Summary
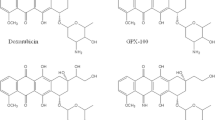
Thirty one patients with solid tumors were entered into a phase I trial with idarubicin, a new anthracycline antibiotic with oral antitumor activity in animals. The drug was scheduled to be given for 4 consecutive weeks at doses ranging from 10 to 20 mg/m2. Leukopenia was the dose-limiting toxicity. Thrombocytopenia was occasionally seen. Since several patients could not receive the third and fourth administrations of the drug at 17.5 and 20 mg/m2, higher doses were administered only for 2 consecutive weeks. With this schedule, the maximum tolerated dose was 25 mg/m2 and leukopenia was again the dose-limiting toxicity. With both schedules, myelosuppression was highly variable and could not be related to prior therapy, bone or liver metastases, or performance status. Other toxicities were mild to moderate and were dominated by nausea and vomiting which were observed in 29% of the patients. Alopecia and mucositis were unfrequent and cardiac toxicity was not observed. Starting doses of 15 mg/m2 for 4 consecutive weeks or 20 mg/m2 for 2 consecutive weeks could be proposed for oral phase II studies with idarubicin, under careful pharmacokinetic monitoring.
Similar content being viewed by others
References
Young RC, Ozols RF, Myers CE: The anthracycline antineoplastic drugs. N Engl J Med 305:139–153, 1981
Von Hoff DD, Layard MW, Basa P, Davis HL, Von Hoff AL, Rozencweig M, Muggia FM: Risk factors for doxorubicin-induced congestive heart failure. Ann Intern Med 91:710–717, 1979
Arcamone F, Bernardi L, Giardino P, Patelli B, Di Marco A, Casazza AM, Pratesi G, Reggiani P: Synthesis and antitumor activity of 4-demethoxydaunorubicin, 4-deme-thoxy-7,9-diepidaunorubicin, and their beta anomers. Cancer Treat Rep 60:549–564, 1980
Casazza AM, Pratesi G, Giuliani F, Di Marco A: Antileukemic activity of 4-demethoxydaunorubicin in mice. Tumori 66:829–834, 1976
Sviluppo RE: IMI30. 4-demethoxydaunorubicin. Farmitalia Carlo Erba Clinical Brochure, 1980
Casazza AM: Experimental evaluation of anthracycline analogs. Cancer Treat Rep 63:835–844, 1979
Berman E, Wittes RE, Leyland-Jones B, Casper ES, Gralla RJ, Howard J, Williams L, Baratz R, Young CW: Phase I and clinical pharmacology studies of intravenous and oral administration of 4-demethoxydaunorubicin in patients with advanced cancer. Cancer Res 43:6096–6101, 1983
Bonfante V, Ferrari L, Villani F, Bonadonna G: Phase I study of 4-demethoxydaunorubicin. Invest N Drugs 1:161–168, 1983
Kaplan S, Martini A, Varini M, Togni P, Cavalli F: Phase I trial of 4-demethoxydaunorubicin with single iv doses. Eur J Cancer Clin Oncol 18:1303–1306, 1982
Kaplan S, Willems Y, Sessa C, Pacciarini MA, Tamassia V, Cavalli F: Phase I trial of 4-demethoxydaunorubicin (idarubicin) with single oral doses. Invest N Drugs 2:281–286, 1984
Daghestani A, Arlin ZA, Gee TS, Kempin S, Mertelsmann R, Budman D, Schulman P, Cassidy C, Raymond V, Clarkson BD, Young CW: Treatment of acute leukemia in relapse with 4-demethoxydaunorubicin. Proc Am Soc Clin Oncol 2:181, 1983
Torti MT, Bristow MR, Howes AE, Aston D, Stockdale FE, Carter SK, Kohler M, Brown BW, Billingham ME: Reduced cardiotoxicity of doxorubicin delivered on a weekly schedule. Ann Intern Med 99:745–749, 1983
WHO Handbook for reporting results of cancer treatment. WHO, Geneva, 1979
Rozencweig M, Crespeigne N, Nicaise C, Gerard B, Atassi G, Tagnon H, Kenis Y: Phase I trial of 1,2,4-triglycidylurazol (TGU, NSC 332488) with a daily x 5 schedule. Proc Am Assoc Cancer Res 25:201, 1984
Dodion P, Davis TA, Rozencweig M, Watthieu M, Crespeigne N, Beer M, Bachur NR: Human pharmacokinetics of 4-demethoxydaunorubicin. Invest N Drugs 2:119, 1984
Dodion P: Clinical pharmacology in oncology. Recent advances. Eur J Cancer Clin Oncol 19:1499–1507, 1983
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Dodion, P., Finet, C., Crespeigne, N. et al. Phase I study of oral idarubicin given with a weekly schedule. Invest New Drugs 4, 31–38 (1986). https://doi.org/10.1007/BF00172013
Issue Date:
DOI: https://doi.org/10.1007/BF00172013