Abstract
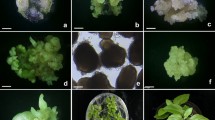
Cell layer strips composed of the epidermis and 7–9 layers of subepidermal cells were isolated from the 3–4 terminal internodes of Brassica napus cv Westar plants at the early flowering stage. The strips were precultured for one day in modified liquid MS [11] medium and subsequently incubated for 17–18 h in a 0.4 M mannitol solution containing 1% Macerozyme and 1% Cellulase ‘Onozuka’ R-10. Protoplast yield was 2–2.8×106 per 1.0g of tissue. Protoplasts were cultured at 1×105/ml in three different media: S1 [13], B [12] and L[8]. The first cell divisions occurred after 2–8 days of culture at frequencies of 20–54%. The highest growth rate of colonies was obtained in L medium containing 0.4 M sucrose and 2% Ficoll. After 4 weeks, green calli, 1–2 mm in diameter were transferred onto B5 [2] medium with 3 mgl-1 zeatin, 1% sucrose, 0.1 M mannitol and 0.5% agarose for shoot regeneration. Up to 20% of the calli regenerated shoots which subsequently were rooted and established in soil in the greenhouse.
Similar content being viewed by others
References
Chuong PV, Pauls KP, Beversdorf WD (1985) A simple culture method for Brassica hypocotyl protoplasts. Plant Cell Rep 4:4–6
Gamborg OL, Miller RA, Ojima K (1968) Nutrient requirements of suspension cultures of soybean root cells. Exp Cell Res 50:151–158
Glimelius K (1984) High growth rate and regeneration capacity of hypocotyl protoplasts in some Brassicaceae. Physiol Plant 61:38–44
Kartha KK, Michayluk MR, Kao KN, Gamborg OL, Constabel F (1974) Callus formation and plant regeneration from mesophyll protoplasts of rape plants (Brassica napus L. cv. Zephyr). Plant Sci Lett 3:265–271
Klimaszewska K, Keller WA (1985) High frequency plant regeneration from thin cell layer explants of Brassica napus. Plant Cell Tissue Organ Culture 4:183–197
Kohlenbach HW, Körber M, Li L-C (1982) Cytodifferentiation of protoplasts isolated from a stem embryo system of Brassica napus to trachery elements. Z Pflanzenphysiol 107:367–371
Li L-C, Kohlenbach HW (1982) Somatic embryogenesis in quite a direct way in cultures of mesophyll protoplasts of Brassica napus L. Plant Cell Rep 1:209–211
Lichter R (1981) Anther culture of Brassica napus in a liquid culture medium. Z Pflanzenphysiol 103:229–237
Lörz H, Larkin PJ, Thomson J and Scowcroft WR (1983) Improved protoplast culture on agarose media. Plant Cell Tissue Organ Culture 2:217–226
Lu DY, Pental D, Cocking EC (1982) Plant regeneration from seedling cotyledon protoplasts. Z Pflanzenphysiol 107:59–63
Murashige T, Skoog F (1962) A revised medium for rapid growth and bioassays with tobacco cultures. Physiol Plant 15:473–497
Pelletier G, Primard C, Vedel F, Cherit P, Remy R, Rousselle P., Renard M (1983) Intergeneric cytoplasmic hybridization in Cruciferae by protoplast fusion. Mol gen Genet 191:244–250
Schenck HR, Hoffmann F (1979) Callus and root regeneration from mesophyll protoplasts of basic Brassica species: B. campestris, B. oleracea and B. nigra. Z Pflanzenzüchtg 82:354–360
Schenck HR, Röbbelen G (1982) Somatic hybrids by fusion of protoplasts from Brassica oleracea and B. campestris. Z Pflanzenzüchtg 89:278–288
Shillito RD, Paszkowski J, Potrykus J (1983) Agarose plating and a bead type culture technique to enable and stimulate development of protoplast-derived colonies in a number of plant species. Plant Cell Rep 2:244–247
Thomas E, Hoffmann F, Potrykus J, Wenzel G (1976) Protoplast regeneration and stem embryogenesis of haploid androgenetic rape. Molec gen Genet 145:245–247
Xu Z-H, Davey MR, Cocking EC (1982) Plant regeneration from root protoplasts of Brassica. Plant Sci Lett 24:117–121
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Klimaszewska, K., Keller, W.A. Plant regeneration from stem cortex protoplasts of Brassica napus . Plant Cell Tiss Organ Cult 8, 225–233 (1987). https://doi.org/10.1007/BF00040949
Received:
Revised:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00040949