Abstract
Introduction
Synthesis of evidence on the long-term use of first-line biologic therapy in patients with early rheumatoid arthritis (RA) is required. We compared the efficacy of up to 5 years’ treatment with first-line tumor necrosis factor inhibitors (TNFis) versus other treatment strategies in this population.
Methods
Previous systematic reviews, PubMed and the Cochrane Central Register of Controlled Trials were searched for randomized controlled trials (RCTs) involving treatment of methotrexate-naïve RA patients with first-line TNFis. Literature was synthesized qualitatively, and a meta-analysis conducted to evaluate American College of Rheumatology (ACR) responses, clinical remission defined by any standard measure, and Health Assessment Questionnaire Disability Index (HAQ) at Years 2 and/or 5.
Results
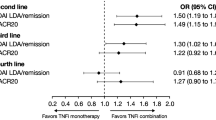
Ten RCTs involving 4306 patients [first-line TNFi, n = 2234; other treatment strategies (control), n = 2072] were included in the meta-analysis. Three studies were double-blind for the first 2 years, while seven were partly/completely open label during this period. Five studies reported data at Year 5; all were open label at this time point. At Year 2, ACR50 response, ACR70 response and remission rates were significantly improved with first-line TNFi versus control in double-blind RCTs [log-odds ratio (OR) 0.32 [95% confidence interval (CI) 0.02, 0.62; p = 0.035], log-OR 0.48 (95% CI 0.20, 0.77; p = 0.001), and log-OR 0.44 (95% CI 0.13, 0.74; p = 0.005), respectively], but not in open-label studies. No significant between-group differences were observed in mean HAQ at Year 2 in double-blind or open-label RCTs or in ACR response or remission outcomes at Year 5.
Conclusion
In double-blind studies, 2-year efficacy outcomes were significantly improved with first-line TNFi versus other treatment strategies in patients with MTX-naïve RA. No significant differences in these outcomes were observed when data from open-label RCTs were considered on their own. Further data on the efficacy of TNFi therapy over ≥ 2 years in patients with methotrexate-naïve RA are required.
Plain Language Summary
Plain language summary available for this article.



Similar content being viewed by others
References
Silman A, Hochberg M. Epidemiology of the rheumatic diseases. 2nd ed. New York: Oxford University Press; 2001.
Curtis JR, Singh JA. Use of biologics in rheumatoid arthritis: current and emerging paradigms of care. Clin Ther. 2011;33:679–707.
Monaco C, Nanchahal J, Taylor P, Feldmann M. Anti-TNF therapy: past, present and future. Int Immunol. 2015;27:55–62.
Watson K, Symmons D, Griffiths I, Silman A. The British Society for Rheumatology biologics register. Ann Rheum Dis. 2005;64(Suppl 4):iv42–3.
van Nies JA, Krabben A, Schoones JW, Huizinga TW, Kloppenburg M, van der Helm-van Mil AH. What is the evidence for the presence of a therapeutic window of opportunity in rheumatoid arthritis? A systematic literature review. Ann Rheum Dis. 2014;73:861–70.
Dennison EM, Packham J, Hyrich K. The BSRBR-RA at 15 years. Rheumatology (Oxford). 2016;55:2093–5.
Singh JA, Hossain A, Mudano AS, et al. Biologics or tofacitinib for people with rheumatoid arthritis naive to methotrexate: a systematic review and network meta-analysis. Cochrane Database Syst Rev. 2017;5:CD012657.
Cai W, Gu Y, Cui H, et al. The efficacy and safety of mainstream medications for patients with DMARD-naive rheumatoid arthritis: a network meta-analysis. Front Pharmacol. 2018;9:138.
Donahue KE, Gartlehner G, Schulman ER, et al. Drug therapy for early rheumatoid arthritis: a systematic review update. Agency for Healthcare Research and Quality (US). Report No: 18-EHC015-EF. 2018.
van der Velde G, Pham B, Machado M, et al. Cost-effectiveness of biologic response modifiers compared to disease-modifying antirheumatic drugs for rheumatoid arthritis: a systematic review. Arthritis Care Res (Hoboken). 2011;63:65–78.
