Abstract
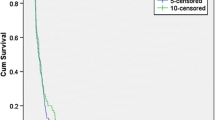
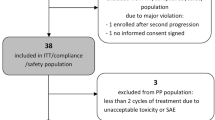
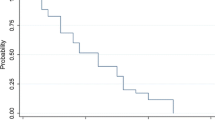
There is no standard therapy for recurrent anaplastic glioma (AG). Salvage therapies include alkylator-based chemotherapy, re-resection with or without carmustine implants, re-irradiation and bevacizumab. Bendamustine is a novel bifunctional alkylator with CNS penetration never previously evaluated in AG. Assess response and toxicity of bendamustine in recurrent AG in a phase II trial. Adults with radiation and temozolomide refractory recurrent AG were treated with bendamustine. A cycle of bendamustine was defined as two consecutive days of treatment (100 mg/m2/day) administered once every 4 weeks. Success of treatment was defined as progression free survival (PFS) at 6 months of 40 % or better. Twenty-six adults [16 males; 10 females: median age 40 years (range 30–65)] were treated, 12 at first recurrence and 17 at second recurrence. Prior salvage therapy included re-resection (14), chemotherapy (11) and re-radiation (2). Grade 3 treatment-related toxicities included lymphopenia (11 patients; Grade 4 in 3), myalgia, pneumonia, diarrhea, leukopenia, allergic reaction and thrombocytopenia in one patient each. One patient discontinued therapy due to toxicity. There were five instances of bendamustine dose delays all due to lymphopenia. There were no dose reductions due to toxicity. The median number of cycles of therapy was 3 (range 1–8). Best radiographic response was progressive disease in 12 (46 %), stable disease in 13 (50 %) and partial response in 1 (4 %). Median, 6- and 12-month PFS was 2.7 months (range 1–52), 27 and 8 % respectively. In patients with recurrent AG refractory to Z, bendamustine has manageable toxicity and modest single agent activity though not meeting pre-specified study criteria.


Similar content being viewed by others
References
Louis DN, Ohgaki H, Wiestler OD, Cavenee WK et al (2007) WHO classification of tumors of the central nervous system. Acta Neuropathol. doi:10.1007/s00401-007-0243-4
Louis DN, Perry A, Burger P, Ellison DW, Reifenberger G, von Deimling A et al (2014) International Society of Neuropathology—s. Brain Pathol Zurich Switz 24:429–435
Eckel-Passow JE, Lachance DH, Molinaro AM, Walsh KM, Decker PA, Sicotte H et al (2015) Glioma groups based on 1p/19q, IDH, and TERT promoter mutations in tumors. N Engl J Med 372:2499–2508
Suzuki H, Aoki K, Chiba K, Sato Y, Shiozawa Y, Shiraishi Y et al (2015) Mutational landscape and clonal architecture in grade II and III gliomas. Nat Genet 47:458–468
Reuss DE, Sahm F, Schrimpf D, Wiestler B, Capper D, Koelsche C et al (2015) ATRX and IDH1-R132H immunohistochemistry with subsequent copy number analysis and IDH sequencing as a basis for an “integrated” diagnostic approach for adult astrocytoma, oligodendroglioma and glioblastoma. Acta Neuropathol 129:133–146
Ellison DW (2015) Multiple molecular data sets and the classification of adult diffuse gliomas. N Engl J Med 372(26):2555–2557
Brat DJ, Verhaak RG et al (2015) Comprehensive, integrative genomic analysis of diffuse lower-grade gliomas. N Engl J Med 372(26):2481–2498
Ceccarelli M, Barthel FP, Malta et al (2016) Molecular profiling reveals biologically discrete subsets and pathways of progression in diffuse glioma. Cell 164(3):550–563
Van den Bent MJ et al (2013) Adjuvant procarbazine, lomustine, and vincristine chemotherapy in newly diagnosed anaplastic oligodendroglioma: long-term follow-up of EORTC brain tumor group study 26951. J Clin Oncol 31:344–350
Cairncross G et al (2013) Phase III trial of chemoradiotherapy for anaplastic oligodendroglioma: long-term results of RTOG 9402. J Clin Oncol 31:337–343
Cairncross JG, Wang M, Jenkins RB et al (2014) Benefit from procarbazine, lomustine, and vincristine in oligodendroglial tumors Is associated with mutation of IDH. J Clin Oncol 32:783–790
Weller M, van den Bent M, Hopkins K et al (2014) EANO guideline for the diagnosis and treatment of anaplastic gliomas and glioblastoma. Lancet Oncol 15:e395–e403
Wick W, Wiestler B, Platten M (2015) Treatment of anaplastic glioma. Cancer Treat Res 163:89–101
