Abstract
Background
Previously we described the outcome of children with spinal cord astrocytoma treated with irinotecan and cisplatin (I/C). We here report the review of the initial institutional experience using this combination for children with low-grade glioma (LGG).
Procedure
I/C chemotherapy consisted of weekly cisplatin (30 mg/m2) and irinotecan (50–65 mg/m2) for a total maximum of 16 doses, administered in an outpatient basis.
Results
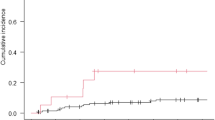
Between November 2002 and December 2009, 46 children (median age 6.3 years; range 0.3–17.7) with glioma were treated. We here report the cohort of 31 patients with LGG. Patients received a median of 16 cycles of I/C (range 8–16). The overall objective response [complete response (CR) + partial response (PR)] and disease control (CR + PR + stable disease) rates to I/C treatment were 6.5% [95% confidence interval (CI), 0.8–21.4%] and 93.5% (95% CI 78.6–99.2%), respectively. Disease control persisted for a median of 65 months. Toxicity was predominantly myelosuppression only seen in heavily pretreated patients. Survival analysis shows 5-year event-free survival (EFS) of 54% and 5-year overall survival (OS) of 80%.
Conclusion
I/C chemotherapy produced disease control and clinical improvement in a majority of children with low-grade glioma, with manageable toxicity.

Similar content being viewed by others
References
Ries LAG, Melbert D, Krapcho M, et al. (2008) SEER statistics review, 1975–2005. National Cancer Institute, Bethesda. Available at http://www.cbtrus.org.
Davis FG, Kupelian V, Freels S, McCarthy B, Surawicz T. Prevalence estimates for primary brain tumors in the United States by behavior and major histology groups. Neuro Oncol. 2001;3:152–8.
Louis DN, Ohgaki H, Wiestler OD, Webster WK. WHO classification of tumours of the central nervous system. Lyon: IARC; 2016.
Sturm D, Pfister SM, Jones DTW. Pediatric gliomas: current concepts on diagnosis, biology, and clinical management. J Clin Oncol. 2017;35:2370–7.
Gnekow AK, Falkenstein F, von Hornstein S, Zwiener I, Berkefeld S, Bison B, et al. Long-term follow-up of the multicenter, multidisciplinary treatment study HIT-LGG-1996 for low-grade glioma in children and adolescents of the German Speaking Society of Pediatric Oncology and Hematology. Neuro Oncol. 2012;14:1265–84.
Qaddoumi I, Sultan I, Broniscer A. Pediatric low-grade gliomas and the need for new options for therapy: why and how? Cancer Biol Ther. 2009;8:4–10.
Mora J, Cruz O, Gala S, Navarro R. Successful treatment of childhood intramedullary spinal cord astrocytomas with irinotecan and cisplatin. Neuro Oncol. 2007;9:39–46.
http://evs.nci.nih.gov/ftp1/CTCAE/CTCAE_4.03_2010-06-14_QuickReference_5x7.pdf
R Core Team (2015) R: A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna. URL https://www.R-project.org/. Accessed 15 Jan 2016.
Garzón M, García-Fructuoso G, Guillén A, Suñol M, Mora J, Cruz O. Low-grade gliomas in children. Single institutional experience in 198 cases. Childs Nerv Syst. 2015;31:1447–59.
Ater JL, Zhou T, Holmes E, Mazewski CM, Booth TN, Freyer DR, et al. Randomized study of two chemotherapy regimens for treatment of low-grade glioma in young children: a report from the Children’s Oncology Group. J Clin Oncol. 2012;30:2641–7.
Bouffet E, Jakacki R, Goldman S, Hargrave D, Hawkins C, Shroff M, et al. Phase II study of weekly vinblastine in recurrent or refractory pediatric low-grade glioma. J Clin Oncol. 2012;30:1358–63.
Gururangan S, Fisher MJ, Allen JC, Herndon JE 2nd, Quinn JA, Reardon DA, et al. Temozolomide in children with progressive low-grade glioma. Neuro Oncol. 2007;9:161–8.