National Institute for Health and Care Excellence. Adalimumab, etanercept, infliximab, certolizumab pegol, golimumab, tocilizumab and abatacept for rheumatoid arthritis not previously treated with DMARDs or after conventional DMARDs only have failed. 2015. https://www.nice.org.uk/guidance/ta375/documents/rheumatoid-arthritis-adalimumab-etanercept-infliximab-certolizumab-pegol-golimumab-abatacept-and-tocilizumab-review-id537-appraisal-consultation-document2. Accessed Oct 11, 2018.
Smolen JS, Landewe R, Bijlsma J, et al. EULAR recommendations for the management of rheumatoid arthritis with synthetic and biological disease-modifying antirheumatic drugs: 2016 update. Ann Rheum Dis. 2017;76:960–77.
Putrik P, Ramiro S, Kvien TK, et al. Variations in criteria regulating treatment with reimbursed biologic DMARDs across European countries. Are differences related to country’s wealth? Ann Rheum Dis. 2014;73:2010–21.
Pentek M, Poor G, Wiland P, et al. Biological therapy in inflammatory rheumatic diseases: issues in Central and Eastern European countries. Eur J Health Econ. 2014;15(Suppl 1):S35–43.
Baji P, Pentek M, Czirjak L, et al. Efficacy and safety of infliximab-biosimilar compared to other biological drugs in rheumatoid arthritis: a mixed treatment comparison. Eur J Health Econ. 2014;15(Suppl 1):S53–64.
Brodszky V, Baji P, Balogh O, Pentek M. Budget impact analysis of biosimilar infliximab (CT-P13) for the treatment of rheumatoid arthritis in six Central and Eastern European countries. Eur J Health Econ. 2014;15(Suppl 1):S65–71.
The Center for Biosimilars. Biosimilar competition has led to consistent price reduction in Europe. 2017. https://www.centerforbiosimilars.com/news/biosimilar-competition-has-led-to-consistent-price-reduction-in-europe. Accessed Oct 11, 2018.
Haddaway NR, Collins AM, Coughlin D, Kirk S. The role of Google Scholar in evidence reviews and its applicability to grey literature searching. PLoS ONE. 2015;10:e0138237.
Hobbs KF, Cohen MD. Rheumatoid arthritis disease measurement: a new old idea. Rheumatology (Oxford). 2012;51(Suppl 6):vi21–7.
DAS28.nl. Alternative validated formulae. 2018. http://www.das-score.nl/das28/en/difference-between-the-das-and-das28/how-to-measure-the-das28/how-to-calculate-the-das28/alternative-validated-formulae.html. Accessed Aug 28, 2018.
Higgins JPT, Green S. Cochrane Handbook for Systematic Reviews of Interventions Version 5.1.0 [updated March 2011]. 2011. The Cochrane Collaboration. http://handbook.cochrane.org.
St Clair EW, van der Heijde DM, Smolen JS, et al. Combination of infliximab and methotrexate therapy for early rheumatoid arthritis: a randomized, controlled trial. Arthritis Rheum. 2004;50:3432–43.
DerSimonian R, Laird N. Meta-analysis in clinical trials. Control Clin Trials. 1986;7:177–88.
Bejarano V, Quinn M, Conaghan PG, et al. Effect of the early use of the anti-tumor necrosis factor adalimumab on the prevention of job loss in patients with early rheumatoid arthritis. Arthritis Rheum. 2008;59:1467–74.
Goekoop-Ruiterman YP, de Vries-Bouwstra JK, Allaart CF, et al. Clinical and radiographic outcomes of four different treatment strategies in patients with early rheumatoid arthritis (the BeSt study): a randomized, controlled trial. Arthritis Rheum. 2005;52:3381–90.