Wick W, Hartmann C, Engel C et al (2009) NOA-04 randomized phase III trial of sequential radiochemotherapy of anaplastic glioma with procarbazine, lomustine and vincristine or temozolomide. J Clin Oncol 27:5874–5880
Perry JR, Belanger K, Mason WP et al (2010) Phase II trial of continuous dose-intense temozolomide in recurrent malignant glioma: RESCUE study. J Clin Oncol 28:2051–2057
Yung WK, Prados MD, Yaya-Tur R et al (1999) Multicenter phase II trial of temozolomide in patients with anaplastic astrocytoma or anaplastic oligoastrocytoma at first relapse, temodal brain tumor group. J Clin Oncol 17:2762–2771
Jaeckle KA, Hess KR, Yung WK et al (2003) Phase II evaluation of temozolomide and 13-cis-retinoic acid for the treatment of recurrent and progressive malignant glioma: a North American brain tumor consortium study. J Clin Oncol 1:2305–2311
Nabors LB, Ammirati M, Bierman PJ et al (2013) Central nervous system cancers. J Natl Compr Cancer Netw 11(9), 1114–1151
Brem H, Piantadosi S, Burger PC et al (1995) Placebo-controlled trial of safety and efficacy of intraoperative controlled delivery by biodegradable polymers of chemotherapy for recurrent gliomas. Lancet 345:1008–1012
Combs SE, Thilmann C, Edler L et al (2005) Efficacy of fractionated stereotactic reirradiation in recurrent gliomas: long-term results in 172 patients treated in a single institution. J Clin Oncol 23:8863–8869
Fogh SE, Andrews DW, Glass J et al (2010) Hypofractionated stereotactic radiation therapy: an effective therapy for recurrent high-grade gliomas. J Clin Oncol 28:3048–3053
Chamberlain MC, Johnston S (2009) Salvage chemotherapy with bevacizumab for recurrent alkylator-refractory anaplastic astrocytoma. J Neuro-Oncol 91:359–367
Kreisl TN, Zhang W, Odia Y et al (2011) A phase II trial of single-agent bevacizumab in patients with recurrent anaplastic glioma. Neuro-Oncology 13:1143–1150
Chamberlain MC (2015) Salvage therapy with lomustine for temozolomide refractory recurrent anaplastic astrocytoma: a retrospective study. J Neuro-Oncol 122:329–338
Cheson BD, Rummel MJ (2009) Bendamustine: rebirth of an old drug. J Clin Oncol 27:1492–1501
Keating MJ, Bach C, Yasothan U, Kirkpatrick P (2008) Bendamusttine. Nat Rev Drug Discov 7(6):473–474
Leoni LM, Bailey B, Reifert J et al (2008) Bendamustine (treanda) displays a distinct pattern of cytotoxicity and unique mechanistic features compared with other alkylating agents. Clin Cancer Res 14(1):309–317
Bendamustine hydrochloride (CEP-18083) Investigators brochure, Cephalon Edition 2. Release date: March 4, 2010
Chamberlain MC, Johnston SA (2011) Salvage chemotherapy with single agent bendamustine for recurrent glioblastoma. J Neuro-Oncol 105(3):523–530
Chamberlain MC (2014) Salvage therapy with bendamustine for methotrexate refractory recurrent primary CNS lymphoma: a retrospective case series. J Neuro-Oncol 18(1):155–162
MacDonald DR, Cascino TL, Schold SC et al (1990) Response criteria for phase II studies of supratentorial malignant glioma. J Clin Oncol 8:1277–1280
Wong ET, Hess KR, Gleason MJ et al (1999) Outcomes and prognostic factors in recurrent glioma patients enrolled onto phase II clinical trials. J Clin Oncol 17:2572–2578
Fine HA, Wen PY, Maher EA, Viscosi E, Batchelor T, Lakhani N, Figg WD, Purow BW, Borkowf CB (2003) Phase II trial of thalidomide and carmustine for patients with recurrent high-grade gliomas. J Clin Oncol 21(12):2299–2304
Acknowledgments
The authors want to thank Sandra Johnston and Alisa Clein for their organizational and administrative assistance in developing the study and manuscript.
Disclosure
MC Chamberlain and Bryan Kim collected and analyzed data. No personal communications cited in the manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors report no conflicts of interest.
Rights and permissions
About this article
Cite this article
Chamberlain, M.C., Colman, H., Kim, B.T. et al. Salvage therapy with bendamustine for temozolomide refractory recurrent anaplastic gliomas: a prospective phase II trial. J Neurooncol 131, 507–516 (2017). https://doi.org/10.1007/s11060-016-2241-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11060-016-2241-7