Abstract
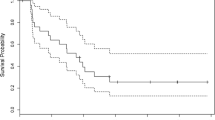
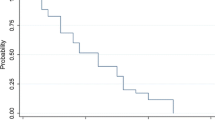
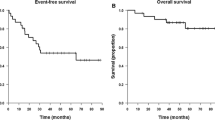
Twenty-four patients with recurrent malignant glioma were treated with intravenous BCNU (80 mg/m2/day × 3 days) alternating with AZQ (8 mg/m2/day × 5 days) every 6–8 weeks. Twenty patients received two or more courses of chemotherapy, ten anaplastic astrocytomas (AA), eight glioblastomas (GBM), and two malignant oligodendrogliomas (Oligo). All had prior surgery and irradiation; one had prior chemotherapy. Median age was 37.5 years. The median Zubrod performance status (PS) was 1. Three patients (15%) achieved response status, and 7 (35%) had stable disease with median times to tumor progression (MTP) of 56 wks and 35 wks. MTP for patients with progression was 11 weeks. No GBM was responsive to chemotherapy and none of the ten patients with stable or responsive disease were older than fifty years. Dose limiting toxicity was consisted of thrombocytopenia and leukopenia. Young patients with recurrent AA and good PS appear more likely to respond to alternating BCNU/AZQ chemotherapy. The overall response rate (response plus stable) of 50% was comparable to that of BCNU alone and the hematologic toxicity was cumulative.
Similar content being viewed by others
References
Green SB, Byar DP, Walker MD, et al: Comparisons of carmutine, procarbazine, and high-dose methylprednisolone as additions to surgery and radiotherapy for the treatment of malignant glioma. Cancer Treatment Report 67: 121–132, 1983
Bigner DD, Schold SC, Bigner SH, Bullard DE, et al: How heterogeneous are gliomas? Cancer Treatment Reports 2: 45–49, 1981
Yung WKA, Shapiro JR, Shapiro WR: Heterogeneous chemosensitivities of subpopulations of human glioma cells in culture. Cancer Res 42: 992–998, 1982
Schold SC, Friedman HS, Bjornsson T, et al: Treatment of patients with recurrent primary brain tumors with AZQ. Neurology 34: 615–619, 1984
Taylor SA, McCracken JD, Eyne HJ, et al: Phase II study of AZQ in patients with central nervous system malignancies: A Southwest Oncology Group Study. J Neurooncol 3: 131–135, 1985
Fenn LG, Yung WKA, Leavens ME, et al: A Phase II trial of 2,5-diaziridinyl 3,6-bis(carboethoxy amino) 1,4-benzoquinone in recurrent primary brain tumors. J Neuro-oncol 2: 13–17, 1984
Haid M, Khandekar JD, Christ M, et al: Aziridinylbenzoquinone in recurrent, progressive glioma of the central nervous system: A Phase II study by the Illinois Cancer Council. Cancer 56: 1311–1315, 1985
Maral J, Poisson M, Pertuiset BF, et al: Phase II evaluation of diaziquone in the treatment of human malignant glioma. J Neuro-oncol 3: 245–250, 1985
Wilson CB, Gutin N, Boldrey EB, et al: Single-agent chemotherapy of brain tumors. A five-year review. Arch Neurol 33: 739–744, 1976
Edward MS, Levin VA, Wilson CB: Brain tumor chemotherapy: An evaluation of agents in current use for Phase II and III trials. Cancer Treatment Reports 64: 1179–1205, 1980
Bjornsson TD, Schold SC, Friedman HS, et al.: Pharmacokinetics of diaziquone after three different dosage regimens. Cancer Treatment Report 69: 1383–1385, 1985
Curt GA, Kelley JA, Kufta CV, et al: Phase II and pharmacokinetic study of AZQ in high grade gliomas. Cancer Res 43: 6102–6105, 1983
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Yung, W., Harris, M., Bruner, J. et al. Intravenous BCNU and AZQ in patients with recurrent malignant gliomas. J Neuro-Oncol 7, 237–240 (1989). https://doi.org/10.1007/BF00172916
Issue Date:
DOI: https://doi.org/10.1007/BF00172916