Goekoop-Ruiterman YP, de Vries-Bouwstra JK, Allaart CF, et al. Comparison of treatment strategies in early rheumatoid arthritis: a randomized trial. Ann Intern Med. 2007;146:406–15.
van der Kooij SM, Goekoop-Ruiterman YP, de Vries-Bouwstra JK, et al. Drug-free remission, functioning and radiographic damage after 4 years of response-driven treatment in patients with recent-onset rheumatoid arthritis. Ann Rheum Dis. 2009;68:914–21.
Klarenbeek NB, Guler-Yuksel M, van der Kooij SM, et al. The impact of four dynamic, goal-steered treatment strategies on the 5-year outcomes of rheumatoid arthritis patients in the BeSt study. Ann Rheum Dis. 2011;70:1039–46.
Markusse IM, Dirven L, Han KH, et al. Continued participation in a ten-year tight control treat-to-target study in rheumatoid arthritis: why keep patients doing their best? Arthritis Care Res (Hoboken). 2015;67:739–45.
Akdemir G, Markusse IM, Dirven L, et al. Effectiveness of four dynamic treatment strategies in patients with anticitrullinated protein antibody-negative rheumatoid arthritis: a randomised trial. RMD Open. 2016;2:e000143.
Markusse IM, Akdemir G, Dirven L, et al. Long-term outcomes of patients with recent-onset rheumatoid arthritis after 10 years of tight controlled treatment: a randomized trial. Ann Intern Med. 2016;164:523–31.
Bergstra SA, Landewe RBM, Huizinga TWJ, Allaart CF. Rheumatoid arthritis patients with continued low disease activity have similar outcomes over 10 years, regardless of initial therapy. Rheumatology (Oxford). 2017;56:1721–8.
Emery P, Bingham CO 3rd, Burmester GR, et al. Certolizumab pegol in combination with dose-optimised methotrexate in DMARD-naive patients with early, active rheumatoid arthritis with poor prognostic factors: 1-year results from C-EARLY, a randomised, double-blind, placebo-controlled phase III study. Ann Rheum Dis. 2017;76:96–104.
Weinblatt ME, Bingham CO 3rd, Burmester GR, et al. A Phase III study evaluating continuation, tapering, and withdrawal of certolizumab pegol after one year of therapy in patients with early rheumatoid arthritis. Arthritis Rheumatol. 2017;69:1937–48.
Atsumi T, Yamamoto K, Takeuchi T, et al. The first double-blind, randomised, parallel-group certolizumab pegol study in methotrexate-naive early rheumatoid arthritis patients with poor prognostic factors, C-OPERA, shows inhibition of radiographic progression. Ann Rheum Dis. 2016;75:75–83.
Atsumi T, Tanaka Y, Yamamoto K, et al. Clinical benefit of 1-year certolizumab pegol (CZP) add-on therapy to methotrexate treatment in patients with early rheumatoid arthritis was observed following CZP discontinuation: 2-year results of the C-OPERA study, a phase III randomised trial. Ann Rheum Dis. 2017;76:1348–56.
Emery P, Breedveld FC, Hall S, et al. Comparison of methotrexate monotherapy with a combination of methotrexate and etanercept in active, early, moderate to severe rheumatoid arthritis (COMET): a randomised, double-blind, parallel treatment trial. Lancet. 2008;372:375–82.
Emery P, Breedveld F, van der Heijde D, et al. Two-year clinical and radiographic results with combination etanercept-methotrexate therapy versus monotherapy in early rheumatoid arthritis: a two-year, double-blind, randomized study. Arthritis Rheum. 2010;62:674–82.
Durez P, Malghem J, Nzeusseu Toukap A, et al. Treatment of early rheumatoid arthritis: a randomized magnetic resonance imaging study comparing the effects of methotrexate alone, methotrexate in combination with infliximab, and methotrexate in combination with intravenous pulse methylprednisolone. Arthritis Rheum. 2007;56:3919–27.
Bathon JM, Martin RW, Fleischmann RM, et al. A comparison of etanercept and methotrexate in patients with early rheumatoid arthritis. N Engl J Med. 2000;343:1586–93.
