory type (CI-neurons) in crab muscles. In: Wiese K (ed) Frontiers in crustacean neurobiology. Advances in life sciences. Birkhäuser, Basel, pp 271–278
Reichert H, Rowell CHF (1985) Integration of nonphaselocked exteroceptive information in the control of rhythmic flight in the locust. J Neurophysiol 53:1201–1218
Robertson RM, Pearson KG (1982) A preparation for the intracellular analysis of neuronal activity during flight in the locust. J Comp Physiol 146:311–320
Rossignol S, Lund JP, Drew T (1988) The role of sensory inputs in regulating patterns of rhythmical movements in higher vertebrates. In: Cohen AH, Rossignol S, Grillner S (eds) Neural control of rhythmic movements in vertebrates. Wiley, NewYork Chichester Brisbane Toronto Singapore, pp 201–283
Schmidt J (1991) Gemeinsame Hemmneurone bei Heuschrecken: Einfluß der Sensorik und Verschaltung mit Interneuronen. PhD thesis, Konstanz
Schmilz J, Haßfeld G (1989) The treading-on-tarsus reflex in stick insects: phase-dependence and modifications of the motor output during walking. J Exp Biol 143:373–388
Schmitz J, Büschges A, Kittmann R (1991) Intracellular recordings from nonspiking interneurons in a semi-intact, tethered walking insect. J Neurobiol 22:907–921
Sillar KT, Roberts A (1988) A neuronal mechanism for sensory gating during locomotion in a vertebrate. Nature 331:262–265
Sillar KT, Skorupski P (1986) Central input to primary afferent neurones in crayfish, Pacifastacus leniusculus, is correlated with rhythmic output of thoracic ganglia. J Neurophysiol 55:678–688
Snodgrass RE (1929) The thoracic mechanism of a grasshopper and its antecedents. Smithson Misc Coll 82:1–111
Stein RB, Pearson KG (1971) Predicted amplitude and form of action potentials recorded from unmyelinated nerve fibers.J Theor Biol 32:539–558
Theophilidis G, Burns MD (1983) The innervation of the mesothoracic flexor tibiae muscle of the locust. J Exp Biol 105:373–388
Theophilidis G, Burns MD (1990) The firing pattern of the locust (Schistocerca gregaria americana) mesothoracic femoral motor axons in resistance reflexes and during walking on a treadmill. J Insect Physiol 36:513–522
Wiens TJ (1989) Common and specific inhibition in leg muscles of decapods: sharpened distinctions. J Neurobiol 20:458–469
Wilson JA (1979) The structure and function of serially homologous leg motor neurons in the locust. I. Anatomy. J Neurobiol 10:41–65
Wolf H (1990) Activity patterns of inhibitory motoneurones and their impact on leg movement in tethered walking locusts. J Exp Biol 152:281–304
Wolf H, Schmidt J (1990) Sensory input to locust common inhibitory motoneurons during walking. Verh Dtsch Zool Ges 83:437
Wolf H, Ronacher B, Reichert H (1988) Patterned synaptic drive to locust flight motoneurons after hemisection of thoracic ganglia. J Comp Physiol A 163:761–769
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Wolf, H. Reflex modulation in locusts walking on a treadwheel intracellular recordings from motoneurons. J Comp Physiol A 170, 443–462 (1992). https://doi.org/10.1007/BF00191461
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00191461