Summary
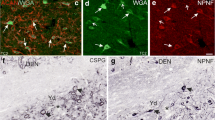
Vestibular neurons from adult rats were studied by immunofluorescence microscopy using ten polyclonal and monoclonal antibodies reacting with different subunits of neurofilament (NF) polypeptides. Most antibodies strongly immunolabeled axons and dendrites. With the majority of antibodies the vestibular ganglion cells showed two distinct subpopulations: those with intense immunoreactivity and those without it. The intensely immunoreactive somata, staining with anti-70 kDa and anti-200 kDa NF antibodies but not with anti-150 kDa NF antibodies, comprised 32% of the ganglion cells (range: 27–40%). These cells were larger than the weakly immunoreactive majority of cells. Comparisons of adjacent sections revealed that at least a part of the distinctly immunostained cells were the same regardless of the antibody used. Thus, about one-third of the vestibular ganglion cells seem to form a subpopulation containing 200 kDa and 70 kDa subunits of NF polypeptides. Similar compositions of NF subunits have been demonstrated in such cells as the large dorsal root ganglion cells. The high percentage of neuronal somata with phosporylated subunits of NF polypeptides demonstrated in this study is exceptional in other normal neurons and has been demonstrated only in experimental models and disease states.
Similar content being viewed by others
References
Anderton BH, Breinburg D, Downes MJ, Green PJ, Tomlinson BE, Ulrich J, Wood JN, Kahn J (1982) Monoclonal antibodies show that neurofibrillary tangles and neurofilaments share antigenic determinants. Nature 298:84–86
Anniko M, Thornell L-E, Gustavsson H, Virtanen I (1986) Intermediate filaments in the newborn inner ear of the mouse. ORL 48:98–106
Ballantyne J, Engstrom H (1969) Morphology of the vestibular ganglion cells. J Laryngol Otol 83:19–42
Berglund AM, Ryugo DK (1986) A monoclonal antibody labels type II neurons of the spiral ganglion. Brain Res 383:327–332
Dahl D (1983) Immunohistochemical differences between neurofilaments in perikarya, dendrites and axons. Exp Cell Res 149:397–408
Debus E, Weber K, Osborn M (1983) Monoclonal antibodies specific for glial fibrillary acidic (GFA) protein and for each of the neurofilament triplet polypeptides. Differentiation 25: 193–203
Geisler N, Weber K (1981) Self-assembly in vitro of the 68,000 molecular weight component of the mammalian neurofilament triplet proteins into intermediate-sized filaments. J Mol Biol 151:565–571
Hoffman PN, Thompson GW, Griffin JW, Price DL (1985) Changes in neurofilament transport coincide temporally with alterations in the caliber of axons in regenerating motor fibers. J Cell Biol 101:1332–1340
Kellerhals B, Engstrom H, Ades HW (1967) Die Morphologie des Ganglion Spirale cochleae. Acta Otolaryngol (Stockh) 226:1–78
Lasek RJ, Hoffman PN (1976) The neuronal cytoskeleton, axonal transport and axonal growth. In: Goldman R, Pollard T, Rosenbaum J (eds) Cell mobility. Book C. Microtubules and related proteins. Cold Spring Harbor Conference on Cell Proliferation, vol 3. Cold Spring Harbor Laboratory, New York, pp 1021–1049
Lazarides E (1980) Intermediate filaments as mechanical integrators of cellular space. Nature 283:249–265
Mencarelli C, Bugnoli M, Contorni M, Moscatelli A, Ruggiero P, Palluni V (1987) Phosphorylated epitopes of neurofilaments have been conserved during chordate evoluation. Biochem Biophys Res Commun 149:807–814
Price J (1985) An immunohistochemical and quantitative examination of dorsal root ganglion neuronal subpopulations. J Neurosci 5:2051–2059
Richter E, Spoendlin H (1981) Scarpa's ganglion in the cat. Acta Otolaryngol (Stockh) 92:423–436
Romand R, Hafidi A, Despres G (1988) Immunocytochemical localization of neurofilament protein subunits in the spiral ganglion of the adult rat. Brain Res 462:167–173
Rosenbluth J (1962) The fine structure of acoustic ganglia in the rat. J Cell Biol 12:329–359
Rosenfeld J, Dorman ME, Griffin JW, Sternberger LA, Sternberger NH, Price DL (1987) Distribution of neurofilament antigens after axonal injury. J Neuropathol Exp Neurol 46:269–282
Spassova I (1982) Fine structure of the neurones and synapsis of the vestibular ganglion of the cat. J Hirnforsch 23:652–669
Spoendlin H (1971) Degeneration behaviour of the cochlear nerve. Arch Klin Exp Ohren- Nasen- Kehlkopfheilkd 200: 275–291
Sternberger LA, Sternberger NH (1983) Monoclonal antibodies distinguish phosphorylated and nonphosphorylated forms of neurofilaments in situ. Proc Natl Acad Sci USA 80:6126–6130
Sternberger NH, Sternberger LA, Ulrich J (1985) Aberrant neurofilament phosphorylation in Alzheimer disease. Proc Natl Acad Sci USA 82:4274–4276
Thornell L-E, Anniko M, Virtanen I (1987) Cytoskeletal organization of the human inner ear. I. Expression of intermediate filaments in vestibular organs. Acta Otolaryngol (Stockh) 437:5–27
Troncoso JC, Sternberger NH, Sternberger LA. Hoffman PL, Price DL (1986) Immunocytochemical studies of neurofilament antigens in the neurofibrillary pathology induced by aluminium. Brain Res 364:295–300
Virtanen I, Miettinen M, Lehto V-P, Kariniemi A-L, Paasivuo R (1985) Diagnostic application of monoclonal antibodies to intermediate filaments. Ann Sci 455:635–648
Ylikoski J, Eränkö L, Päivärinta H (1984) Substance P-like immunoreactivity in the rabbit inner ear. J Laryngol Otol 98:759–765
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Ylikoski, J., Pirvola, U. & Virtanen, I. Expression of neurofilament proteins in the vestibular ganglion cells of the rat. Eur Arch Otorhinolaryngol 247, 211–214 (1990). https://doi.org/10.1007/BF00178986
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00178986