Genovese MC, Bathon JM, Martin RW, et al. Etanercept versus methotrexate in patients with early rheumatoid arthritis: two-year radiographic and clinical outcomes. Arthritis Rheum. 2002;46:1443–50.
Genovese MC, Bathon JM, Fleischmann RM, et al. Long term safety, efficacy, and radiographic outcome with etanercept treatment in patients with early rheumatoid arthritis. J Rheumatol. 2005;32:1232–42.
Emery P, Fleischmann RM, Moreland LW, et al. Golimumab, a human anti-tumor necrosis factor alpha monoclonal antibody, injected subcutaneously every four weeks in methotrexate-naive patients with active rheumatoid arthritis: twenty-four-week results of a phase III, multicenter, randomized, double-blind, placebo-controlled study of golimumab before methotrexate as first-line therapy for early-onset rheumatoid arthritis. Arthritis Rheum. 2009;60:2272–83.
Emery P, Fleischmann RM, Doyle MK, et al. Golimumab, a human anti-tumor necrosis factor monoclonal antibody, injected subcutaneously every 4 weeks in patients with active rheumatoid arthritis who had never taken methotrexate: 1-year and 2-year clinical, radiologic, and physical function findings of a phase III, multicenter, randomized, double-blind, placebo-controlled study. Arthritis Care Res (Hoboken). 2013;65:1732–42.
Baker JF, Conaghan PG, Emery P, Baker DG, Ostergaard M. Validity of early MRI structural damage end points and potential impact on clinical trial design in rheumatoid arthritis. Ann Rheum Dis. 2016;75:1114–9.
Emery P, Fleischmann RM, Strusberg I, et al. Efficacy and safety of subcutaneous golimumab in methotrexate-naive patients with rheumatoid arthritis: five-year results of a randomized clinical trial. Arthritis Care Res (Hoboken). 2016;68:744–52.
Baker JF, Conaghan PG, Emery P, Baker DG, Ostergaard M. Relationship of patient-reported outcomes with MRI measures in rheumatoid arthritis. Ann Rheum Dis. 2017;76:486–90.
Soubrier M, Puechal X, Sibilia J, et al. Evaluation of two strategies (initial methotrexate monotherapy vs its combination with adalimumab) in management of early active rheumatoid arthritis: data from the GUEPARD trial. Rheumatology (Oxford). 2009;48:1429–34.
Detert J, Bastian H, Listing J, et al. Induction therapy with adalimumab plus methotrexate for 24 weeks followed by methotrexate monotherapy up to week 48 versus methotrexate therapy alone for DMARD-naive patients with early rheumatoid arthritis: HIT HARD, an investigator-initiated study. Ann Rheum Dis. 2013;72:844–50.
Yamanaka H, Ishiguro N, Takeuchi T, et al. Recovery of clinical but not radiographic outcomes by the delayed addition of adalimumab to methotrexate-treated Japanese patients with early rheumatoid arthritis: 52-week results of the HOPEFUL-1 trial. Rheumatology (Oxford). 2014;53:904–13.
Takeuchi T, Yamanaka H, Ishiguro N, et al. Adalimumab, a human anti-TNF monoclonal antibody, outcome study for the prevention of joint damage in Japanese patients with early rheumatoid arthritis: the HOPEFUL 1 study. Ann Rheum Dis. 2014;73:536–43.
Tanaka Y, Yamanaka H, Ishiguro N, et al. Low disease activity for up to 3 years after adalimumab discontinuation in patients with early rheumatoid arthritis: 2-year results of the HOPEFUL-3 Study. Arthritis Res Ther. 2017;19:56.
Nam JL, Villeneuve E, Hensor EM, et al. Remission induction comparing infliximab and high-dose intravenous steroid, followed by treat-to-target: a double-blind, randomised, controlled trial in new-onset, treatment-naive, rheumatoid arthritis (the IDEA study). Ann Rheum Dis. 2014;73:75–85.