Jakacki RI, Bouffet E, Adamson PC, Pollack IF, Ingle AM, Voss SD, et al. A phase 1 study of vinblastine in combination with carboplatin for children with low-grade gliomas: a Children’s Oncology Group phase 1 consortium study. Neuro Oncol. 2011;13:910–5.
Ronghe M, Hargrave D, Bartels U, Tabori U, Vaidya S, Chandler C, et al. Vincristine and carboplatin chemotherapy for unresectable and/or recurrent low-grade astrocytoma of the brainstem. Pediatr Blood Cancer. 2010;55:471–7.
Scheinemann K, Bartels U, Tsangaris E, Hawkins C, Huang A, Dirks P, et al. Feasibility and efficacy of repeated chemotherapy for progressive pediatric low-grade gliomas. Pediatr Blood Cancer. 2011;57:84–8.
Lassaletta A, Scheinemann K, Zelcer SM, Hukin J, Wilson BA, Jabado N, et al. Phase II weekly vinblastine for chemotherapy-naïve children with progressive low-grade glioma: a Canadian Pediatric Brain Tumor Consortium study. J Clin Oncol. 2016;34:3537–43.
Krishnatry R, Zhukova N, Guerreiro Stucklin AS, Pole JD, Mistry M, Fried I, et al. Clinical and treatment factors determining long-term outcomes for adult survivors of childhood low-grade glioma: a population-based study. Cancer. 2016;122:1261–9.
Jagasia MH, Langer CJ, Johnson DH, Yunus F, Rodgers JS, Schlabach LL, et al. Weekly irinotecan and cisplatin in advanced non-small cell lung cancer: a multicenter phase II study. Clin Cancer Res. 2001;7:68–73.
Yancey A, Harris MS, Egbelakin A, Gilbert J, Pisoni DB, Renbarger J. Risk factors for cisplatin-associated ototoxicity in pediatric oncology patients. Pediatr Blood Cancer. 2012;59:144–8.
Brock PR, Knight KR, Freyer DR, Campbell KC, Steyger PS, Blakley BW, et al. Platinum-induced ototoxicity in children: a consensus review on mechanisms, predisposition, and protection, including a new International Society of Pediatric Oncology Boston ototoxicity scale. J Clin Oncol. 2012;30:2408–17.
Coradini PP, Cigana L, Selistre SG, Rosito LS, Brunetto AL. Ototoxicity from cisplatin therapy in childhood cancer. J Pediatr Hematol Oncol. 2007;29:355–60.
Bertolini P, Lassalle M, Mercier G, Raquin MA, Izzi G, Corradini N, et al. Platinum compound-related ototoxicity in children: long-term follow-up reveals continuous worsening of hearing loss. J Pediatr Hematol Oncol. 2004;26:649–55.
Kushner BH, Budnick A, Kramer K, Modak S, Cheung NK. Ototoxicity from high dose use of platinum compounds in patients with neuroblastoma. Cancer. 2006;107:417–22.
Skinner R, Parry A, Price L, Cole M, Craft AW, Pearson AD. Persistent nephrotoxicity during 10-year follow-up after cisplatin or carboplatin treatment in childhood: relevance of age and dose as risk factors. Eur J Cancer. 2009;45:3213–9.
Hare CB, Elion GB, Houghton PJ, Houghton JA, Keir S, Marcelli SL, et al. Therapeutic efficacy of the topoisomerase I inhibitor 7–ethyl–10–(4–[1–piperidino]–1– piperidino)-carbonyloxy-camptothecin against pediatric and adult central nervous system tumor xenografts. Cancer Chemother Pharmacol. 1997;39:187–91.
Turner CD, Gururangan S, Eastwood J, Bottom K, Watral M, Beason R, et al. Phase II study of irinotecan (CPT-11) in children with high-risk malignant brain tumors: the duke experience. Neuro Oncol. 2002;4:102–8.
Souid AK, Dubowy RL, Blaney SM, Hershon L, Sullivan J, McLeod WD, et al. Phase I clinical and pharmacologic study of weekly cisplatin and irinotecan combined with amifostine for refractory solid tumors. Clin Cancer Res. 2003;9:703–10.