Bissell LA, Hensor EM, Kozera L, et al. Improvement in insulin resistance is greater when infliximab is added to methotrexate during intensive treatment of early rheumatoid arthritis-results from the IDEA study. Rheumatology (Oxford). 2016;55:2181–90.
Marcora SM, Chester KR, Mittal G, Lemmey AB, Maddison PJ. Randomized phase 2 trial of anti-tumor necrosis factor therapy for cachexia in patients with early rheumatoid arthritis. Am J Clin Nutr. 2006;84:1463–72.
Leirisalo-Repo M, Kautiainen H, Laasonen L, et al. Infliximab for 6 months added on combination therapy in early rheumatoid arthritis: 2-year results from an investigator-initiated, randomised, double-blind, placebo-controlled study (the NEO-RACo Study). Ann Rheum Dis. 2013;72:851–7.
Rantalaiho V, Kautiainen H, Korpela M, et al. Targeted treatment with a combination of traditional DMARDs produces excellent clinical and radiographic long-term outcomes in early rheumatoid arthritis regardless of initial infliximab. The 5-year follow-up results of a randomised clinical trial, the NEO-RACo trial. Ann Rheum Dis. 2014;73:1954–61.
Kuusalo L, Puolakka K, Kautiainen H, et al. Impact of physicians’ adherence to treat-to-target strategy on outcomes in early rheumatoid arthritis in the NEO-RACo trial. Scand J Rheumatol. 2015;44:449–55.
Kuusalo LA, Puolakka KT, Kautiainen H, et al. Intra-articular glucocorticoid injections should not be neglected in the remission targeted treatment of early rheumatoid arthritis: a post hoc analysis from the NEO-RACo trial. Clin Exp Rheumatol. 2016;34:1038–44.
Kuusalo L, Puolakka K, Kautiainen H, et al. Patient-reported outcomes as predictors of remission in early rheumatoid arthritis patients treated with tight control treat-to-target approach. Rheumatol Int. 2017;37:825–30.
Vaananen T, Vuolteenaho K, Kautiainen H, et al. Glycoprotein YKL-40: a potential biomarker of disease activity in rheumatoid arthritis during intensive treatment with csDMARDs and infliximab.Evidence from the randomised controlled NEO-RACo trial. PLoS ONE. 2017;12:0183294.
Kuusalo L, Puolakka K, Kautiainen H, et al. High burden of adverse events is associated with reduced remission rates in early rheumatoid arthritis. Clin Rheumatol. 2018;37:1689–94.
Horslev-Petersen K, Hetland ML, Junker P, et al. Adalimumab added to a treat-to-target strategy with methotrexate and intra-articular triamcinolone in early rheumatoid arthritis increased remission rates, function and quality of life. The OPERA Study: an investigator-initiated, randomised, double-blind, parallel-group, placebo-controlled trial. Ann Rheum Dis. 2014;73:654–61.
Andersen T, Hvid M, Johansen C, et al. Interleukin-23 in early disease development in rheumatoid arthritis. Scand J Rheumatol. 2015;44:438–42.
Axelsen MB, Eshed I, Horslev-Petersen K, et al. A treat-to-target strategy with methotrexate and intra-articular triamcinolone with or without adalimumab effectively reduces MRI synovitis, osteitis and tenosynovitis and halts structural damage progression in early rheumatoid arthritis: results from the OPERA randomised controlled trial. Ann Rheum Dis. 2015;74:867–75.
Greisen SR, Moller HJ, Stengaard-Pedersen K, et al. Macrophage activity assessed by soluble CD163 in early rheumatoid arthritis: association with disease activity but different response patterns to synthetic and biologic DMARDs. Clin Exp Rheumatol. 2015;33:498–502.
Horslev-Petersen K, Hetland ML, Ornbjerg LM, et al. Clinical and radiographic outcome of a treat-to-target strategy using methotrexate and intra-articular glucocorticoids with or without adalimumab induction: a 2-year investigator-initiated, double-blinded, randomised, controlled trial (OPERA). Ann Rheum Dis. 2016;75:1645–53.
Kragstrup TW, Greisen SR, Nielsen MA, et al. The interleukin-20 receptor axis in early rheumatoid arthritis: novel links between disease-associated autoantibodies and radiographic progression. Arthritis Res Ther. 2016;18:61.
Krintel SB, Dehlendorff C, Hetland ML, et al. Prediction of treatment response to adalimumab: a double-blind placebo-controlled study of circulating microRNA in patients with early rheumatoid arthritis. Pharmacogenomics J. 2016;16:141–6.
Heftdal LD, Stengaard-Pedersen K, Ornbjerg LM, et al. Soluble CD206 plasma levels in rheumatoid arthritis reflect decrease in disease activity. Scand J Clin Lab Invest. 2017;77:385–9.
Kristensen AM, Stengaard-Pedersen K, Hetland ML, et al. Expression of soluble CD83 in plasma from early-stage rheumatoid arthritis patients is not modified by anti-TNF-alpha therapy. Cytokine. 2017;96:1–7.
Ornbjerg LM, Ostergaard M, Jensen T, et al. Hand bone loss in early rheumatoid arthritis during a methotrexate-based treat-to-target strategy with or without adalimumab-a substudy of the optimized treatment algorithm in early RA (OPERA) trial. Clin Rheumatol. 2017;36:781–9.
Brahe CH, Ostergaard M, Johansen JS, et al. Predictive value of a multi-biomarker disease activity score for clinical remission and radiographic progression in patients with early rheumatoid arthritis: a post hoc study of the OPERA trial. Scand J Rheumatol. 2018. https://doi.org/10.1080/03009742.2018.1464206.
Sode J, Krintel SB, Carlsen AL, et al. Plasma microRNA profiles in patients with early rheumatoid arthritis responding to adalimumab plus methotrexate vs methotrexate alone: a placebo-controlled clinical trial. J Rheumatol. 2018;45:53–61.
Smolen JS, Emery P, Fleischmann R, et al. Adjustment of therapy in rheumatoid arthritis on the basis of achievement of stable low disease activity with adalimumab plus methotrexate or methotrexate alone: the randomised controlled OPTIMA trial. Lancet. 2014;383:321–32.
Kavanaugh A, Fleischmann RM, Emery P, et al. Clinical, functional and radiographic consequences of achieving stable low disease activity and remission with adalimumab plus methotrexate or methotrexate alone in early rheumatoid arthritis: 26-week results from the randomised, controlled OPTIMA study. Ann Rheum Dis. 2013;72:64–71.
Emery P, Smolen JS, Ganguli A, et al. Effect of adalimumab on the work-related outcomes scores in patients with early rheumatoid arthritis receiving methotrexate. Rheumatology (Oxford). 2016;55:1458–65.
Breedveld FC, Weisman MH, Kavanaugh AF, et al. The PREMIER study: a multicenter, randomized, double-blind clinical trial of combination therapy with adalimumab plus methotrexate versus methotrexate alone or adalimumab alone in patients with early, aggressive rheumatoid arthritis who had not had previous methotrexate treatment. Arthritis Rheum. 2006;54:26–37.
van der Heijde D, Breedveld FC, Kavanaugh A, et al. Disease activity, physical function, and radiographic progression after longterm therapy with adalimumab plus methotrexate: 5-year results of PREMIER. J Rheumatol. 2010;37:2237–46.
Strand V, Rentz AM, Cifaldi MA, Chen N, Roy S, Revicki D. Health-related quality of life outcomes of adalimumab for patients with early rheumatoid arthritis: results from a randomized multicenter study. J Rheumatol. 2012;39:63–72.
Landewe R, Smolen JS, Florentinus S, Chen S, Guerette B, van der Heijde D. Existing joint erosions increase the risk of joint space narrowing independently of clinical synovitis in patients with early rheumatoid arthritis. Arthritis Res Ther. 2015;17:133.