Couec ML, André N, Thebaud E, Minckes O, Rialland X, Corradini N, et al. Bevacizumab and irinotecan in children with recurrent or refractory brain tumors: toxicity and efficacy trends. Pediatr Blood Cancer. 2012;59:34–8.
Hwang EI, Jakacki RI, Fisher MJ, Kilburn LB, Horn M, Vezina G, et al. Long-term efficacy and toxicity of bevacizumab based therapy in children with recurrent low-grade gliomas. Pediatr Blood Cancer. 2013;60:776–82.
Gururangan S, Chi SN, Young Poussaint T, Onar-Thomas A, Gilbertson RJ, Vajapeyam S, et al. Lack of efficacy of bevacizumab plus irinotecan in children with recurrent malignant glioma and diffuse brainstem glioma: a Pediatric Brain Tumor Consortium study. J Clin Oncol. 2010;28:3069–75.
Doireau V, Grill J, Zerah M, Lellouch-Tubiana A, Couanet D, Chastagner P, et al. Chemotherapy for unresectable and recurrent intramedullary glial tumours in children. Brain Tumours Subcommittee of the French Society of Paediatric Oncology (SFOP). Br J Cancer. 1999;81:835–84.
Massimino M, Spreafico F, Cefalo G, Riccardi R, Tesoro-Tess JD, Gandola L, et al. High response rate to cisplatin/etoposide regimen in childhood low-grade glioma. J Clin Oncol. 2002;20:4209–16.
Packer RJ, Ater J, Allen J, Phillips P, Geyer R, Nicholson HS, et al. Carboplatin and vincristine chemotherapy for children with newly diagnosed and progressive low-grade gliomas. J Neurosurg. 1997;86:747–54.
Mahoney DH Jr, Cohen ME, Friedman HS, Kepner JL, Gemer L, Langston JW, et al. Carboplatin is effective therapy for young children with progressive optic pathway tumors: a Pediatric Oncology Group phase II study. Neuro Oncol. 2001;2:213–20.
Massimino M, Spreafico F, Riva D, Biassoni V, Poggi G, Solero C, et al. A lower-dose, lower-toxicity cisplatin and etoposide regimen for childhood progressive low-grade glioma. J Neuro Oncol. 2010;100:65–71.
Fisher MJ, Loguidice M, Gutmann DH, Listernick R, Ferner RE, Ullrich NJ, et al. Visual outcomes in children with neurofibromatosis type 1-associated optic pathway glioma following chemotherapy: a multicenter retrospective analysis. Neuro Oncol. 2012;14:790–7.
Moreno L, Bautista F, Ashley S, Duncan C, Zacharoulis S. Does chemotherapy affect the visual outcome in children with optic pathway glioma? A systematic review of the evidence. Eur J Cancer. 2010;46:2253–9.
Gnekow AK, Walker DA, Kandels D, Picton S, Perilongo G, Grill J, et al. A European randomised controlled trial of the addition of etoposide to standard vincristine and carboplatin induction as part of an 18-month treatment programme for childhood (≤16 years) low grade glioma—a final report. Eur J Cancer. 2017;81:206–25.
Ater JL, Xia C, Mazewski CM, Booth TN, Freyer DR, Packer RJ, et al. Nonrandomized comparison of neurofibromatosis type 1 and non-neurofibromatosis type 1 children who received carboplatin and vincristine for progressive low-grade glioma: a report from the Children’s Oncology Group. Cancer. 2016;122:1928–36.
Acknowledgements
The authors thank the nursing and multidisciplinary staff for their support in the patient care.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
All procedures performed were in accordance with the ethical standards of the institution research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards. For this retrospective study specific consent was not required.
Rights and permissions
About this article
Cite this article
Mora, J., Perez-Jaume, S. & Cruz, O. Treatment of childhood astrocytomas with irinotecan and cisplatin. Clin Transl Oncol 20, 500–507 (2018). https://doi.org/10.1007/s12094-017-1741-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12094-017-1741-z