Stephens S, Botteman MF, Cifaldi MA, van Hout BA. Modelling the cost-effectiveness of combination therapy for early, rapidly progressing rheumatoid arthritis by simulating the reversible and irreversible effects of the disease. BMJ Open. 2015;5:e006560.
Moller B, Everts-Graber J, Florentinus S, Li Y, Kupper H, Finckh A. Low hemoglobin and radiographic damage progression in early rheumatoid arthritis: secondary analysis from a phase III trial. Arthritis Care Res (Hoboken). 2018;70:861–8.
Quinn MA, Conaghan PG, O’Connor PJ, et al. Very early treatment with infliximab in addition to methotrexate in early, poor-prognosis rheumatoid arthritis reduces magnetic resonance imaging evidence of synovitis and damage, with sustained benefit after infliximab withdrawal: results from a twelve-month randomized, double-blind, placebo-controlled trial. Arthritis Rheum. 2005;52:27–35.
clinicaltrials.gov. NCT03492658. 2018. https://clinicaltrials.gov/ct2/show/NCT03492658. Accessed July 25, 2018.
clinicaltrials.gov. NCT02504268. 2017. https://clinicaltrials.gov/ct2/show/NCT02504268. Accessed July 15, 2018.
clinicaltrials.gov. NCT00901550. 2012. https://clinicaltrials.gov/ct2/show/NCT00901550. Accessed July 15, 2018.
clinicaltrials.gov. NCT00480272. 2017. https://clinicaltrials.gov/ct2/show/NCT00480272. Accessed July 15, 2018.
clinicaltrials.gov. NCT03160001. 2018. https://clinicaltrials.gov/ct2/show/NCT03160001. Accessed July 15, 2018.
clinicaltrials.gov. NCT01491815. 2018. https://clinicaltrials.gov/ct2/show/NCT01491815. Accessed July 15, 2018.
clinicaltrials.gov. NCT02935387. 2018. https://clinicaltrials.gov/ct2/show/NCT02935387. Accessed July 15, 2018.
clinicaltrials.gov. NCT00523692. 2007. https://clinicaltrials.gov/ct2/show/NCT00523692. Accessed July 15, 2018.
clinicaltrials.gov. NCT01245452. 2013. https://clinicaltrials.gov/ct2/show/NCT01245452. Accessed July 15, 2018.
clinicaltrials.gov. NCT02837146. 2016. https://clinicaltrials.gov/ct2/show/NCT02837146. Accessed July 15, 2018.
clinicaltrialregister.eu. EUCTR2011-004017-17-GB. 2013. https://www.clinicaltrialsregister.eu/ctr-search/trial/2011-004017-17/GB. Accessed July 15, 2018.
clinicaltrialregister.eu. EUCTR2010-023910-30-GB. 2011. https://www.clinicaltrialsregister.eu/ctr-search/trial/2010-023910-30/GB. Accessed July 15, 2018.
cris.nih.go.kr. KCT0000089. https://cris.nih.go.kr/cris/en/search/search_result_st01.jsp?seq=1201. Accessed July 15, 2018.
isrctn.com. ISRCTN49682259. 2015. http://www.isrctn.com/ISRCTN49682259?q=&filters=conditionCategory:Musculoskeletal%20Diseases. Accessed July 15, 2018.
Albert DA. Are all biologics the same? Optimal treatment strategies for patients with early rheumatoid arthritis: systematic review and indirect pairwise meta-analysis. J Clin Rheumatol. 2015;21:398–404.
Hifinger M, Hiligsmann M, Ramiro S, et al. Economic considerations and patients’ preferences affect treatment selection for patients with rheumatoid arthritis: a discrete choice experiment among European rheumatologists. Ann Rheum Dis. 2017;76:126–32.
Taylor PC, Alten R, Reino JJG, et al. Factors influencing the use of biologic therapy and adoption of treat-to-target recommendations in current European rheumatology practice. Patient Prefer Adherence. 2018;12:2007–14.
Nagy G, van Vollenhoven RF. Sustained biologic-free and drug-free remission in rheumatoid arthritis, where are we now? Arthritis Res Ther. 2015;17:181.
Nam JL, Villeneuve E, Hensor EM, et al. A randomised controlled trial of etanercept and methotrexate to induce remission in early inflammatory arthritis: the EMPIRE trial. Ann Rheum Dis. 2014;73:1027–36.
Stamm TA, Machold KP, Aletaha D, et al. Induction of sustained remission in early inflammatory arthritis with the combination of infliximab plus methotrexate: the DINORA trial. Arthritis Res Ther. 2018;20:174.
Combe B, Landewe R, Daien CI, et al. 2016 update of the EULAR recommendations for the management of early arthritis. Ann Rheum Dis. 2017;76:948–59.
Stevenson M, Archer R, Tosh J, et al. Adalimumab, etanercept, infliximab, certolizumab pegol, golimumab, tocilizumab and abatacept for the treatment of rheumatoid arthritis not previously treated with disease-modifying antirheumatic drugs and after the failure of conventional disease-modifying antirheumatic drugs only: systematic review and economic evaluation. Health Technol Assess. 2016;20:1–610.
Dorner T, Strand V, Cornes P, et al. The changing landscape of biosimilars in rheumatology. Ann Rheum Dis. 2016;75:974–82.
Acknowledgements
Funding
The authors received no funding for the analyses reported here. The article processing charges were funded by Celltrion Healthcare Co., Ltd (Incheon, Republic of Korea).
At the time of publication of the current article, the authors are performing additional work related to the analyses reported here; funding for this work will be provided by Celltrion Healthcare Co., Ltd.
Authorship
All named authors meet the International Committee of Medical Journal Editors (ICMJE) criteria for authorship for this manuscript, take responsibility for the integrity of the work as a whole, and have given final approval for the article to be published.
Medical Writing, Editorial, and Other Assistance
Medical writing support for this article (including copyediting and fact checking) was provided to the authors by Rick Flemming, PhD CMPP and Emma Evans, PhD CMPP at Aspire Scientific Limited (Bollington, UK) and was funded by Celltrion Healthcare Co., Ltd.
Disclosures
László Gulácsi has received consultancy and lecturing fees from Astellas, BMS, Celltrion, Egis Pharmaceuticals, GSK, Hikma, Hospira, Lilly Hungaria Ltd, MSD Hungary, Pfizer, Roche, Sandoz and UCB. Zsombor Zrubka used to be a full-time employee of Egis Pharmaceuticals, Janssen Cilag, Sandoz and Pfizer. Valentin Brodszky has received grants and personal fees from Celltrion, Egis Pharmaceuticals, Pfizer and Sager Pharma. Fanni Rencz has received consultancy fees from Celltrion and Hospira. Rieke Alten has received honoraria from Celltrion. Zoltán Szekanecz has received consultancy and lecturing fees from AbbVie, Amgen, BMS, Lilly, MSD, Novartis, Pfizer, Roche and UCB. Márta Péntek has received grants and personal fees from Celltrion, Egis Pharmaceuticals, Merck, Pfizer and Sager Pharma.
Compliance with Ethics Guidelines
This article does not contain any new studies with human or animal subjects performed by any of the authors.
Data Availability
All data used in this systematic review and meta-analysis are available in the published sources.
Author information
Authors and Affiliations
Corresponding author
Additional information
Enhanced Digital Features
To view enhanced digital features for this article go to https://doi.org/10.6084/m9.figshare.7498988.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Gulácsi, L., Zrubka, Z., Brodszky, V. et al. Long-Term Efficacy of Tumor Necrosis Factor Inhibitors for the Treatment of Methotrexate-Naïve Rheumatoid Arthritis: Systematic Literature Review and Meta-Analysis. Adv Ther 36, 721–745 (2019). https://doi.org/10.1007/s12325-018-0869-8
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12325-018-0869-8